GSK1379725A (AU1) is the first small molecule selective for BPTF over Brd4, as an inhibitor of Brd4 and a selective BPTF ligand with Kd of 2.8 μM. AU1 is active in a cell-based reporter assay. No binding is detected with Brd4. Three new Brd4 inhibitors with submicromolar affinity were also discovered. Brd4 hits were validated in a thermal stability assay and potency determined via fluorescence anisotropy. The speed, ease of interpretation, and low protein concentration needed for protein-observed (19)F NMR experiments in a multiprotein format offers a new method to discover and characterize selective ligands for bromodomain-containing proteins.
Physicochemical Properties
| Molecular Formula | C23H23FN6O3 |
| Molecular Weight | 450.465527772903 |
| Exact Mass | 450.181 |
| CAS # | 1802251-00-8 |
| Related CAS # | (S)-GSK1379725A;2359618-49-6 |
| PubChem CID | 44525934 |
| Appearance | White to off-white solid powder |
| LogP | 3.4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 33 |
| Complexity | 662 |
| Defined Atom Stereocenter Count | 0 |
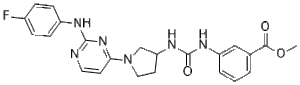
| SMILES | FC1C=CC(=CC=1)NC1=NC=CC(=N1)N1CCC(C1)NC(NC1C=CC=C(C(=O)OC)C=1)=O |
| InChi Key | LLVPDBRSUHNJQQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H23FN6O3/c1-33-21(31)15-3-2-4-18(13-15)27-23(32)28-19-10-12-30(14-19)20-9-11-25-22(29-20)26-17-7-5-16(24)6-8-17/h2-9,11,13,19H,10,12,14H2,1H3,(H,25,26,29)(H2,27,28,32) |
| Chemical Name | 3-(3-{1-[2-(4-Fluoro-phenylamino)-pyrimidin-4-yl]-pyrrolidin-3-yl}-ureido)-benzoic acid methyl ester |
| Synonyms | AU1; AU-1; AU 1; GSK1379725A; GSK 1379725 A; GSK-1379725-A; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
BPTF bromodomain (Ki = 1.8 μM, determined by HTRF assay) [1] - Brd4 bromodomain BD1 (Ki = 0.5 μM, determined by HTRF assay) [1] - Brd4 bromodomain BD2 (Ki = 2.3 μM, determined by HTRF assay) [1] |
| ln Vitro |
There is an upper limit on the chemical exchange rate based on the separation of the bound and unbound resonances (171 Hz) from the NMR titration of GSK1379725A. The upper limit of Kd, if a series of protein-small molecule interactions (e.g., chymotrypsin: proflavin k1=1.2×108 M-1 s-1) have a correlation rate of 1×108 M-1 s-1 as their upper limit, follows. An estimate of 8 μM was made based on this experiment. By using unlabeled BPTF as a supplementary direct binding assay, ITC was utilized to assess non-fluorinated proteins with greater accuracy. The intermediate exchange resonance broadening we discovered by PrOF NMR is consistent with the Kd of 2.8 μM that we measured. A full selectivity panel against other bromodomains is still required, even though GSK1379725A has been demonstrated to be selective against Brd4. Of the five cell assays with an EC50 of 500 nM, only GSK1379725A was shown to be active using a ChEMBL database search. Furthermore, the kinase activity of GSK1379725A has not been documented, despite the growing popularity of PKIS libraries for screening [1]. Binds specifically to the bromodomains of BPTF and Brd4 with moderate affinity, showing no significant binding to other bromodomains (e.g., Brd2, Brd3, CBP) with Ki > 20 μM [1] - Inhibited the interaction between BPTF bromodomain and acetylated histone H4 peptide (H4K5ac/K8ac/K12ac/K16ac) in 19F NMR binding assays, with a binding affinity consistent with HTRF-derived Ki values [1] - Blocked Brd4 bromodomain (BD1/BD2) binding to acetylated histone H3 peptide (H3K14ac) in HTRF competition assays, reducing the signal intensity by ~70% at 10 μM concentration [1] - Exhibited weak inhibitory effect on Brd4-dependent transcription in a HEK293T reporter gene assay, with ~30% inhibition at 20 μM GSK1379725A [1] |
| Enzyme Assay |
Protein-observed 19F NMR binding assay: Recombinant BPTF bromodomain protein was labeled with a fluorine-containing probe and incubated with various concentrations of GSK1379725A in NMR buffer. The mixture was transferred to NMR tubes, and 19F NMR spectra were recorded at 25°C. Chemical shift changes of the fluorine probe were monitored to assess binding, and the dissociation constant (Ki) was calculated by fitting the chemical shift data to a 1:1 binding model [1] - HTRF competition assay for bromodomain-acetylated peptide interaction: Recombinant BPTF or Brd4 (BD1/BD2) bromodomain protein was incubated with biotinylated acetylated histone peptide (H4K5ac/K8ac/K12ac/K16ac for BPTF; H3K14ac for Brd4) and europium-labeled anti-biotin antibody in assay buffer. GSK1379725A was added at different concentrations, and the mixture was incubated at 37°C for 1 hour. Time-resolved fluorescence resonance energy transfer (HTRF) signal was measured, and the Ki value was determined by analyzing the competition curve [1] |
| Cell Assay |
HEK293T Brd4-dependent reporter gene assay: HEK293T cells were transfected with a luciferase reporter plasmid driven by a Brd4-responsive promoter and a Renilla luciferase plasmid as an internal control. After 24 hours of transfection, cells were treated with GSK1379725A (0.1-20 μM) for 16 hours. Luciferase activity was measured using a dual-luciferase assay system, and the inhibition rate was calculated relative to the vehicle control [1] - Bromodomain selectivity profiling assay: Recombinant bromodomain proteins from 24 different human bromodomain-containing proteins were incubated with GSK1379725A (10 μM) in HTRF assay buffer. Binding affinity was assessed by measuring the competition between GSK1379725A and acetylated histone peptides, and selectivity was determined by comparing Ki values across all tested bromodomains [1] |
| References |
[1]. Dual Screening of BPTF and Brd4 Using Protein-Observed Fluorine NMR Uncovers New Bromodomain Probe Molecules. ACS Chem Biol. 2015 Oct 16;10(10):2246-56. |
| Additional Infomation |
GSK1379725A is a small-molecule probe compound identified through dual screening of BPTF and Brd4 bromodomains using protein-observed fluorine NMR [1] - Its mechanism of action involves binding to the acetyllysine-binding pocket of BPTF and Brd4 bromodomains, thereby blocking their interaction with acetylated histones and interfering with chromatin-dependent transcriptional regulation [1] - Serves as a valuable chemical tool for studying the biological functions of BPTF and Brd4 bromodomains, particularly their cooperative roles in transcriptional control and chromatin remodeling [1] - Exhibits moderate potency and good selectivity, making it suitable for in vitro mechanistic studies but not for in vivo applications due to limited solubility and bioavailability (not formally tested but inferred from structural properties) [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~221.99 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.55 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (5.55 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.55 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2199 mL | 11.0995 mL | 22.1990 mL | |
| 5 mM | 0.4440 mL | 2.2199 mL | 4.4398 mL | |
| 10 mM | 0.2220 mL | 1.1100 mL | 2.2199 mL |