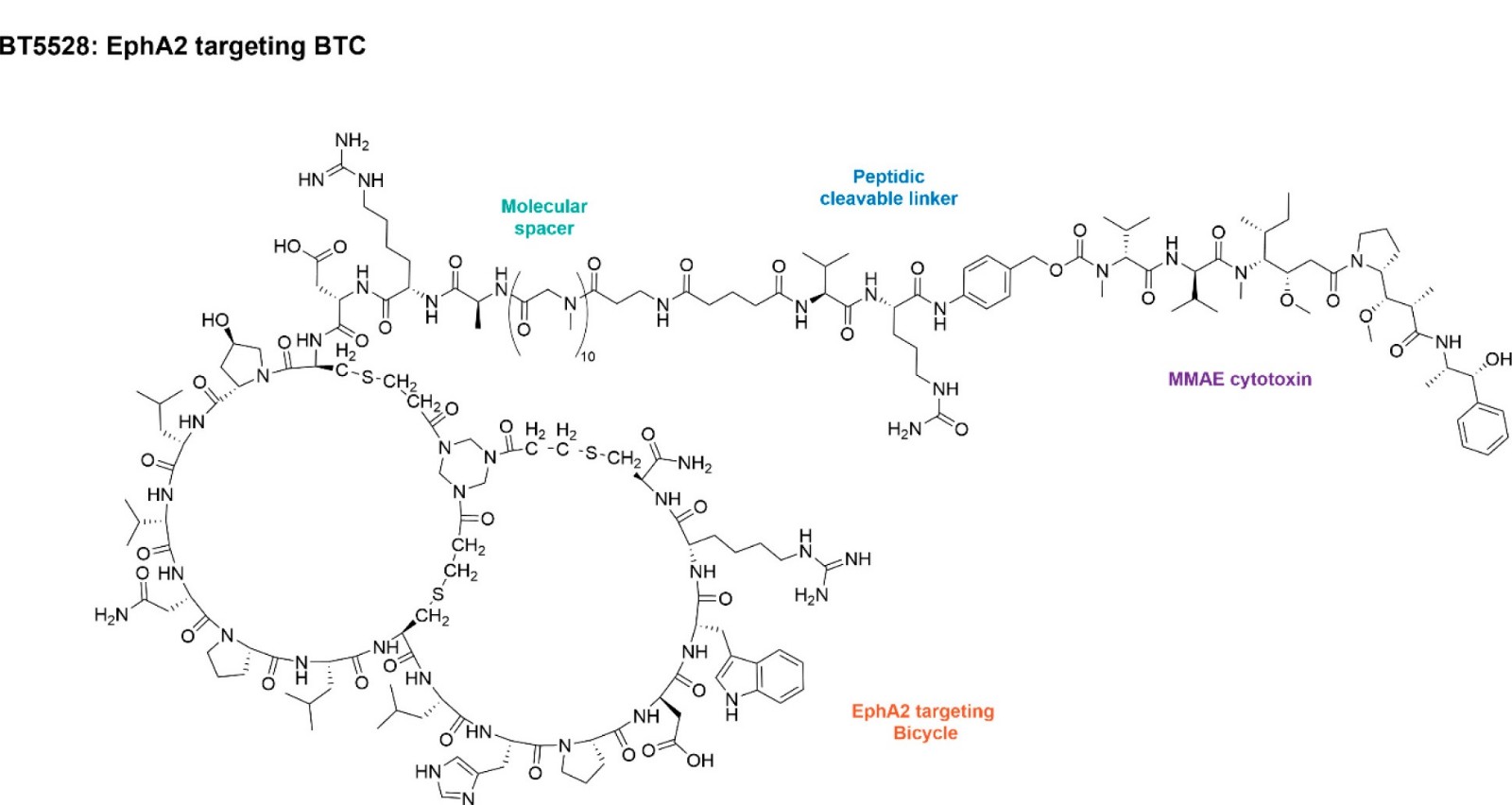
BT5528 (BT-5528) is a Bicycle Toxin Conjugate (BTC) or PDC/peptide-drug conjugate with anticaner activity. It is an EphA2 activator and is composed of a bicyclic peptide targeting the tumor antigen EphA2, linked to a cytotoxin (monomethyl auristatin E [MMAE]) via a tumor microenvironment cleavable linker. Bicycles are a novel class of chemically synthesized constrained peptides, developed by Bicycle Therapeutics. EphA2 is reported to be overexpressed in a range of solid tumors, contributes to oncogenesis, tumor-associated angiogenesis and metastasis. Intracellular EphA2 signaling converges on pathways that are integral to cell growth, proliferation, migration and invasion. Increased EphA2 expression has been identified as a resistance mechanism to EGFR Tyrosine Kinase Inhibitor based therapy. BT5528 mechanism of action is dependent on tumor penetration, target binding and release of MMAE toxin payload. BTCs offer advantages over antibody-toxin conjugates exhibiting rapid penetration of dense tumors and decreased extra-tumor exposure. BT5528 exhibited a favorable preclinical profile supporting the initiation of a first-in-human study to investigate safety and efficacy of BT5528 in indications with evidence of EphA2 expression including non-small-cell lung cancer (NSCLC), ovarian cancer, triple-negative breast cancer (TNBC), gastric/upper gastrointestinal (GI), pancreatic and urothelial cancers. Methods: BT5528-100 (NCT04180371) is a Ph I/II study to evaluate safety and tolerability of weekly BT5528 alone and in combination with Q4W nivolumab. Each dose escalation utilizes a 3+3 design which converts to a Bayesian design to determine MTD or MAD and RP2D for BT5528 with and without nivolumab. Eligible patients must have advanced solid tumors associated with EphA2 expression which have recurred after exhausting standard treatment options. Patients must have available tumor tissue and acceptable hematologic and organ function, with exclusions for uncontrolled brain metastases, thromboembolic events, bleeding disorders, uncontrolled hypertension, CYP3A4 inhibitors/inducers or, for the nivolumab cohorts, autoimmune disease. On-study tumor and blood samples will be collected for biomarker evaluations including tumor EphA2 expression, ADA, and candidate response biomarkers for BT5528 alone and combination with nivolumab. Pharmacokinetic data will be reported for C1D1 and D15 for BT5528 and MMAE. The expansion phase will enroll specific tumor types to evaluate clinical activity of BT5528. Enrollment is ongoing. Clinical trial information: NCT04180371.
Physicochemical Properties
| Molecular Formula | C201H315N53O52S3 |
| Molecular Weight | 4402.17074227333 |
| Exact Mass | 4401.286 |
| CAS # | 2648849-70-9 |
| PubChem CID | 166177190 |
