Physicochemical Properties
| Molecular Formula | C17H12FN3 |
| Molecular Weight | 277.30 |
| Exact Mass | 277.101 |
| CAS # | 2449301-27-1 |
| PubChem CID | 137319705 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.33±0.1 g/cm3(Predicted) |
| Boiling Point | 540.4±50.0 °C(Predicted) |
| LogP | 4.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 21 |
| Complexity | 441 |
| Defined Atom Stereocenter Count | 0 |
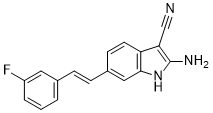
| SMILES | C1=CC(=CC(=C1)F)/C=C/C2=CC3=C(C=C2)C(=C(N3)N)C#N |
| InChi Key | CGXBPMZRTMXEIA-SNAWJCMRSA-N |
| InChi Code | InChI=1S/C17H12FN3/c18-13-3-1-2-11(8-13)4-5-12-6-7-14-15(10-19)17(20)21-16(14)9-12/h1-9,21H,20H2/b5-4+ |
| Chemical Name | 2-amino-6-[(E)-2-(3-fluorophenyl)ethenyl]-1H-indole-3-carbonitrile |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | IC50: 160 nM (USP20/Ub-Rho)[1] |
| ln Vitro | USP20-mediated breaking of protein-ubiquitin bonds is inhibited by GSK2643943A [2]. GSK2643943A sensitizes SCC9 cells to oHSV-1-induced oncolysis at 1 μM and 5 μM over night [2]. When 0.01 MOI T1012G was added to SCC9, the generation of virus increased significantly in response to GSK2643943A (1 μM) [2]. |
| ln Vivo | In SCC9 tumors, GSK2643943A (5 mg/kg, ip, daily for 6 days) improves oHSV-1-induced oncolysis[2]. In SCC7 cells, GSK2643943A (2.5 mg/kg, ip, daily for 9 days) regulates the replication of oHSV-1 T1012G and oncolysis[2]. |
| Cell Assay |
Cell Viability Assay[2] Cell Types: SCC9 cells Tested Concentrations: 1 μM, 5 μM (GSK+0.01 MOI T1012) 1 μM (GSK+0.01 MOI/ 1 MOI T1012) Incubation Duration:overnight Experimental Results: Displayed a significant drop in viability ( R50%) (5 μM GSK+0.01 MOI T1012 infection) and 50% loss of SCC9 viability (1 μM GSK+0.01 MOI T1012 infection) . Remarkably diminished the viability of SCC9 upon exposure to 1 MOI T1012G infection. Western Blot Analysis[2] Cell Types: SCC9 cells Tested Concentrations: 1 μM Incubation Duration: 3h, 9 h and 20 h Experimental Results: Generally up-regulated the expression of viral proteins at various phases. RT-PCR[2] Cell Types: SCC9 cells Tested Concentrations: 1 μM Incubation Duration: 9 h Experimental Results: Dramatically increased the accumulation of viral ICP8 and VP16 mRNA in SCC9 cells. |
| Animal Protocol |
Animal/Disease Models: The subcutaneous (sc) xenograft model[2]. (SCC9 or SCC7 cells (8×106 cells or 1×106 cells), 5weeks old, female, BALB/c nude mice or C3H/HeN mice, four groups, n = 6-7, per group)[2] Doses: 5 mg/kg Route of Administration: GSK2643943A (alone): intraperitoneal (ip) administration, daily, for 6 days. GSK2643943A (combination): intraperitoneal (ip) administration, daily for 6 days + intratumoral injection with 50 mL of 1×106 PFU T1012G in PBS on day 1, day 4, and day 7. Experimental Results: Caused a visible drop of tumor volumes and Dramatically decreased the tumor volumes in mice with combined treatment of GSK2643943A and oHSV-1 T1012G. Increased slightly viral ICP0 and gD mRNA accumulation in SCC9 tumors. Animal/Disease Models: The SCC7 mouse model[2]. Doses: 2.5 mg/kg Route of Administration: GSK2643943A (alone): intraperitoneal (ip) administration, daily, for 9 days. GSK2643943A (combination): intraperitoneal (ip) administration, daily, for 9 days + intratumoral injection, with 50 mL of 1×107 PFU T1012G in PBS on days 1, 4, 7, and 10. |
| References |
[1]. Targeting the Ubiquitin Proteasome System in Cancer. Shahzad, Hafiz Naveed (2018). Neoplasm Targeting the Ubiquitin Proteasome System in Cancer. , 10.5772/intechopen.69560(Chapter 8). [2]. USP18 and USP20 restrict oHSV-1 replication in resistant human oral squamous carcinoma cell line SCC9 and affect the viability of SCC9 cells. Mol Ther Oncolytics. 2021 Nov 11;23:477-487. |
Solubility Data
| Solubility (In Vitro) | DMSO: ≥ 125 mg/mL (450.78 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.50 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (7.50 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6062 mL | 18.0310 mL | 36.0620 mL | |
| 5 mM | 0.7212 mL | 3.6062 mL | 7.2124 mL | |
| 10 mM | 0.3606 mL | 1.8031 mL | 3.6062 mL |