Imepitoin, formerly known as AWD 131-138 and ELB 138, is a novel partial BZD (benzodiazepine) receptor agonist that has the potential use as an antiepileptic drug for the treatment of canine idiopathic epilepsy. Imepitoin is a centrally acting anti-epileptic which crosses the blood brain barrier. It inhibits seizures via potentiation of the GABA a receptor-mediated inhibitory effects on the neurones. Imepitoin exerts significant anticonvulsant efficacy without tolerance in a dog seizure model as well as in epileptic dogs with spontaneously recurrent seizures. These data thus substantiate that partial agonism at the BZD site of GABAA receptors offers advantages versus full agonism and constitutes a valuable approach for treatment of seizures.
Physicochemical Properties
| Molecular Formula | C13H14CLN3O2 | |
| Molecular Weight | 279.08 | |
| Exact Mass | 279.077 | |
| CAS # | 188116-07-6 | |
| Related CAS # |
|
|
| PubChem CID | 3083511 | |
| Appearance | White to off-white solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 421.8±55.0 °C at 760 mmHg | |
| Flash Point | 208.9±31.5 °C | |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C | |
| Index of Refraction | 1.664 | |
| LogP | 0.04 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 2 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 19 | |
| Complexity | 374 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | IQHYCZKIFIHTAI-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C13H14ClN3O2/c14-10-1-3-11(4-2-10)17-9-12(15-13(17)18)16-5-7-19-8-6-16/h1-4H,5-9H2 | |
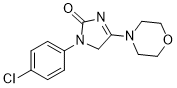
| Chemical Name | 3-(4-chlorophenyl)-5-morpholin-4-yl-4H-imidazol-2-one | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
GABAₐ receptor (benzodiazepine binding site): recombinant rat GABAₐ receptor isoforms (α1β2γ2, α1β3γ2, α2β2γ2, α3β2γ2, α5β2γ2), low-affinity partial agonist[1] Benzodiazepine receptor (central nervous system): low-affinity partial agonist [2][3] |
||
| ln Vitro |
Recombinant gamma-aminobutyric acid A (GABA(A)) receptors of the subunit compositions alpha1beta2gamma2, alpha1beta3gamma2, alpha2beta2gamma2, alpha3beta2gamma2, and alpha5beta2gamma2) were dose-dependently stimulated GABA currents by AWD 131–138. This allosteric stimulation amounted, on average, to roughly 12–21% of the maximal stimulation attained with diazepam at 10 microM AWD 131–138. The stimulation threshold ranged from 0.3 to 1.0 microM [1]. 1. GABAₐ receptor activation: Imepitoin (formerly AWD 131-138/ELB 138) dose-dependently stimulated GABA currents in Xenopus oocytes expressing recombinant rat GABAₐ receptors of subunit compositions α1β2γ2, α1β3γ2, α2β2γ2, α3β2γ2 and α5β2γ2; at 10 μM, the allosteric stimulation reached 12-21% of the maximal stimulation induced by diazepam, with a stimulation threshold of 0.3-1.0 μM; 1 μM flumazenil (benzodiazepine antagonist) counteracted the current stimulation by 10 μM Imepitoin, confirming its action at the benzodiazepine binding site of GABAₐ receptors [1] |
||
| ln Vivo |
At cumulative doses up to 18.0 mg/kg im (plasma levels over 2100 ng/ml), AWD 131–138 did not change response rates or cause midazolam-like responding. Within three sessions of studying AWD 131-138 (10-100 microg/kg/injection) by substitution, responding decreased to vehicle substitution levels. Sufficient drug was self-administered during the first session (approximately 3.5 mg/kg) at the dose of 100 microg/kg iv AWD 131-138 to produce plasma levels above 1000 ng/ml; however, responding dropped to vehicle levels over the next two sessions [2]. In the PTZ dog model, prolonged oral administration of ELB 138 at twice-daily doses of either 5 or 40 mg/kg for a duration of 5 weeks was not linked to a decrease in the drug's anticonvulsant efficacy [3]. 1. Discriminative stimulus and reinforcing effects (squirrel monkeys): Imepitoin (up to 18.0 mg/kg i.m., plasma levels over 2100 ng/ml) did not produce midazolam-like discriminative responding or alter response rates in squirrel monkeys trained to discriminate midazolam (0.3 mg/kg i.m.) from vehicle; in monkeys trained to self-administer cocaine (56.0 μg/kg/injection), substitution with Imepitoin (10-100 μg/kg/injection i.v.) led to responding declining to vehicle levels within three sessions; at 100 μg/kg i.v., monkeys self-administered ~3.5 mg/kg of Imepitoin in the first session (plasma levels >1000 ng/ml), but responding dropped to vehicle levels in subsequent sessions, indicating low abuse liability [2] 2. Anticonvulsant efficacy (dogs): Imepitoin potently increased the pentylenetetrazole (PTZ) seizure threshold in dogs; twice-daily oral administration of Imepitoin (5 or 40 mg/kg) for 5 weeks did not cause loss of anticonvulsant efficacy in the PTZ seizure model; injection of flumazenil after 5 weeks of Imepitoin treatment precipitated only mild abstinence symptoms (especially at 5 mg/kg b.i.d.) compared with diazepam; in newly diagnosed epileptic dogs, Imepitoin markedly reduced seizure frequency and severity, with efficacy comparable to phenobarbital/primidone but better tolerability; in chronic epileptic dogs, add-on treatment with Imepitoin reduced seizure frequency and severity in most cases [3] |
