Physicochemical Properties
| Molecular Formula | C20H26N2O2 |
| Molecular Weight | 326.4326 |
| Exact Mass | 326.199 |
| CAS # | 1435-55-8 |
| Related CAS # | 1476-98-8 (mono-hydrochloride);1668-97-9 (mono-hydrochloride) |
| PubChem CID | 91503 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 498.4±30.0 °C at 760 mmHg |
| Melting Point | 169-170 ºC |
| Flash Point | 255.2±24.6 °C |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.626 |
| LogP | 3.77 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 24 |
| Complexity | 432 |
| Defined Atom Stereocenter Count | 4 |
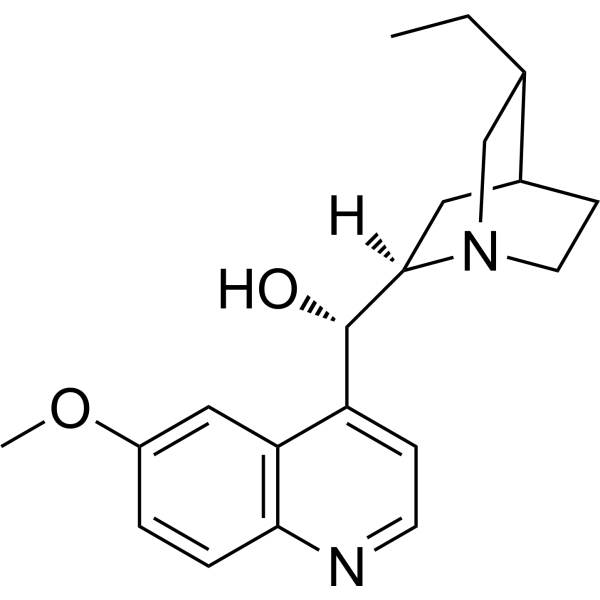
| SMILES | CC[C@H]1CN2CC[C@H]1C[C@@H]2[C@H](C3=C4C=C(C=CC4=NC=C3)OC)O |
| InChi Key | LJOQGZACKSYWCH-LHHVKLHASA-N |
| InChi Code | InChI=1S/C20H26N2O2/c1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18/h4-6,8,11,13-14,19-20,23H,3,7,9-10,12H2,1-2H3/t13-,14-,19+,20-/m0/s1 |
| Chemical Name | (S)-[(2R,4S,5R)-5-ethyl-1-azabicyclo[2.2.2]octan-2-yl]-(6-methoxyquinolin-4-yl)methanol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Hydroquinidine is a class Ia antiarrhythmic drug. The study mentions it inhibits the transient outward potassium current (Ito) of the cardiac action potential, which is implicated in Brugada syndrome. [2] |
| ln Vivo |
This clinical study demonstrates the in vivo (human) efficacy of long-term Hydroquinidine treatment in patients with Short QT Syndrome (SQTS). In 17 treated patients, Hydroquinidine (mean dose 584 ± 53 mg/day) significantly prolonged the QTc interval from a baseline of 331 ± 3 ms to 391 ± 5 ms (mean prolongation 60 ± 6 ms; p < 0.001). In a matched-period analysis of 15 patients treated long-term (mean 6 ± 1 years), the percentage of patients experiencing life-threatening arrhythmic events (LAE: cardiac arrest or sudden cardiac death) dropped from 40% in an equal period before treatment to 0% during treatment (p = 0.03). The number of LAE per patient dropped from 0.73 ± 0.30 to 0 (p = 0.026). In a separate analysis of 16 SQTS patients who had survived a prior cardiac arrest, the annual rate of LAE dropped from 12% (10 events over 82 person-years) off treatment to 0% (0 events over 44 person-years) on Hydroquinidine treatment (p = 0.028). [1] This clinical study (human) evaluated the efficacy of Hydroquinidine in patients with Brugada syndrome (BrS). In asymptomatic BrS patients with inducible ventricular tachycardia/ventricular fibrillation (VT/VF) undergoing electrophysiologic (EP)-guided therapy (n=29), Hydroquinidine (mean dose 609 ± 89 mg/day) prevented VT/VF inducibility in 22 patients (76%). [2] Among 21 asymptomatic, inducible patients who received long-term Hydroquinidine therapy (mean follow-up 17 ± 13 months), 17 (81%) remained asymptomatic. Two patients experienced syncope during follow-up; one case was associated with major QTc prolongation (530 ms) and documented non-sustained VT runs, while the other was unexplained but possibly linked to non-compliance. Two other patients discontinued Hydroquinidine due to late gastrointestinal intolerance. [2] In a separate group of 10 asymptomatic, inducible patients who received an implantable cardioverter-defibrillator (ICD) instead of long-term Hydroquinidine (due to persistent inducibility or early intolerance), one appropriate ICD shock occurred during a mean follow-up of 13 ± 8 months. [2] In four symptomatic BrS patients with a history of multiple appropriate ICD shocks, long-term Hydroquinidine therapy prevented shock recurrence and VT/VF episodes in all cases during a mean follow-up of 14 ± 8 months. A dramatic reduction in ventricular arrhythmia salvos was documented in one patient. [2] |
| ADME/Pharmacokinetics |
The study monitored plasma levels of Hydroquinidine. The mean plasma level in treated patients was 2.7 ± 3.3 µmol/L. The stated usual therapeutic concentration range (by fluorimetry) is 3 to 6 µmol/L. Dose adjustments were made based on plasma levels and electrophysiologic study results. The standard dose was a "slow-release" preparation of 300 mg twice daily (600 mg/day). The dose was increased to 900 mg/day if VT/VF remained inducible and the plasma level was below 3 µmol/L. [2] |
| Toxicity/Toxicokinetics |