| Appearance | Typically exists as solid at room temperature |
| LogP | -6.1 |
| Hydrogen Bond Donor Count | 35 |
| Hydrogen Bond Acceptor Count | 58 |
| Rotatable Bond Count | 95 |
| Heavy Atom Count | 309 |
| Complexity | 10900 |
| Defined Atom Stereocenter Count | 31 |
| SMILES | CC[C@@H](C)[C@H]([C@H](CC(=O)N1CCC[C@@H]1[C@H]([C@H](C)C(=O)N[C@@H](C)[C@@H](C2=CC=CC=C2)O)OC)OC)N(C)C(=O)[C@@H](C(C)C)NC(=O)[C@@H](C(C)C)N(C)C(=O)OCC3=CC=C(C=C3)NC(=O)[C@@H](CCCNC(=O)N)NC(=O)[C@@H](C(C)C)NC(=O)CCCC(=O)NCCC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N(C)CC(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN=C(N)N)C(=O)N[C@@H](CC(=O)O)C(=O)N[C@H]4CSCCC(=O)N5CN6CN(C5)C(=O)CCSC[C@H](NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H]7CCCN7C(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CSCCC6=O)NC(=O)[C@@H](NC(=O)[C@@H]8CCCN8C(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H]9C[C@H](CN9C4=O)O)CC(C)C)C(C)C)CC(=O)N)CC(C)C)CC(C)C)CC1=CNC=N1)CC(=O)O)CC1=CNC2=CC=CC=C21)CCCCN=C(N)N)C(=O)N |
| InChi Key | YZUGAHYDHBVJAU-KCKBOPTCSA-N |
| InChi Code | InChI=1S/C201H315N53O52S3/c1-34-117(16)172(147(304-32)88-156(264)251-73-46-56-143(251)174(305-33)118(17)176(280)217-119(18)173(278)122-49-36-35-37-50-122)246(30)197(301)170(115(12)13)235-193(297)171(116(14)15)247(31)201(303)306-102-121-61-63-124(64-62-121)218-178(282)131(55-45-71-213-200(208)302)221-191(295)168(113(8)9)233-150(258)60-44-59-149(257)210-72-65-152(260)237(21)93-158(266)239(23)95-160(268)241(25)97-162(270)243(27)99-164(272)245(29)101-165(273)244(28)100-163(271)242(26)98-161(269)240(24)96-159(267)238(22)94-157(265)236(20)92-151(259)216-120(19)177(281)219-129(53-40-42-69-211-198(204)205)179(283)224-136(86-166(274)275)185(289)232-142-105-309-78-68-155(263)250-108-248-107-249(109-250)154(262)67-77-308-104-141(231-182(286)133(80-111(4)5)225-188(292)144-57-48-75-253(144)195(299)139(85-148(202)256)229-192(296)169(114(10)11)234-186(290)134(81-112(6)7)226-190(294)146-84-126(255)91-254(146)196(142)300)187(291)222-132(79-110(2)3)181(285)228-138(83-125-90-209-106-215-125)194(298)252-74-47-58-145(252)189(293)227-137(87-167(276)277)184(288)223-135(82-123-89-214-128-52-39-38-51-127(123)128)183(287)220-130(54-41-43-70-212-199(206)207)180(284)230-140(175(203)279)103-307-76-66-153(248)261/h35-39,49-52,61-64,89-90,106,110-120,126,129-147,168-174,214,255,278H,34,40-48,53-60,65-88,91-105,107-109H2,1-33H3,(H2,202,256)(H2,203,279)(H,209,215)(H,210,257)(H,216,259)(H,217,280)(H,218,282)(H,219,281)(H,220,287)(H,221,295)(H,222,291)(H,223,288)(H,224,283)(H,225,292)(H,226,294)(H,227,293)(H,228,285)(H,229,296)(H,230,284)(H,231,286)(H,232,289)(H,233,258)(H,234,290)(H,235,297)(H,274,275)(H,276,277)(H4,204,205,211)(H4,206,207,212)(H3,208,213,302)/t117-,118+,119+,120+,126-,129+,130+,131-,132+,133+,134+,135+,136+,137+,138+,139+,140+,141+,142+,143-,144+,145+,146+,147+,168-,169+,170-,171-,172-,173+,174+/m1/s1 |
