Physicochemical Properties
| Molecular Formula | C22H47NO5 |
| Molecular Weight | 405.61228 |
| Exact Mass | 405.345 |
| CAS # | 145040-09-1 |
| PubChem CID | 21119850 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.051±0.06 g/cm3(Predicted) |
| Boiling Point | 593.8±50.0 °C(Predicted) |
| Vapour Pressure | 1.42E-16mmHg at 25°C |
| LogP | 3.031 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 28 |
| Complexity | 371 |
| Defined Atom Stereocenter Count | 8 |
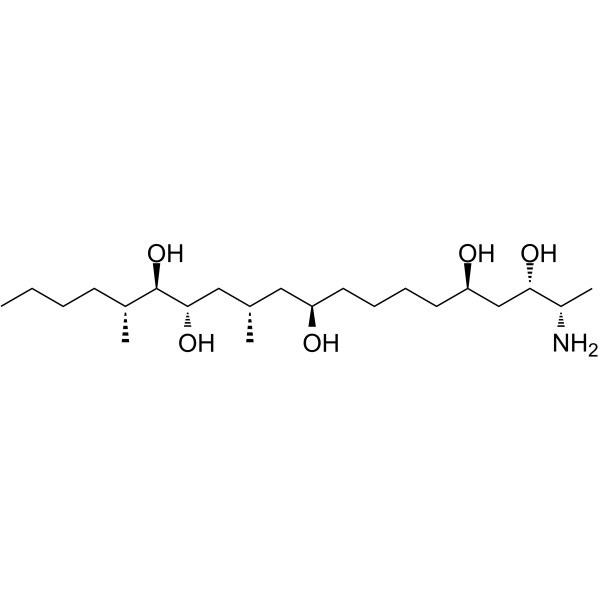
| SMILES | CCCC[C@@H]([C@@H]([C@@H](C[C@@H](C[C@H](CCCC[C@H](C[C@@H]([C@@H](N)C)O)O)O)C)O)O)C |
| InChi Key | UWWVLQOLROBFTD-GADKELDLSA-N |
| InChi Code | InChI=1S/C22H47NO5/c1-5-6-9-16(3)22(28)21(27)13-15(2)12-18(24)10-7-8-11-19(25)14-20(26)17(4)23/h15-22,24-28H,5-14,23H2,1-4H3/t15-,16+,17-,18+,19+,20-,21-,22+/m0/s1 |
| Chemical Name | (2S,3S,5R,10R,12S,14S,15R,16R)-2-amino-12,16-dimethylicosane-3,5,10,14,15-pentol |
| Synonyms | AP1 Metabolite; Aminopentol; Aminopentol formate salt; 145040-09-1; (2S,3S,5R,10S,12R,14R,15S,16S)-2-amino-12,16-dimethylicosane-3,5,10,14,15-pentol; 3,5,10,14,15-Eicosanepentol, 2-amino-12,16-dimethyl-, (2S,3S,5R,10R,12S,14S,15R,16R)-; 3,5,10,14,15-Eicosanepentol, 2-amino-12,16-dimethyl-, (2S-(2R*,3R*,5S*,10S*,12R*,14R*,15S*,16S*))-; 2-amino-12,16-dimethylicosane-3,5,10,14,15-pentol; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Hydrolysis product of Fumonisin B1 |
| ln Vitro | Fumonisin B1 (FB1) is the predominant member of a family of mycotoxins produced by Fusarium moniliforme (Sheldon) and related fungi. Certain foods also contain the aminopentol backbone (AP1) that is formed upon base hydrolysis of the ester-linked tricarballylic acids of FB1. Both FB1 and, to a lesser extent, AP1 inhibit ceramide synthase due to structural similarities between fumonisins (as 1-deoxy-analogs of sphinganine) and sphingoid bases. To explore these structure-function relationships further, erythro- and threo-2-amino, 3-hydroxy- (and 3, 5-dihydroxy-) octadecanes were prepared by highly stereoselective syntheses. All of these analogs inhibit the acylation of sphingoid bases by ceramide synthase, and are themselves acylated with Vmax/Km of 40-125 for the erythro-isomers (compared with approximately 250 for D-erythro-sphinganine) and 4-6 for the threo-isomers. Ceramide synthase also acylates AP1 (but not FB1, under the conditions tested) to N-palmitoyl-AP1 (PAP1) with a Vmax/Km of approximately 1. The toxicity of PAP1 was evaluated using HT29 cells, a human colonic cell line. PAP1 was at least 10 times more toxic than FB1 or AP1 and caused sphinganine accumulation as an inhibitor of ceramide synthase. These studies demonstrate that: the 1-hydroxyl group is not required for sphingoid bases to be acylated; both erythro- and threo-isomers are acylated with the highest apparent Vmax/Km for the erythro-analogs; and AP1 is acylated to PAP1, a new category of ceramide synthase inhibitor as well as a toxic metabolite that may play a role in the diseases caused by fumonisins [2]. |
| References |
[1]. Effects of aminopentol on in utero development in rats. Food Chem Toxicol. 2006 Feb;44(2):161-9. [2]. Acylation of naturally occurring and synthetic 1-deoxysphinganines by ceramide synthase. Formation of N-palmitoyl-aminopentol produces a toxic metabolite of hydrolyzed fumonisin, AP1, and a new category of ceramide synthase inhibitor. J B. [3]. Induction of apoptosis by fumonisin B1 in HT29 cells is mediated by the accumulation of endogenous free sphingoid bases. Toxicol Appl Pharmacol. 1998 Feb;148(2):252-60. |
