GNE-477 is a novel, potent and efficacious dual inhibitor of PI3K (IC50=4 nM)/mTOR(Ki=21 nM). The PI3K/AKT/mTOR signaling pathway is regarded as an attractive area of research for the identification of oral small molecule inhibitors. PI3K-α, β, δ, and were all found to be inhibited in vitro by GNE-477, with IC50 values of 4, 86, 6, and 15 nM, respectively. A direct comparison of GNE-477 with its des-methyl analog in vivo showed that mice and dogs also exhibit the trend of decreased in vivo clearance that is seen in rats. In contrast to GNE-477, which had a low clearance in dogs, the des-methyl analog was cleared at a rate that was two-thirds that of hepatic blood flow. In a study evaluating the tumor growth inhibition of a PC3 tumor xenograft10 over a period of 14 days, stasis was reached at a dose of GNE-477 of 20 mg/kg QD, and significant inhibition was discovered with doses as low as 1 mg/kg QD. GNE-477 was generally well tolerated throughout this study, as evidenced by acceptable weight loss levels comparable to those in the vehicle cohort.
Physicochemical Properties
| Molecular Formula | C21H28N8O3S2 |
| Molecular Weight | 504.62882 |
| Exact Mass | 504.172 |
| Elemental Analysis | C, 49.98; H, 5.59; N, 22.21; O, 9.51; S, 12.71 |
| CAS # | 1032754-81-6 |
| Related CAS # | 1032754-81-6 |
| PubChem CID | 25207689 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 693.6±65.0 °C at 760 mmHg |
| Flash Point | 373.3±34.3 °C |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.717 |
| LogP | 1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 34 |
| Complexity | 779 |
| Defined Atom Stereocenter Count | 0 |
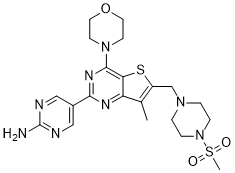
| SMILES | NC1=NC=C(C2=NC(N3CCOCC3)=C4C(C(C)=C(CN5CCN(S(=O)(C)=O)CC5)S4)=N2)C=N1 |
| InChi Key | AKKCGLXULFRAET-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H28N8O3S2/c1-14-16(13-27-3-5-29(6-4-27)34(2,30)31)33-18-17(14)25-19(15-11-23-21(22)24-12-15)26-20(18)28-7-9-32-10-8-28/h11-12H,3-10,13H2,1-2H3,(H2,22,23,24) |
| Chemical Name | 5-[7-methyl-6-[(4-methylsulfonylpiperazin-1-yl)methyl]-4-morpholin-4-ylthieno[3,2-d]pyrimidin-2-yl]pyrimidin-2-amine |
| Synonyms | GNE-477; GNE477; GNE 477 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
PI3Kα (IC50 = 4 nM); mTOR (IC50 = 21 nM) GNE-477 (Compound 8) has improved potency in the MCF7.1 cell proliferation assay with an EC50 of 143 nM[1]. GNE-477 is a dual inhibitor of PI3K and mTOR kinase. It inhibits PI3K-α with an IC₅₀ of 4 nM, PI3K-β with an IC₅₀ of 86 nM, PI3K-δ with an IC₅₀ of 6 nM, PI3K-γ with an IC₅₀ of 15 nM, and mTOR kinase (Ki(app)) with a value of 21 nM. [1] |
| ln Vitro |
GNE-477 (Compound 8) has improved potency in the MCF7.1 cell proliferation assay with an EC50 of 143 nM[1]. GNE-477 inhibited the proliferation of MCF7.1 cells (a HER2-transfected clone of MCF7 with a PI3K E545K activating mutation) with an EC₅₀ of 15 nM. It also inhibited the proliferation of PC3 (a PTEN-negative cancer cell line) with an EC₅₀ of 174 nM. [1] GNE-477 inhibited the proliferation of MCF7.1 cells (a HER2-transfected clone of MCF7 with a PI3K E545K activating mutation) with an EC₅₀ of 15 nM. It also inhibited the proliferation of PC3 (a PTEN-negative cancer cell line) with an EC₅₀ of 174 nM. [1] |
| ln Vivo |
GNE-477 also exhibits stasis in a PC3 tumor growth inhibition study. In a 14-day experiment examining the tumor growth inhibition of a PC3 tumor xenograft, stasis is reached at a dose of 20 mg/kg QD, and significant inhibition is seen at doses as low as 1 mg/kg QD. The acceptable levels of weight loss comparable to those seen with the animals in the vehicle cohort during this study show that GNE-477 is generally well tolerated.[1] In a PC3 tumor xenograft model in mice, oral administration of GNE-477 at 20 mg/kg once daily (QD) for 14 days resulted in tumor stasis. Significant tumor growth inhibition was observed at doses as low as 1 mg/kg QD. Partial responses (tumor volume reduction >50%) and complete responses (100% reduction) were observed in some animals during the study. [1] In a PC3 tumor xenograft model in mice, oral administration of GNE-477 at 20 mg/kg once daily (QD) for 14 days resulted in tumor stasis. Significant tumor growth inhibition was observed at doses as low as 1 mg/kg QD. Partial responses (tumor volume reduction >50%) and complete responses (100% reduction) were observed in some animals during the study. [1] |
| Enzyme Assay |
The biochemical potency against PI3K isoforms and mTOR kinase was determined using enzyme inhibition assays. [1] The biochemical potency against PI3K isoforms and mTOR kinase was determined using enzyme inhibition assays. [1] |
| Cell Assay |
Cell proliferation assays were conducted using MCF7.1 and PC3 cell lines. Cells were treated with GNE-477, and proliferation was measured, likely using a viability or metabolic activity endpoint, to determine EC₅₀ values. [1] Cell proliferation assays were conducted using MCF7.1 and PC3 cell lines. Cells were treated with GNE-477, and proliferation was measured, likely using a viability or metabolic activity endpoint, to determine EC₅₀ values. [1] |
