Physicochemical Properties
| Molecular Formula | C36H62O8 |
| Molecular Weight | 622.8727 |
| Exact Mass | 622.444 |
| CAS # | 39262-14-1 |
| PubChem CID | 9852086 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 723.1±60.0 °C at 760 mmHg |
| Melting Point | 178 °C |
| Flash Point | 391.1±32.9 °C |
| Vapour Pressure | 0.0±5.3 mmHg at 25°C |
| Index of Refraction | 1.572 |
| LogP | 5.5 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 44 |
| Complexity | 1070 |
| Defined Atom Stereocenter Count | 15 |
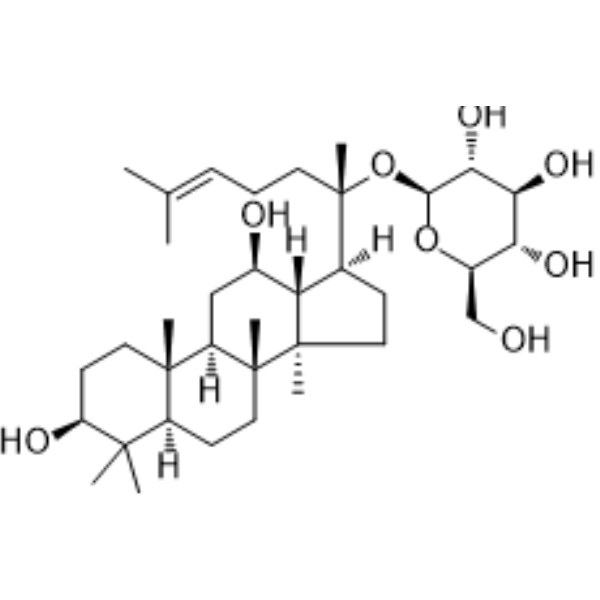
| SMILES | CC(=CCC[C@@](C)([C@H]1CC[C@@]2([C@@H]1[C@@H](C[C@H]3[C@]2(CC[C@@H]4[C@@]3(CC[C@@H](C4(C)C)O)C)C)O)C)O[C@H]5[C@@H]([C@H]([C@@H]([C@H](O5)CO)O)O)O)C |
| InChi Key | FVIZARNDLVOMSU-IRFFNABBSA-N |
| InChi Code | InChI=1S/C36H62O8/c1-20(2)10-9-14-36(8,44-31-30(42)29(41)28(40)23(19-37)43-31)21-11-16-35(7)27(21)22(38)18-25-33(5)15-13-26(39)32(3,4)24(33)12-17-34(25,35)6/h10,21-31,37-42H,9,11-19H2,1-8H3/t21-,22+,23+,24-,25+,26-,27-,28+,29-,30+,31-,33-,34+,35+,36-/m0/s1 |
| Chemical Name | (2S,3R,4S,5S,6R)-2-[(2S)-2-[(3S,5R,8R,9R,10R,12R,13R,14R,17S)-3,12-dihydroxy-4,4,8,10,14-pentamethyl-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl]-6-methylhept-5-en-2-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Ginsenoside CK, a bacterial metabolite of G-Rb1, has anti-inflammatory properties primarily by inhibiting inducible nitric oxide synthase (iNOS), cyclooxygenase (COX)-2, and pro-inflammatory cytokines. Ginsenoside CK reduces pro-inflammatory cytokine expression by inhibiting IAK-1, MAPK, IKK-α, and NF-κB activity in LPS-treated mouse peritoneal macrophages. Ginsenoside CK reduced iNOS and COX-2 expression by decreasing NF-κB activation in LPS-stimulated RAW264.7 cells. Ginsenoside CK decreased the inflammatory response in zymosan-treated bone marrow-derived macrophages (BMDM) and RAW264.7 cells by reducing pro-inflammatory cytokine release, MAPK activation, and ROS generation. Ginsenoside CK also shown anti-inflammatory action in LPS-stimulated microglial cells. Ginsenoside CK reduces inflammation by regulating ROS levels, MAPK, NF-κB, and AP-1 activity [1]. Ginsenoside CK, the major metabolite of ginsenoside in the gastrointestinal tract, suppresses NF-κB activation via PXR. Ginsenoside CK inhibits NF-κB activation, leading to improved recovery from DSS-induced colitis. Ginsenoside CK decreased TNF-α-induced elevation of IL-1β and iNOS mRNA levels while restoring PXR and CYP3A4 mRNA levels in LS174T cells [2]. Ginsenoside CK is an intestine metabolite of 20(S)-protopanaxadiol derivatives. It inhibits the activity of CYP2C9 in human liver microsomes. The IC50 value is 32.0 ± 3.6 μM. It reduces the activity of CYP2A6 in human liver microsomes. The activity weakly inhibits human liver microsome CYP2D6 activity, with an IC50 value of 63.6±4.2 μM and an IC50 value exceeding 100 μM [4]. |
| ln Vivo | The body weight of mice with collagen-induced arthritis (CIA) grew gradually and was notably lower than that of normal DBA/1 mice beginning on the third day following emulsion injection. Mice given lotion injection on day 32 began to restore body weight when given ginsenoside CK at doses of 28, 56, and 112 mg/kg. On day 50, the body weight of the mice treated with ginsenoside CK (56 and 112 mg/kg) and methotrexate (MTX) (2 mg/kg) was considerably higher than that of the CIA mice. On day 24 following immunization, edema in the hind paws began. Treatment was given to CIA mice from day 28 to day 50. Every four days starting on day 24, arthritic scores were recorded. On day 51, ginsenoside CK (56 and 112 mg/kg) dramatically decreased the arthritis scores in mice [3]. |
| References |
[1]. Role of ginsenosides, the main active components of Panax ginseng, in inflammatory responsesand diseases. J Ginseng Res. 2017 Oct;41(4):435-443. [2]. Ginsenoside metabolites, rather than naturally occurring ginsenosides, lead to inhibition of human cytochrome P450 enzymes. Toxicol Sci. 2006 Jun;91(2):356-64. [3]. Ginsenosides Regulate PXR/NF-κB Signaling and Attenuate Dextran Sulfate Sodium-Induced Colitis. Drug Metab Dispos. 2015 Aug;43(8):1181-9. [4]. Ginsenoside compound K suppresses the abnormal activation of T lymphocytes in mice with collagen-induced arthritis. Acta Pharmacol Sin. 2014 May;35(5):599-612. |
| Additional Infomation |
Ginsenoside C-K is a ginsenoside found in Panax species that is dammarane which is substituted by hydroxy groups at the 3beta, 12beta and 20 pro-S positions, in which the hydroxy group at position 20 has been converted to the corresponding beta-D-glucopyranoside, and in which a double bond has been introduced at the 24-25 position. It has a role as a plant metabolite, an antineoplastic agent, a hepatoprotective agent, an anti-allergic agent and an anti-inflammatory agent. It is a beta-D-glucoside, a 12beta-hydroxy steroid, a ginsenoside, a tetracyclic triterpenoid, a 3beta-hydroxy steroid and a 3beta-hydroxy-4,4-dimethylsteroid. It derives from a hydride of a dammarane. ginsenoside C-K has been reported in Panax ginseng and Fusarium sacchari with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 100 mg/mL (~160.55 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.75 mg/mL (4.42 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.75 mg/mL (4.42 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.75 mg/mL (4.42 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6055 mL | 8.0274 mL | 16.0547 mL | |
| 5 mM | 0.3211 mL | 1.6055 mL | 3.2109 mL | |
| 10 mM | 0.1605 mL | 0.8027 mL | 1.6055 mL |