Physicochemical Properties
| Molecular Formula | C25H24N4O2 |
| Molecular Weight | 412.48 |
| Exact Mass | 412.189 |
| CAS # | 1916505-13-9 |
| PubChem CID | 141437001 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 31 |
| Complexity | 656 |
| Defined Atom Stereocenter Count | 0 |
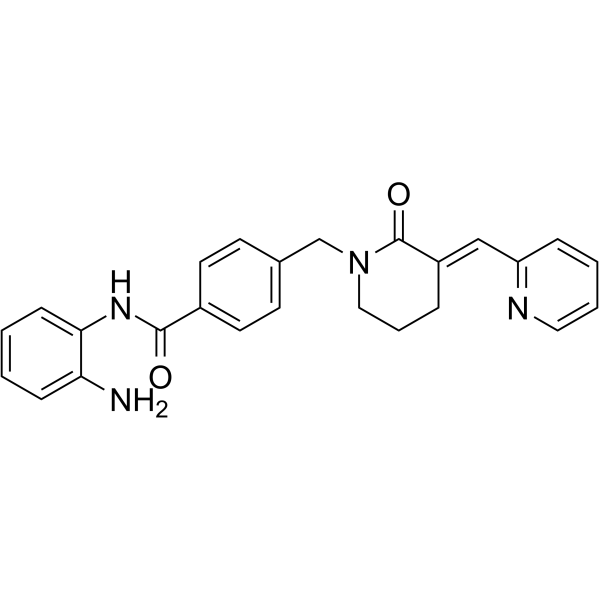
| SMILES | C1C/C(=C\C2=CC=CC=N2)/C(=O)N(C1)CC3=CC=C(C=C3)C(=O)NC4=CC=CC=C4N |
| InChi Key | NBUJRTVDQYEPKP-CAPFRKAQSA-N |
| InChi Code | InChI=1S/C25H24N4O2/c26-22-8-1-2-9-23(22)28-24(30)19-12-10-18(11-13-19)17-29-15-5-6-20(25(29)31)16-21-7-3-4-14-27-21/h1-4,7-14,16H,5-6,15,17,26H2,(H,28,30)/b20-16+ |
| Chemical Name | N-(2-aminophenyl)-4-[[(3E)-2-oxo-3-(pyridin-2-ylmethylidene)piperidin-1-yl]methyl]benzamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | HDAC-IN-31 (compound 24g) (2 µM) inhibits the growth of TMD-8, HCT 116, A549, and MDA-MB-231 cells at growth rates of 2.32%, 44.01%, 48.53%, and 64.94% [1]. HDAC-IN-31's (1 µM) IC50 values against HDAC 1, HDAC 2, HDAC 3, HDAC 8, and 81.20%, 84.43%, 88.07%, and 92.34% were, in that order, 84.9, 168.0, 442.7, and >10000 nM. According to reference [1], the corresponding enzyme activity of HDAC4, HDAC5, HDAC 7, HDAC9, HDAC 6, and HDAC 11 are 96.88% and 91.98%. In a dose-dependent manner, HDAC-IN-31 (2.5, 5, 7.5, 10 µM; 24 h) enhances the expression of HDAC1, Ace-H3, Ace-H4, Cleaved PARP, and Cleaved Caspase-3 [1]. Inducing apoptosis and cell cycle arrest in the G2/M phase, HDAC-IN-31 (0–4 µM; 24 h) works in a dose-dependent manner [1]. |
| ln Vivo | HDAC-IN-31 exhibits strong dose dependence and good bioavailability at 2 mg/kg intravenously and 10–100 mg/kg orally [1]. In the TMD-8 xenograft model, HDAC-IN-31 (50, 100 mg/kg; oral, daily, for 21 days) demonstrated good anti-tumor efficacy without obvious toxicity [1]. HDAC-IN-31 pharmacokinetic parameters in mice [1]. The parameters are as follows: parameter unit: 24 g (25 mg/kg); Cmax: ng·h·mL-1 3100±231 T1/2(po) h 4.4±0.3 AUC0-inf(iv) ng·h·mL-1 1040±142 AUC0-inf (po) ng·h·mL-1 5180±252 MRTPO h 2.6±0.4 F % 39.9±2.1 ICR mice; intravenous injection: 2 mg/kg; oral: 25 mg/kg]. HDAC-IN-31's pharmacokinetic characteristics in tumor models [1]. Parameter unit po (25 mg/kg) po (50 mg/kg) po (100 mg/kg) Cmax ng·h·mL-1 1700±317 14700±1024 10700±1001 AUC0-t ng·h·mL-1 1220±242 9710±314 9740±230 AUC0-inf ng·h·mL-1 1230±165 9730±341 9770±332 MRT0-t h 0.750±0.043 0.812±0.023 1.43±0.56 MRT0-inf h 0.805±0。 086 0.821± 0.041 1.51±0.32 mice;25,50, and 100 mg/kg administered orally[1]. HDAC-IN-31's pharmacokinetic characteristics in tumor models [1]. Units of PK parameter: iv (2 mg/kg), po (10 mg/kg), po (100 mg/kg), Cmax ng·h·mL-1 3960±413 58300±1352 T1/2 h 0.427±0.016 1.31±0.27 1.63± 0.52 AUC0-inf ng·h·mL-1 1250±132 2670±286 57200±1047 MRT·h 0.402±0.032 0.919±0.052 0.897±0.041 CL mL·kg·min-1 27.2±1.2 F % 45.6±1.2 91.8 ± 2.3 ICR mice; 2 mg/kg intravenously; 10, 100 mg/kg orally [1]. HDAC-IN-31's pharmacokinetic characteristics in tumor models [1]. PK parameter unit: monkey IV (1 mg/kg) po (10 mg/kg) IV (1 mg/kg) po (10 mg/kg) Cmax ng·h·mL-1 8520±301 4740±243 T1/2 h 4.31±0.56 9.14±0.32 1.65±0.41 1.51±0.33 AUC0-inf ng·h·mL-1 15700±1842, 53200±1241 2550±365 15100±2004 MRT h 3.41±0.12 8.28±0.32 2.2 6±0.41 2.71±0.32 CL mL·kg·min-1 1.35±0.21 6.72±0.35 Vdss L·kg-1 0.34±0.22 0.55±0.04 F % 27.6±2.1 58.9±1.2 Dogs and monkeys; monkeys: intravenously 1 mg/kg, oral 10 mg/kg [1]. |
