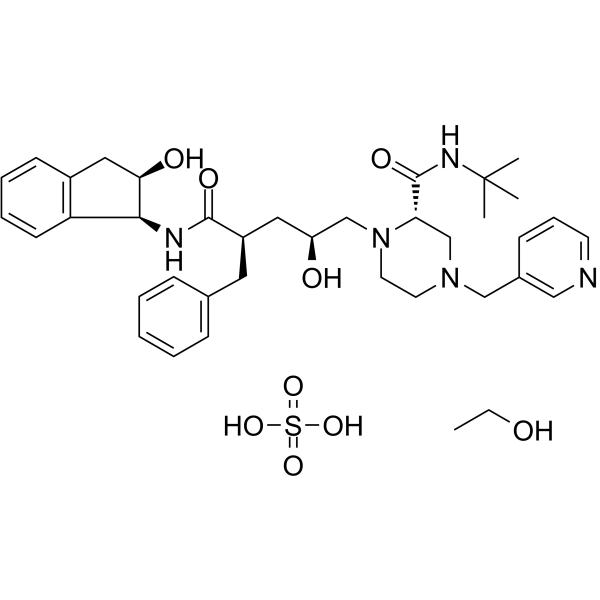
Indinavir sulfate ethanolate (MK-639 ethanolate) is an orally bioactive and selective HIV-1 protease inhibitor with antiviral and anticancer activity. It inhibits protease with a Ki of 0.54 nM. Indinavir sulfate ethanolate is also a SARS-CoV 3CLpro inhibitor.
Physicochemical Properties
| Molecular Formula | C38H55N5O9S |
| Molecular Weight | 757.94 |
| Exact Mass | 757.372 |
| CAS # | 2563866-80-6 |
| Related CAS # | Indinavir sulfate;157810-81-6;Indinavir;150378-17-9 |
| PubChem CID | 11954281 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 53 |
| Complexity | 1040 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | CCO.CC(C)(C)NC(=O)[C@@H]1CN(CCN1C[C@H](C[C@@H](CC2=CC=CC=C2)C(=O)N[C@@H]3[C@@H](CC4=CC=CC=C34)O)O)CC5=CN=CC=C5.OS(=O)(=O)O |
| InChi Key | QDNVAYDEAGXHTB-NOYQBWMBSA-N |
| InChi Code | InChI=1S/C36H47N5O4.C2H6O.H2O4S/c1-36(2,3)39-35(45)31-24-40(22-26-12-9-15-37-21-26)16-17-41(31)23-29(42)19-28(18-25-10-5-4-6-11-25)34(44)38-33-30-14-8-7-13-27(30)20-32(33)43;1-2-3;1-5(2,3)4/h4-15,21,28-29,31-33,42-43H,16-20,22-24H2,1-3H3,(H,38,44)(H,39,45);3H,2H2,1H3;(H2,1,2,3,4)/t28-,29+,31+,32-,33+;;/m1../s1 |
| Chemical Name | (2S)-1-[(2S,4R)-4-benzyl-2-hydroxy-5-[[(1S,2R)-2-hydroxy-2,3-dihydro-1H-inden-1-yl]amino]-5-oxopentyl]-N-tert-butyl-4-(pyridin-3-ylmethyl)piperazine-2-carboxamide;ethanol;sulfuric acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In PBMC cells, indinavir sulfate ethanolate (0-50 µM; 18 h) inhibits the lymphocyte cell cycle in the G0/G1 phase and reduces the ability of the cells to proliferate [1]. In vitro, indinavir sulfate ethanolate (40 µM–40 nM; 5 days) inhibits Huh7 and SK-HEP-1 hepatocarcinoma cells' ability to invade cells and activate MMPs-2 (40 µM–40 nM; 48 h)[2]. |
| ln Vivo | Hepatocarcinoma cell growth is inhibited in vivo by indinavir sulfate ethanolate (70 mg/kg; ig; once daily for 3 weeks)[2]. |
| Cell Assay |
Cell Viability Assay[1] Cell Types: PBMCs (from healthy and HIV-infected volunteers) Tested Concentrations: 0-50 µM Incubation Duration: 18 h (pretreatment; stimulation with anti-CD3 for an additional 48 hrs (hours)) Experimental Results: Blocked anti-CD3-induced cell-cycle progression in a dose-dependent manner. Resulted in dose-dependent reduction of lymphoproliferative responses. Cell Invasion Assay[2] Cell Types: Huh7 and SK-HEP-1 cells Tested Concentrations: 40 µM-40 nM Incubation Duration: 5 days Experimental Results: decreased ability to invade an in vitro constituted extracellular matrix for both cell lines treated compared with the untreated cells. Western Blot Analysis[2] Cell Types: Huh7 and SK-HEP-1 cells Tested Concentrations: 40 µM-40 nM Incubation Duration: 48 h Experimental Results: Blocked the conversion of latent MMP-2 to its 62/64- kDa active form. |
| Animal Protocol |
Animal/Disease Models: Nude mice(sc into Huh7 and SK-HEP-1 cells)[2] . Doses: 70 mg/kg Route of Administration: po (oral gavage); one time/day for 3 weeks Experimental Results: Delaied the growth of sc implanted hepatocarcinoma xenografts in nude mice compared with placebo. |
| References |
[1]. The HIV protease inhibitor Indinavir inhibits cell-cycle progression in vitro in lymphocytes of HIV-infected and uninfected individuals. Blood. 2001 Jul 15;98(2):383-9. [2]. Evaluation of antitumoral properties of the protease inhibitor indinavir in a murine model of hepatocarcinoma. Clin Cancer Res. 2006 Apr 15;12(8):2634-9. [3]. Kinetic, stability, and structural changes in high-resolution crystal structures of HIV-1 protease with drug-resistant mutations L24I, I50V, and G73S. J Mol Biol. 2005 Dec 9;354(4):789-800. [4]. A search for medications to treat COVID-19 via in silico molecular docking models of the SARS-CoV-2 spike glycoprotein and 3CL protease. Travel Med Infect Dis. 2020 May-Jun;35:101646. |
| Additional Infomation |
Drug Indication Crixivan is indicated in combination with antiretroviral nucleoside analogues for the treatment of HIV-1 infected adults. , |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3194 mL | 6.5968 mL | 13.1937 mL | |
| 5 mM | 0.2639 mL | 1.3194 mL | 2.6387 mL | |
| 10 mM | 0.1319 mL | 0.6597 mL | 1.3194 mL |