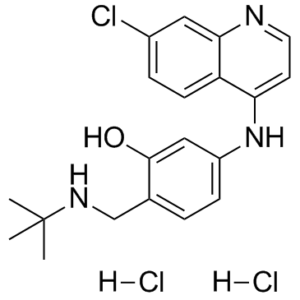
GSK369796 dihydrochloride is a novel, potent and effective antimalarial and inhibits hERG potassium ion channel repolarization with an IC50 of 7.5 μM. It is a 4-aminoquinoline drug candidate selected and developed as part of a public-private partnership between academics at Liverpool, MMV, and GSK pharmaceuticals. This molecule was rationally designed based on chemical, toxicological, pharmacokinetic, and pharmacodynamic considerations and was selected based on excellent activity against Plasmodium falciparum in vitro and rodent malaria parasites in vivo. The optimized chemistry delivered this novel synthetic quinoline in a two-step procedure from cheap and readily available starting materials. The molecule has a full industry standard preclinical development program allowing first into humans to proceed. Employing chloroquine (1) and amodiaquine (2) as comparator molecules in the preclinical plan, the first preclinical dossier of pharmacokinetic, toxicity, and safety pharmacology has also been established for the 4-aminoquinoline antimalarial class. These studies have revealed preclinical liabilities that have never translated into the human experience. This has resulted in the availability of critical information to other drug development teams interested in developing antimalarials within this class.
Physicochemical Properties
| Molecular Formula | C20H24CL3N3O |
| Molecular Weight | 428.7831 |
| Exact Mass | 427.098 |
| CAS # | 1010411-21-8 |
| Related CAS # | 459133-38-1;1010411-21-8 (HCl); |
| PubChem CID | 51071988 |
| Appearance | Light yellow to yellow solid powder |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 27 |
| Complexity | 428 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | UDVALKJFXQVZSI-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H22ClN3O.2ClH/c1-20(2,3)23-12-13-4-6-15(11-19(13)25)24-17-8-9-22-18-10-14(21)5-7-16(17)18;;/h4-11,23,25H,12H2,1-3H3,(H,22,24);2*1H |
| Chemical Name | 2-[(tert-Butylamino)methyl]-5-[(7-chloroquinolin-4-yl)amino]phenol Dihydrochloride |
| Synonyms | GSK369796 dihydrochloride; GSK 369796; GSK-369796; GSK369796; N-tert-Butyl isoquine. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | GSK369796 DiHClide has an IC50 of 11.2±2.2, 12.6±5.3, and 13.2±3.2 nM, respectively, which inhibits the development of Plasmodium falciparum strains 3D7c, HB3c, and K1d in vitro. In mice, GSK369796 dihydrochloride (chemical 4) exhibited stronger protein binding (93% vs.74%) than desethylamodiaquine, although human protein binding (88% vs.86%) was comparable. GSK369796 DiHClide has an IC50 of 7.5±0.8 μM and can also prevent hERG potassium channel repolarization[1]. |
| ln Vivo | Plasmodium berghei ANKA is inhibited in vivo by GSK369796 DiHClide, with ED50 and ED90 values of 2.8 and 4.7 mg/kg, respectively [1]. |
| References |
[1]. Candidate selection and preclinical evaluation of N-tert-butyl isoquine (GSK369796), an affordable and effective 4-aminoquinoline antimalarial for the 21st century. J Med Chem. 2009 Mar 12;52(5):1408-15. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~233.22 mM) H2O : ~50 mg/mL (~116.61 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3322 mL | 11.6610 mL | 23.3220 mL | |
| 5 mM | 0.4664 mL | 2.3322 mL | 4.6644 mL | |
| 10 mM | 0.2332 mL | 1.1661 mL | 2.3322 mL |