| Additional Infomation |
(2S,3S,5R,10R,12S,14S,15R,16R)-2-amino-12,16-dimethylicosane-3,5,10,14,15-pentol is a pentol that is (3S,5R,10R,12S,14S,15R,16R)-12,16-dimethylicosane-3,5,10,14,15-pentol in which the pro-S hydrogen at position 2 is substituted by an amino group. It results from hydrolysis of the O-acyl bonds of fumonisin B1. It is a pentol and a primary amino compound. It is a conjugate base of a (2S,3S,5R,10R,12S,14S,15R,16R)-2-amino-12,16-dimethylicosane-3,5,10,14,15-pentol(1+). Aminopentol (AP1), the backbone and main hydrolysis product of the mycotoxin fumonisin B1 (FB1), is present in corn-based foods which are consumed daily as a substantial part of the diet in some areas of the world. The toxicity of FB1 has been attributed to altered sphingolipid metabolism, but the toxicity of AP1 is less certain. Epidemiological correlations and in vitro studies have suggested that AP1 can increase neural tube defects (NTDs), but no in vivo developmental study of AP1 was done prior to this study. AP1 was given once daily to rats by gavage on gestation days (GD) 3-16 at doses of 0, 15, 30, 60, or 120 mg/kg. Reproductive and developmental parameters were measured at GD 17, one day after the last dose, and on GD 20. In addition, on GD 17, maternal and fetal tissues were analyzed for sphingolipid content. Conclusions: AP1 reduced dam body weight gain, but was less toxic than FB1. AP1 was not teratogenic, did not affect tissue sphingolipid ratios, did not alter reproduction or development of fetuses, and produced no dose-related histopathological effects in dams. [1] Fumonisin B1 (FB1) and aminopentol (AP1) (which is formed by hydrolysis of FB1) are found in corn contaminated with some strains of Fusarium moniliforme. Incubation of HT29 cells (a human colonic cell line) with FB1 or AP1 caused a significant reduction in cell number; AP1 was less potent, with 50 microM AP1 causing the same reduction (ca. 30% after 24 h) as 10 microM FB1. The reduction in cell number reflected increases in DNA fragmentation and the percentage of apoptotic cells. Both FB1 and AP1 caused the accumulation of sphinganine (25- and 35-fold by 10 microM FB1 and 50 microM AP1, respectively); thus, concentrations of FB1 and AP1 that caused comparable reductions in cell number were also similar with respect to elevation of sphinganine, a compound that is growth inhibitory and cytotoxic. Inhibition of the first step of sphingolipid biosynthesis with ISP-1 prevented the elevation in sphinganine, DNA fragmentation, and apoptosis induced by FB1. Therefore, these effects of FB1 on HT29 cells can be attributed to the accumulation of sphinganine. Since consumption of food contaminated with Fusarium moniliforme (Sheldon) exposes colonic cells to these mycotoxins, the possibility that FB1 and AP1 are toxic for intestinal cells in vivo should be evaluated, especially in the light of the recent report (Bhat et al., Clin. Toxicol. 35, 249, 1997) describing intestinal disturbances in humans after consumption of moldy corn and sorghum containing fumonisins. [3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4654 mL | 12.3271 mL | 24.6542 mL | |
| 5 mM | 0.4931 mL | 2.4654 mL | 4.9308 mL | |
| 10 mM | 0.2465 mL | 1.2327 mL | 2.4654 mL |