In this study, Hydroquinidine therapy was generally well-tolerated. Two out of 17 patients (12%) discontinued treatment within one week due to gastrointestinal intolerance (diarrhea). No pro-arrhythmic events were observed during the treatment period. The study mentions that the tolerability profile was better than in a previous study on Brugada syndrome, possibly due to the lower mean daily dose used (584 ± 53 mg/day vs. 738 ± 25 mg/day). [1] During the trial period, drug intolerance occurred in 7 of 35 patients (20%), primarily diarrhea, leading to Hydroquinidine withdrawal in 2 patients. One patient experienced a reversible Hydroquinidine-induced hepatitis (three-fold increase in transaminases) with nausea and asthenia after one week of treatment, leading to drug discontinuation. [2] One patient experienced syncope associated with a major QTc interval prolongation to 530 ms (from a baseline of 425 ms) and documented runs of monomorphic VT one month after starting Hydroquinidine. This was considered a possible proarrhythmic event. A QTc >500 ms was observed only in this patient. [2] Overall, 5 of 35 patients (14%) discontinued Hydroquinidine due to extracardiac side effects (gastrointestinal intolerance or hepatitis). [2] |
| References |
[1]. Hydroquinidine Prevents Life-Threatening Arrhythmic Events in Patients With Short QT Syndrome. J Am Coll Cardiol. 2017 Dec 19;70(24):3010-3015. [2]. Hydroquinidine therapy in Brugada syndrome. J Am Coll Cardiol. 2004 May 19;43(10):1853-60. [3]. Comparison of the effectiveness of dihydroquinidine and quinidine on ventricular ectopy after acute and chronic administration. Cardiovasc Drugs Ther. 1988 Dec;2(5):679-86. |
| Additional Infomation |
Hydroquinidine is under investigation in clinical trial NCT00927732 (Hydroquinidine Versus Placebo in Patients With Brugada Syndrome). See also: Hydroquinine (annotation moved to). Hydroquinidine is a quinidine derivative used off-label to treat Short QT Syndrome (SQTS), a rare inherited arrhythmogenic disorder characterized by an abnormally short QT interval on the electrocardiogram and a high risk of sudden cardiac death. The study protocol involved starting Hydroquinidine at a dose of 3 mg/kg/day and gradually titrating upwards to achieve a QTc interval >360 ms. The mean maintenance dose was 584 ± 53 mg/day (approximately 8 mg/kg/day). The study concludes that long-term treatment with Hydroquinidine is effective in prolonging the QT interval and preventing life-threatening arrhythmic events in high-risk SQTS patients. It suggests the drug could be used as adjunctive therapy to reduce implantable cardioverter-defibrillator (ICD) shocks or as an alternative therapy when an ICD is contraindicated, unavailable, or refused by the patient. The authors advocate for the continued global availability of quinidine drugs for treating life-threatening inherited arrhythmias like SQTS. [1] Hydroquinidine chlorhydrate (slow-release preparation) was used in this study for the management of Brugada syndrome (BrS). [2] The proposed mechanism in BrS is inhibition of the transient outward potassium current (Ito), thereby rebalancing the currents during phase 1 of the epicardial action potential and reducing the substrate for phase 2 re-entry arrhythmias. [2] The treatment protocol involved an initial EP study. Hydroquinidine was then started, and a repeat EP study was performed after at least two weeks to assess suppression of VT/VF inducibility. Long-term therapy was continued if the patient was non-inducible and tolerated the drug. [2] Electrocardiographic changes observed with Hydroquinidine included a slight but significant prolongation of QRS duration (by 13 ± 12 ms) and QTc interval (by 10 ± 7 ms). A reduction or normalization of the characteristic Brugada-type ST-segment elevation was observed in 12 of 35 patients (34%), but this was not correlated with prevention of VT/VF inducibility. [2] The study suggests Hydroquinidine may be an alternative preventive treatment to ICD implantation in selected asymptomatic BrS patients with inducible arrhythmias, but emphasizes the need for careful monitoring of the QTc interval and plasma levels for tolerance and compliance. [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~20.83 mg/mL (~63.81 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.37 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.37 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (6.37 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0634 mL | 15.3172 mL | 30.6344 mL | |
| 5 mM | 0.6127 mL | 3.0634 mL | 6.1269 mL | |
| 10 mM | 0.3063 mL | 1.5317 mL | 3.0634 mL |