Physicochemical Properties
| Molecular Formula | C21H24O6 |
| Molecular Weight | 372.41200 |
| Exact Mass | 372.157 |
| CAS # | 52387-14-1 |
| Related CAS # | 76549-35-4 |
| PubChem CID | 479497 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.246g/cm3 |
| Boiling Point | 570.1ºC at 760 mmHg |
| Melting Point | 107ºC |
| Flash Point | 198.6ºC |
| Index of Refraction | 1.578 |
| LogP | 3.717 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 27 |
| Complexity | 656 |
| Defined Atom Stereocenter Count | 1 |
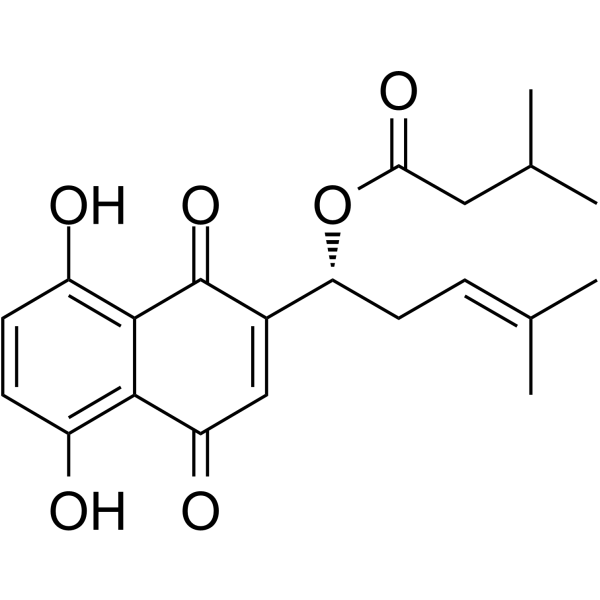
| SMILES | CC(C)CC(=O)OC(CC=C(C)C)C1=CC(=O)C2=C(C=CC(=C2C1=O)O)O |
| InChi Key | UTOUNDHZJFIVPK-QGZVFWFLSA-N |
| InChi Code | InChI=1S/C21H24O6/c1-11(2)5-8-17(27-18(25)9-12(3)4)13-10-16(24)19-14(22)6-7-15(23)20(19)21(13)26/h5-7,10,12,17,22-23H,8-9H2,1-4H3/t17-/m1/s1 |
| Chemical Name | [(1R)-1-(5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl] 3-methylbutanoate |
| Synonyms | Isovalerylshikonin; 76549-35-4; [1-(5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl] 3-methylbutanoate; NSC344556; 52387-14-1; Butanoic acid, 3-methyl-, 1-(1,4-dihydro-5,8-dihydroxy-1,4-dioxo-2-naphthalenyl)-4-methyl-3-pentenyl ester; Alkannin isovalerate; SCHEMBL13389448; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural product |
| ln Vitro |
Antibacterial minimum inhibitory concentration assay and synergistic effects [1] IVS/Isovalerylshikonin was isolated from A. euchroma with 98.7% purity (Supplementary Fig. S1) and showed marginal antibacterial activity against the drug-resistant test strain S. aureus RN4220 with an MIC of 16 mg/L (Table 1; Supplementary Fig. S2). As a positive control, the antibiotic vancomycin inhibited the growth of strain RN4220 with an MIC of 2 mg/L. Growth kinetics, ethidium bromide efflux assay and effect of isovalerylshikonin on induction of msrA at the transcriptional level [1] As shown in Supplementary Fig. S3, the growth curves of bacteria treated with the vehicle or with 0.25 × MIC of Isovalerylshikonin/IVS almost overlapped. The growth of strain RN4220 treated with 0.25 × MIC of STM was almost completely halted within 12 h and grew quickly thereafter. Compared with STM alone, strain RN4220 treated with STM (0.25 × MIC) combined with IVS (0.25 × MIC) grew at a slower rate. Thus, a synergistic effect between STM and IVS against strain RN4220 was observed. Ethidium bromide was quickly depleted to <50% by vehicle-treated bacteria from 1–60 min, followed by a slower reduction, and the final level of fluorescence was ca. 30% (Fig. 1). IVS inhibited bacterial efflux against strain RN4220 (P < 0.05), although this inhibition was weaker than in the positive control carbonyl cyanide m-chlorophenylhydrazone. Expression of msrA mRNA in bacteria treated with vehicle was set as the reference level of induction (1-fold). Strain RN4220 was incubated with STM (32 mg/L, 0.0125 × MIC) resulting in substantial expression of msrA mRNA, which was decreased significantly when the bacteria were incubated with STM and Isovalerylshikonin/IVS (P < 0.01). However, the level of mRNA expression in bacteria treated with STM combined with IVS was higher than in those treated with the vehicle (Supplementary Fig. S4). |
| ln Vivo |
In vivo infection and acute toxicity studies [1] The 7-day survival curve of mice infected with strain RN4220 was recorded. Vehicle group mice died within 24 h of infection, as did those treated with Isovalerylshikonin/IVS (40 mg/kg) alone or with STM (10 mg/kg) alone (Fig. 2). Most mice administered a combination of STM (10 mg/kg) and IVS (10 mg/kg) died within 24 h of infection, with a survival percentage of 20%, and the rest died within the following 24 h. Eight of the combination group mice treated with the combination of STM (10 mg/kg) and IVS (20 mg/kg) died within 2 days (48 h) of being infected by strain RN4220, such that the final 7-day survival percentage of this group was 10%. Combination group mice administered STM (10 mg/kg) and a high dose of IVS (40 mg/kg) died slowly within 24 h of infection, and six died within 3 days of infection, with a final 7-day survival percentage of 40%. Mice in the positive control group injected with vancomycin (110 mg/kg) had a 60% survival rate at 7 days of infection with strain RN4220. Isovalerylshikonin/IVS significantly suppressed bacterial levels in infected mice (Supplementary Fig. S6), increasing the in vivo antibacterial activity of STM. The acute toxicity of IVS in mice was measured and it was found to have a 50% lethal dose (LD50) of 2.584 g/kg, indicating that IVS is a low-toxicity compound (Supplementary Fig. S8). |