| Chemical Name | (3S)-4-[[(9R,13R,15S,18S,21S,24S,30S,33S,36R,39S,42S,48S,51S,54S,57S,60R)-24-(2-amino-2-oxoethyl)-60-carbamoyl-51-(carboxymethyl)-57-[4-(diaminomethylideneamino)butyl]-13-hydroxy-42-(1H-imidazol-4-ylmethyl)-54-(1H-indol-3-ylmethyl)-18,33,39-tris(2-methylpropyl)-4,10,16,19,22,25,31,34,37,40,43,49,52,55,58,65,68-heptadecaoxo-21-propan-2-yl-7,62,71-trithia-1,3,11,17,20,23,26,32,35,38,41,44,50,53,56,59,66-heptadecazahexacyclo[34.31.5.13,66.011,15.026,30.044,48]triheptacontan-9-yl]amino]-3-[[(2S)-2-[[(2S)-2-[[2-[[2-[[2-[[2-[[2-[[2-[[2-[[2-[[2-[[2-[3-[[5-[[(2R)-1-[[(2R)-5-(carbamoylamino)-1-[4-[[[(2R)-1-[[(2R)-1-[[(3S,4R,5R)-1-[(2R)-2-[(1S,2S)-3-[[(1R,2S)-1-hydroxy-1-phenylpropan-2-yl]amino]-1-methoxy-2-methyl-3-oxopropyl]pyrrolidin-1-yl]-3-methoxy-5-methyl-1-oxoheptan-4-yl]-methylamino]-3-methyl-1-oxobutan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]-methylcarbamoyl]oxymethyl]anilino]-1-oxopentan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]amino]-5-oxopentanoyl]amino]propanoyl-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]-methylamino]acetyl]amino]propanoyl]amino]-6-(diaminomethylideneamino)hexanoyl]amino]-4-oxobutanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1] https://ascopubs.org/doi/10.1200/JCO.2020.38.15_suppl.TPS3655 [2].Abstract 5854: bt5528, a bicycle toxin conjugate targeting epha2 has potent anti-tumor activity without bleeding or coagulation abnormalities in preclinical models.Cancer Research. 2018.78(13 Supplement), 5854-5854. [3].MMAE Delivery Using theBicycleToxin Conjugate BT5528. Mol Cancer Ther. 2020 Jul;19(7):1385-1394. |
| Additional Infomation | EphA2-targeting Bicycle Toxin Conjugate BT5528 is a bicyclic peptide targeting Ephrin receptor A2 (EphA2) and conjugated, through an inert sarcosine spacer chain and a valine-citrulline cleavable linker, to the cytotoxic agent monomethyl auristatin E (MMAE), an auristatin derivative and a potent inhibitor of microtubule polymerization, with potential antineoplastic activity. Upon administration of the EphA2-targeting bicycle toxin conjugate BT5528, this agent targets and binds to EphA2-expressing tumor cells. After internalization and enzymatic cleavage of the immunoconjugate within the tumor cell cytosol, free MMAE binds to tubulin and inhibits its polymerization, which may result in G2/M phase arrest and tumor cell apoptosis. The cell-surface receptor EphA2, a member of the ephrin family of receptor tyrosine kinases (RTKs) involved in mammalian development, is overexpressed by a variety of different cancer cell types. EphA2 expression is associated with poor prognosis. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.2272 mL | 1.1358 mL | 2.2716 mL | |
| 5 mM | 0.0454 mL | 0.2272 mL | 0.4543 mL | |
| 10 mM | 0.0227 mL | 0.1136 mL | 0.2272 mL |