Physicochemical Properties
| Molecular Formula | C16H16O5 |
| Molecular Weight | 288.2952 |
| Exact Mass | 288.099 |
| CAS # | 23180-65-6 |
| Related CAS # | (+)-Columbianetin;3804-70-4 |
| PubChem CID | 161409 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 437.7±45.0 °C at 760 mmHg |
| Flash Point | 195.0±28.8 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.572 |
| LogP | 2.26 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 21 |
| Complexity | 481 |
| Defined Atom Stereocenter Count | 1 |
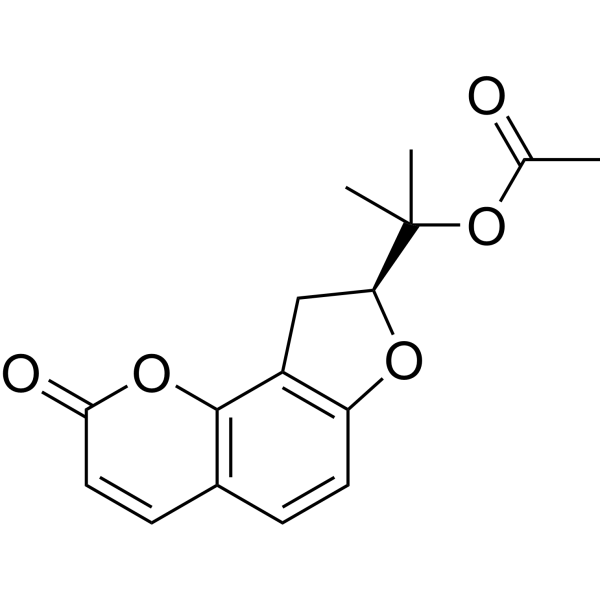
| SMILES | CC(=O)OC(C)(C)[C@@H]1CC2=C(O1)C=CC3=C2OC(=O)C=C3 |
| InChi Key | IQTTZQQJJBEAIM-ZDUSSCGKSA-N |
| InChi Code | InChI=1S/C16H16O5/c1-9(17)21-16(2,3)13-8-11-12(19-13)6-4-10-5-7-14(18)20-15(10)11/h4-7,13H,8H2,1-3H3/t13-/m0/s1 |
| Chemical Name | 2-[(8S)-2-oxo-8,9-dihydrofuro[2,3-h]chromen-8-yl]propan-2-yl acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
- Antipathogenic activity: Columbianetin exhibited inhibitory activity against plant pathogens associated with celery storage decay, including Botrytis cinerea, Fusarium oxysporum, and Rhizoctonia solani. The growth of these pathogens was suppressed when exposed to Columbianetin extracted from resistant celery cultivars, with the inhibitory effect positively correlated to the compound's concentration in celery tissues [1] - Anti-inflammatory activity: Columbianetin inhibited the activation of human mast cells (HMC-1) induced by phorbol 12-myristate 13-acetate (PMA) plus calcium ionophore A23187. At concentrations of 10 μM, 30 μM, and 100 μM, it dose-dependently reduced the release of histamine (inhibition rates of 21.3%, 45.7%, and 68.2% respectively) and tryptase (inhibition rates of 18.9%, 42.5%, and 65.3% respectively) from activated HMC-1 cells. Additionally, it significantly decreased the production of pro-inflammatory cytokines TNF-α, IL-6, and IL-8, with the highest inhibition observed at 100 μM (TNF-α: 72.1% inhibition; IL-6: 69.4% inhibition; IL-8: 67.8% inhibition) [2] |
| Cell Assay |
- Human mast cell activation and inflammatory mediator detection assay: HMC-1 cells were cultured in appropriate medium and seeded into 24-well plates at a density of 1×10⁶ cells/well. The cells were pre-treated with Columbianetin at concentrations of 10 μM, 30 μM, and 100 μM for 1 hour, followed by stimulation with PMA (10 ng/mL) plus A23187 (1 μM) for 6 hours. After incubation, the cell supernatant was collected to measure histamine levels using a fluorometric assay, tryptase activity via a colorimetric assay, and concentrations of TNF-α, IL-6, and IL-8 by enzyme-linked immunosorbent assay (ELISA). Cells without drug treatment but with PMA/A23187 stimulation served as the positive control, and unstimulated cells served as the negative control [2] |
| References |
[1]. Columbianetin, a phytoalexin associated with celery resistance to pathogens during storage. 1995. Volume 39, Issue 6, August 1995, Pages 1347-1350. [2]. Anti-inflammatory effect of Columbianetin on activated human mast cells. Biol Pharm Bull. 2009 Jun;32(6):1027-31. |
| Additional Infomation |
Columbianetin acetate is an acetate ester obtained by formal acetylation of the tertiary hydroxy group of 2-[(8S)-2-oxo-8,9-dihydro-2H-furo[2,3-h][1]benzopyran-8-yl]propan-2-ol. It has a role as a plant metabolite. It is a furanocoumarin and an acetate ester. Columbianetin acetate has been reported in Cnidium monnieri, Murraya siamensis, and other organisms with data available. - Phytochemical background: Columbianetin is a phytoalexin naturally produced in celery (Apium graveolens) plants, particularly in resistant cultivars in response to pathogen infection during post-harvest storage. Its biosynthesis is induced as a defense mechanism against fungal and bacterial pathogens [1] - Anti-inflammatory mechanism: Columbianetin exerts anti-inflammatory effects by suppressing the activation of signaling pathways involved in mast cell degranulation and pro-inflammatory cytokine production, although the specific molecular targets remain uncharacterized [2] - Chemical nature: Columbianetin belongs to the coumarin family of natural products, characterized by a benzopyranone core structure, which is associated with various biological activities including antimicrobial and anti-inflammatory effects [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~346.86 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.67 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.67 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4686 mL | 17.3430 mL | 34.6861 mL | |
| 5 mM | 0.6937 mL | 3.4686 mL | 6.9372 mL | |
| 10 mM | 0.3469 mL | 1.7343 mL | 3.4686 mL |