| Enzyme Assay |
Growth kinetics, ethidium bromide efflux assay, total RNA extraction and real-time PCR [1] Growth kinetics of strain RN4220 were determined as previously described with some modifications. Isovalerylshikonin/IVS was evaluated in a bacterial efflux assay as described previously. Total RNA was isolated from bacteria using TRIzol reagent according to the manufacturer's instructions. The reverse transcription step was carried out using a RevertAidTM First Strand cDNA Synthesis Kit to synthesise cDNA. PCR analysis was performed using an ABI 7300 real-time fluorescent quantitative PCR system. Detailed methods are provided in the Supplementary material. |
| Animal Protocol |
In vivo infection and acute toxicity studies [1] In vivo infection and acute toxicity studies were performed according to previously published methods, with some modifications. |
| Toxicity/Toxicokinetics | Investigation of efficacy and safety in animal models is important in drug development. The synergistic effect between Isovalerylshikonin/IVS and STM against drug-resistant S. aureus infection was shown in vivo in a mouse model. The acute toxicity of IVS with a single exposure in mice was measured with an LD50 of 2.584 g/kg in present study (Supplementary material).[1] |
| References |
[1].Isovalerylshikonin, a new resistance-modifying agent from Arnebia euchroma, supresses antimicrobial resistance of drug-resistant Staphylococcus aureus. Int J Antimicrob Agents. 2019 Jan;53(1):70-73. [2].Use of matrix‐assisted laser desorption/ionization mass spectrometry for the rapid detection of low‐mass components in the Alkanna tinctoria pigments fraction. Journal of mass spectrometry, 1998, 33(1): 89-91. |
| Additional Infomation |
(R)-1-(5,8-Dihydroxy-1,4-dioxo-1,4-dihydro-2-naphthyl)-4-methyl-3-pentenyl isovalerate has been reported in Lithospermum erythrorhizon with data available.
Isovalerylshikonin has been reported in Onosma heterophylla with data available. Antimicrobial resistance is the greatest threat to the treatment of bacterial infectious diseases. The development of resistance-modifying agents (RMAs) represents a promising strategy to mitigate the spread of bacterial antimicrobial resistance. In this study, a natural product, isovalerylshikonin (IVS), was isolated from Arnebia euchroma, a traditional Chinese medicine herb, that exhibited marginal antibacterial activity against drug-resistant Staphylococcus aureus RN4220, with a minimum inhibitory concentration (MIC) of 16 mg/L. In addition, a synergistic effect between IVS and streptomycin (STM) was detected by the microdilution antimicrobial chequerboard assay, with a reduction in the MIC of STM by up to 16-fold against strain RN4220. A bacterial ethidium bromide efflux assay and reverse transcription PCR were performed to investigate the synergistic mechanism. IVS significantly inhibited bacterial efflux and expression of msrA mRNA in vitro. A murine peritonitis/sepsis model was employed to test the in vivo synergistic activity of IVS and STM. IVS synergistically decreased bacterial counts with STM in peritoneal, spleen and liver tissue and increased mouse survival with STM in 7 days. The acute toxicity of IVS was tested and the 50% lethal dose (LD50) of IVS with a single exposure was 2.584 g/kg in mice. Overall, IVS, a low-toxicity RMA, exhibited synergistic antibacterial activities in vitro and in vivo against drug-resistant S. aureus. The effects were mediated by suppression of msrA mRNA expression and reduced bacterial efflux. In addition, these data support that IVS is a potential RMA against microbial resistance caused by the MsrA efflux pump.[1] This study focused on the discovery of a new RMA against the MsrA efflux pump. IVS isolated from A. euchroma was shown to be an effective RMA against microbial resistance caused by the MsrA efflux pump, exhibiting in vitro and in vivo synergistic activities against S. aureus RN4220. The effects were mediated by suppression of msrA expression at the mRNA level and by a reduction of bacterial efflux. In addition, IVS is a low toxicity agent with an LD50 of 2.584 g/kg in mice and may serve as a potential agent for therapeutic use in infections.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6852 mL | 13.4261 mL | 26.8521 mL | |
| 5 mM | 0.5370 mL | 2.6852 mL | 5.3704 mL | |
| 10 mM | 0.2685 mL | 1.3426 mL | 2.6852 mL |