Physicochemical Properties
| Molecular Formula | C11H20N4O6 |
| Molecular Weight | 307.3 |
| Exact Mass | 304.138 |
| Elemental Analysis | C, 43.42; H, 6.62; N, 18.41; O, 31.55 |
| CAS # | 75459-34-6 |
| Related CAS # | 75459-34-6 (free);ADR-925 HCl; |
| PubChem CID | 10402764 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.409±0.06 g/cm3(Predicted) |
| Boiling Point | 644.3±55.0 °C(Predicted) |
| Melting Point | 128-130 °C |
| LogP | -6.7 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 21 |
| Complexity | 414 |
| Defined Atom Stereocenter Count | 1 |
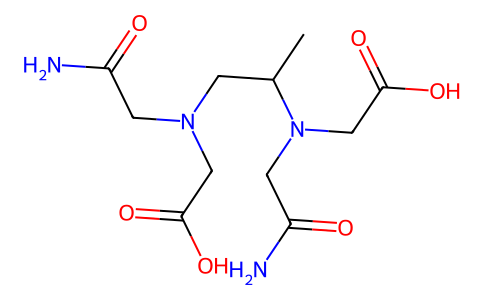
| SMILES | CC(N(CC(=O)O)CC(=O)N)CN(CC(=O)N)CC(=O)O |
| InChi Key | YXWHFCSUHVBWFG-ZETCQYMHSA-N |
| InChi Code | InChI=1S/C11H20N4O6/c1-7(15(4-9(13)17)6-11(20)21)2-14(3-8(12)16)5-10(18)19/h7H,2-6H2,1H3,(H2,12,16)(H2,13,17)(H,18,19)(H,20,21)/t7-/m0/s1 |
| Chemical Name | 2-[(2-amino-2-oxoethyl)-[(2S)-2-[(2-amino-2-oxoethyl)-(carboxymethyl)amino]propyl]amino]acetic acid |
| Synonyms | Icrf198; ICRF 198; 75459-34-6; ADR-925; Adr 925; 2-[(2-amino-2-oxoethyl)-[2-[(2-amino-2-oxoethyl)-(carboxymethyl)amino]propyl]amino]acetic acid; 2-[(2-amino-2-oxoethyl)-[(2S)-2-[(2-amino-2-oxoethyl)-(carboxymethyl)amino]propyl]amino]acetic acid; SCHEMBL30025804; Icrf-198; Icrf 198 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
ADR-925 (ICRF 198) functions as a metal-chelating agent, with its active form chelating metal ions (likely iron) that contribute to oxidative stress and cardiotoxicity [1] |
| ln Vitro |
ADR-925 (ICRF 198) is a one-ring open metabolite of dexrazoxane. In in vitro systems simulating metabolic processes, it can be converted to the active metal-chelating form (the closed-ring form of dexrazoxane metabolites). This conversion is thought to involve enzymatic or chemical reactions that restore the cyclic structure necessary for metal chelation [1] |
| ln Vivo |
In rats, ADR-925 (ICRF 198) (administered as a metabolite of dexrazoxane) undergoes further metabolism to form the active closed-ring metal-chelating metabolite. The conversion occurs in various tissues, including the liver, and possibly through non-enzymatic processes. After formation, the active metabolite is distributed to tissues, where it exerts its chelating effects [1] |
| ADME/Pharmacokinetics |
In rats, ADR-925 (ICRF 198) (administered as a metabolite of dexrazoxane) undergoes further metabolism to form the active closed-ring metal-chelating metabolite. The conversion occurs in various tissues, including the liver, and possibly through non-enzymatic processes. After formation, the active metabolite is distributed to tissues, where it exerts its chelating effects [1] |
| References | [1]. Metabolism of the one-ring open metabolites of the cardioprotective drug dexrazoxane to its active metal-chelating form in the rat. Drug Metab Dispos. 2005 Sep;33(9):1367-72 |
| Additional Infomation |
ADR-925 (ICRF 198) is an intermediate metabolite in the metabolic pathway of dexrazoxane, a cardioprotective drug. Its role is to serve as a precursor to the active metal-chelating form, which helps mitigate cardiotoxicity caused by anthracycline chemotherapy (e.g., doxorubicin) by chelating metal ions involved in generating reactive oxygen species [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2541 mL | 16.2707 mL | 32.5415 mL | |
| 5 mM | 0.6508 mL | 3.2541 mL | 6.5083 mL | |
| 10 mM | 0.3254 mL | 1.6271 mL | 3.2541 mL |