| Cell Assay |
Cell proliferation assay[1] Cell Types: MDA-MB-231, A549, NCI-H460, HCT-116, SK-OV-3, HT-29, COLO 678, NCI-H441, 22Rv1, 786-O, TMD- 8. DOHH-2, CCRF-CEM, SU-DHL-2, REC-1, MOLT-4, HUT-78, RS4; 11 Cell Tested Concentrations: 0-20 µM Incubation Duration: 72 hrs (hours) Experimental Results: Broad spectrum anti- Tumor activity, the IC50 of MDA- is 2.29, 2.85, 1.58, 1.16, 3.17, 2.41, 8.02, 2.62, 1.14, 0.60, 0.31, 0.39, 0.48, 0.51, 0.33, 0.38, 0.80, 0.47 µM MB-231, A549, NCI-H460, HCT-116, SK-OV-3, HT-29, COLO 678, NCI-H441, 22Rv1, 786-O, TMD-8, DOHH-2, CCRF-CEM, SU-DHL-2, respectively For REC-1, MOLT-4, HUT-78, RS4; 11 cells. Western Blot Analysis[1] Cell Types: TMD-8 Cell Tested Concentrations: 2.5, 5, 7.5, 10 µM Incubation Duration: 24 hrs (hours) Experimental Results: Promote acetylation of HDAC1, HDAC2, HDAC3 substrates Ace-H3 and Ace-H4 at certain doses Dependence method. Apoptosis analysis [1] Cell Types: TMD-8 Cell Tested Concentrations: 0.5, 1, 2, 4 µM Incubation Duration: 24 h Experimental Results: Apoptosis was induced in a concentration-dependent manner. Cell cycle an |
| Animal Protocol |
Animal/Disease Models: ICR mouse[1] Doses: 2 mg/kg, intravenous (iv) (iv)injection; oral 25 mg/kg (DMSO/PEG200/saline = 20:20:60, v/v/v) Route of Administration: intravenous (iv) (iv)injection or oral Experimental Results: Demonstrated high oral bioavailability (F=40%). Animal/Disease Models: Mice[1] Doses: 25, 50, 100 mg/kg Route of Administration: Oral Experimental Results: Oral administration did not show significant dose dependence. Animal/Disease Models: ICR mouse[1] Doses: 2, 10, 100mg/kg (hydrochloride) Route of Administration: 2mg/kg, intravenous (iv) (iv)injection; 10, 100 mg/kg Oral Experimental Results: shows good bioavailability , with significant dose dependence. Animal/Disease Models: dogs and monkeys [1] Doses: 1, 10 mg/kg Route of Administration: 1 mg/kg intravenously (iv) (iv)(iv); oral 10 mg/kg Experimental Results: It demonstrated good pharmacokinetic/PK/PK characteristics for different species. Animal/Disease Models: 5-6 weeks, female CB.17 SCID (severe combined immunodeficient) mouse (TMD-8 tumor xenograft) [1] Doses: 50, 100 mg/kg Route of Administration: Po, one time/day for 21 days Experimental Results: Inhibition When the tumor growth rate is 100 mg/kg/d, the effective rate reaches |
| References |
[1]. Design and synthesis of HDAC inhibitors to enhance the therapeutic effect of diffuse large B-cell lymphoma by improving metabolic stability and pharmacokinetic characteristics. Eur J Med Chem. 2022 Feb 5;229:114049. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4244 mL | 12.1218 mL | 24.2436 mL | |
| 5 mM | 0.4849 mL | 2.4244 mL | 4.8487 mL | |
| 10 mM | 0.2424 mL | 1.2122 mL | 2.4244 mL |