||
| Enzyme Assay |
1. Recombinant rat GABAₐ receptor activity assay: Xenopus oocytes were injected with cRNA encoding different recombinant rat GABAₐ receptor subunit combinations (α1β2γ2, α1β3γ2, α2β2γ2, α3β2γ2, α5β2γ2) to express functionally active receptors; the oocytes were exposed to varying concentrations of Imepitoin (0.3-10 μM) in the presence of GABA, and GABA-induced chloride currents were recorded using electrophysiological techniques to evaluate the allosteric stimulation effect of Imepitoin; to confirm the binding site, 1 μM flumazenil was co-administered with 10 μM Imepitoin, and current changes were measured [1] |
||
| Cell Assay |
1. Xenopus oocyte heterologous expression assay: Xenopus oocytes were isolated and prepared for heterologous expression of recombinant rat GABAₐ receptors; cRNA for different GABAₐ receptor subunit combinations (α1β2γ2, α1β3γ2, α2β2γ2, α3β2γ2, α5β2γ2) was microinjected into the oocytes; after a period of incubation to allow receptor expression, the oocytes were placed in a recording chamber, and GABA currents were measured using the two-electrode voltage-clamp technique; different concentrations of Imepitoin were applied to the oocytes to assess dose-dependent stimulation of GABA currents, and the antagonistic effect of flumazenil was also tested [1] |
||
| Animal Protocol |
|
||
| ADME/Pharmacokinetics |
1. Plasma concentration (squirrel monkeys): After intramuscular administration of Imepitoin (up to 18.0 mg/kg) to squirrel monkeys, plasma levels exceeded 2100 ng/ml; after intravenous self-administration of 100 μg/kg/injection (total ~3.5 mg/kg in the first session), plasma levels were above 1000 ng/ml [2] |
||
| Toxicity/Toxicokinetics |
1. Withdrawal symptoms (dogs): After 5 weeks of twice-daily oral administration of Imepitoin (5 or 40 mg/kg), injection of flumazenil precipitated only mild abstinence symptoms, which were significantly less severe than those induced by prolonged diazepam treatment (especially at 5 mg/kg b.i.d.) [3] 2. Behavioral toxicity (squirrel monkeys): Imepitoin (up to 18.0 mg/kg i.m.) did not alter response rates in discriminative stimulus assays, indicating no significant psychomotor toxicity; no self-administration behavior was observed, suggesting low abuse liability [2] 3. Tolerability (dogs): Imepitoin was much better tolerated than phenobarbital/primidone in newly diagnosed epileptic dogs, with no significant adverse effects reported [3] |
||
| References |
[1]. The antiepileptic drug AWD 131-138 stimulates different recombinant isoforms of the rat GABA(A) receptor through the benzodiazepine binding site. Neurosci Lett. 1998 Apr 3;245(2):85-8. [2]. Evaluation of the novel antiepileptic drug, AWD 131-138, for benzodiazepine-like discriminative stimulus and reinforcing effects in squirrel monkeys. Eur J Pharmacol. 2003 Apr 4;465(3):257-65. [3]. Anticonvulsant efficacy of the low-affinity partial benzodiazepine receptor agonist ELB 138 in a dog seizure model and in epileptic dogs with spontaneously recurrent seizures. Epilepsia. 2004 Oct;45(10):1228-39. |
||
| Additional Infomation |
Drug Indication Meeting highlights from the Committee for Medicinal Products for Veterinary Use (CVMP) 16-17 February 202119/02/2021 1. Chemical identity: Imepitoin (former names: AWD 131-138, ELB 138) has the chemical structure 1-(4-chlorophenyl)-4-morpholino-imidazolin-2-one [2][3] 2. Mechanism of action: Imepitoin is a low-affinity partial agonist at the benzodiazepine binding site of GABAₐ receptors, exerting allosteric stimulation of GABA currents without the functional uncoupling between GABAₐ and benzodiazepine sites caused by full agonists (e.g., diazepam) [1][3] 3. Therapeutic potential: Imepitoin has potent anticonvulsant efficacy in dog seizure models and epileptic dogs, with no tolerance development during long-term administration; it has low abuse liability and better tolerability than conventional antiepileptic drugs (phenobarbital/primidone), making it a valuable option for epilepsy treatment [2][3] 4. Clinical limitation context: Full benzodiazepine agonists (e.g., diazepam) have limitations including ataxia, sedation, amnesia, ethanol/barbiturate potentiation, tolerance, dependence, and abuse potential; partial agonists like Imepitoin overcome these limitations due to low intrinsic activity at the benzodiazepine site [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (4.47 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (4.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 1.25 mg/mL (4.47 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5832 mL | 17.9160 mL | 35.8320 mL | |
| 5 mM | 0.7166 mL | 3.5832 mL | 7.1664 mL | |
| 10 mM | 0.3583 mL | 1.7916 mL | 3.5832 mL |