| Animal Protocol |
Mice, Rats and Dogs[1]. The GNE-477 HCl salt is administered intravenously to female nu/nu mice (1 mg/kg) and orally to female nu/nu mice (5 mg/kg) as a solution in 80% PEG. The GNE-477 TFA salt is administered intravenously to male rats (1 mg/kg) and orally to male rats (5 mg/kg) as a solution in 80% PEG. The GNE-477 HCl salt is administered to male beagle dogs as an intravenous solution (1 mg/kg) in 10% HP-CD and as an oral suspension (2 mg/kg) in MCT. GNE-477's effectiveness in the PC3-NCI tumor xenograft model is studied. At the conclusion of the study (day 14), the percentage of tumor growth inhibition (TGI) is assessed and contrasted with the vehicle control group. For the efficacy study, GNE-477 was formulated in a vehicle consisting of 0.5% methylcellulose and 0.2% Tween-80 (MCT). The compound was administered orally (po) once daily (QD) at doses of 1, 5, 20, and 75 mg/kg to mice bearing PC3 tumor xenografts. The study duration was 14 days. Tumor volumes and body weights were monitored. [1] For the efficacy study, GNE-477 was formulated in a vehicle consisting of 0.5% methylcellulose and 0.2% Tween-80 (MCT). The compound was administered orally (po) once daily (QD) at doses of 1, 5, 20, and 75 mg/kg to mice bearing PC3 tumor xenografts. The study duration was 14 days. Tumor volumes and body weights were monitored. [1] |
| ADME/Pharmacokinetics |
GNE-477 showed improved in vivo clearance compared to its des-methyl analog (compound 1). In rats, GNE-477 had a total plasma clearance (Cl) of 22 mL/min/kg, an unbound clearance (Clu) of 220 mL/min/kg, and plasma protein binding (PPB) of 90%. In mice, total clearance was 34 mL/min/kg, unbound clearance was 340 mL/min/kg, and plasma protein binding was 90%. In dogs, total clearance was 4 mL/min/kg, unbound clearance was 80 mL/min/kg, and plasma protein binding was 95%. The low clearance in dogs was particularly notable compared to the high clearance of the des-methyl analog. [1] GNE-477 showed improved in vivo clearance compared to its des-methyl analog (compound 1). In rats, GNE-477 had a total plasma clearance (Cl) of 22 mL/min/kg, an unbound clearance (Clu) of 220 mL/min/kg, and plasma protein binding (PPB) of 90%. In mice, total clearance was 34 mL/min/kg, unbound clearance was 340 mL/min/kg, and plasma protein binding was 90%. In dogs, total clearance was 4 mL/min/kg, unbound clearance was 80 mL/min/kg, and plasma protein binding was 95%. The low clearance in dogs was particularly notable compared to the high clearance of the des-methyl analog. [1] |
| Toxicity/Toxicokinetics |
During the 14-day PC3 xenograft efficacy study, treatment with GNE-477 was generally well-tolerated. All dose groups, including the vehicle group, exhibited acceptable and comparable body weight loss of less than 10% relative to the starting mass. [1] During the 14-day PC3 xenograft efficacy study, treatment with GNE-477 was generally well-tolerated. All dose groups, including the vehicle group, exhibited acceptable and comparable body weight loss of less than 10% relative to the starting mass. [1] |
| References |
[1]. Identification of GNE-477, a potent and efficacious dual PI3K/mTOR inhibitor. Bioorg Med Chem Lett. 2010 Apr 15;20(8):2408-11. |
| Additional Infomation |
GNE-477 is a 6-aryl morpholino thienopyrimidine derivative identified from efforts to improve the dissolution rate of high-melting-point PI3K inhibitors by adding a methyl group to the thienopyrimidine core to disrupt planarity. This modification generally reduced in vivo clearance across species while maintaining potent enzymatic and cellular activity. It represents a potent and efficacious dual PI3K/mTOR inhibitor with a favorable pharmacokinetic profile. [1] GNE-477 is a 6-aryl morpholino thienopyrimidine derivative identified from efforts to improve the dissolution rate of high-melting-point PI3K inhibitors by adding a methyl group to the thienopyrimidine core to disrupt planarity. This modification generally reduced in vivo clearance across species while maintaining potent enzymatic and cellular activity. It represents a potent and efficacious dual PI3K/mTOR inhibitor with a favorable pharmacokinetic profile. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO: 25 ~16.7 mg/mL (33~49.5 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.67 mg/mL (3.31 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 16.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.67 mg/mL (3.31 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 16.7 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9816 mL | 9.9082 mL | 19.8165 mL | |
| 5 mM | 0.3963 mL | 1.9816 mL | 3.9633 mL | |
| 10 mM | 0.1982 mL | 0.9908 mL | 1.9816 mL |