With a Ki of 0.2 nM, GDC-0326 is a brand-new, powerful, and selective PI3K (-Isoform of Phosphoinositide 3-Kinase) inhibitor. Inhibitors of the class I phosphoinositide 3-kinase (PI3K) isoform PI3Kα have drawn a lot of interest due to their potential application in the treatment of cancer. Despite the particular appeal of targeting PI3Kα , it has proven difficult to inhibit this isoform with selectivity. High selectivity is demonstrated by GDC-0326 versus other kinases. In humans, GDC-0326 has a low plasma CL. There are four class I PI3K isoforms (α, β, δ, and γ) ) in the PI3 kinase family. PI3K is the isoform of these that is most frequently linked to cancer.
Physicochemical Properties
| Molecular Formula | C19H22N6O3 |
| Molecular Weight | 382.416383266449 |
| Exact Mass | 382.175 |
| Elemental Analysis | C, 59.67; H, 5.80; N, 21.98; O, 12.55 |
| CAS # | 1282514-88-8 |
| Related CAS # | 1282514-88-8 |
| PubChem CID | 58204997 |
| Appearance | White to off-white solid powder |
| LogP | 1.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 28 |
| Complexity | 564 |
| Defined Atom Stereocenter Count | 1 |
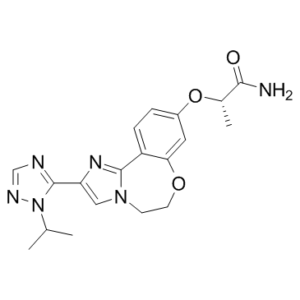
| SMILES | C[C@@H](C(=O)N)OC1=CC2=C(C=C1)C3=NC(=CN3CCO2)C4=NC=NN4C(C)C |
| InChi Key | SIKYDKLGPWRPMZ-LBPRGKRZSA-N |
| InChi Code | InChI=1S/C19H22N6O3/c1-11(2)25-19(21-10-22-25)15-9-24-6-7-27-16-8-13(28-12(3)17(20)26)4-5-14(16)18(24)23-15/h4-5,8-12H,6-7H2,1-3H3,(H2,20,26)/t12-/m0/s1 |
| Chemical Name | (2S)-2-[[2-(2-propan-2-yl-1,2,4-triazol-3-yl)-5,6-dihydroimidazo[1,2-d][1,4]benzoxazepin-9-yl]oxy]propanamide |
| Synonyms | GDC-0326; GDC 0326; GDC0326 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
PI3Kα (Ki = 0.2 nM); PI3Kβ (Ki = 26.6 nM); PI3Kδ (Ki = 4 nM); PI3Kγ (Ki = 10.2 nM) The PI3Kα specific inhibitor GDC-0326 also achieves a very high level of selectivity over other kinases in addition to selectivity over the other class I isoforms. When tested on cytochrome P450 enzymes, GDC-0326 was not an inhibitor[1]. |
| ln Vitro |
The PI3Kα specific inhibitor GDC-0326 also achieves a very high level of selectivity over other kinases in addition to selectivity over the other class I isoforms. When tested on cytochrome P450 enzymes, GDC-0326 was not an inhibitor[1]. Treatment of βTC3 cells (derived from RIP1-Tag2 mouse pancreatic neuroendocrine tumors) with 1 µM GDC-0326 for 2 hours reduced AKT phosphorylation, similar to the pan-PI3K inhibitor GDC-0941[2] In βTC3 cells, GDC-0326 (1 µM) induced apoptosis after 48 hours of treatment, as assessed by cleaved caspase-3 immunofluorescence[2] Pre-treatment of βTC3 and βTC4 cells with GDC-0326 (and BYL719, another p110α inhibitor) under low glucose conditions stimulated cell death[2] Incubation of mouse lung endothelial cells (ECs) with GDC-0326 reduced cell migration, but had no effect on cell proliferation or survival[2] |
| ln Vivo |
GDC-0326 has a consistently low clearance and a high oral bioavailability in all tested species, allowing for significant sustained free drug levels[1]. Daily oral administration of GDC-0326 (12 mg/kg/day) to RIP1-Tag2 mice (a model of pancreatic neuroendocrine tumors) from 12 to 14 weeks of age significantly improved animal lifespan compared to vehicle-treated controls[2] The same treatment regimen resulted in an approximately 60% reduction in total tumor burden[2] GDC-0326 treatment (12 mg/kg/day for 14 days) significantly reduced the incidence of liver micrometastases (0% vs. control) and lymph node (LN) metastasis (incidence reduced by four times) compared to vehicle-treated mice[2] For mice that did show LN infiltration, the average number of T antigen-positive nodes was also decreased (1.0 vs. 1.6 in controls)[2] Treatment did not alter tumor cell proliferation in vivo, as assessed by Ki67 immunostaining[2] Treatment led to a reduced number of hypervascularized “red islets”[2] Treatment significantly decreased tumor vascular area, indicating anti-angiogenic activity[2] Treatment resulted in an overall increase in tumor cell death, as assessed by TUNEL staining[2] |
| Cell Assay |
At 1 × 106 cells per dish, TC3 cells are plated. Cells are treated for 2 hours with vehicle, GDC-0941 (1 μM), GDC-0326 (1 μM), TGX-221 (0.5 μM), IC87114 (5 μM), BKM120 (0.8 μM) or BYL719 (1 μM) 24 hours later, after which they are lysed. AKT phosphorylation assay: Exponentially growing βTC3 cells were treated with vehicle or GDC-0326 (1 µM) for 2 hours. Cells were then lysed, and total cell lysates were subjected to western blot analysis using antibodies against phospho-AKT (S473) and total AKT to assess inhibition of PI3K signaling[2] Apoptosis assay (Cleaved Caspase-3): βTC3 cells were treated with vehicle or GDC-0326 (1 µM) for 48 hours. Cells were then fixed and processed for immunofluorescence staining using an antibody against cleaved caspase-3. The percentage of cleaved caspase-3 positive cells was quantified to measure apoptosis[2] Cell death under low nutrients: βTC3 and βTC4 cells were treated with GDC-0326 under low glucose culture conditions. Cell death was assessed, although the specific assay method (e.g., viability dye, caspase activity) is not detailed in the main text[2] Endothelial cell migration assay: Mouse lung endothelial cells were incubated with GDC-0326. Their migratory capacity was assessed, while proliferation and survival were also monitored. The specific migration assay method (e.g., transwell, scratch wound) is not detailed[2] |
| Animal Protocol |
Mice: GDC-0326 is administered intravenously to female NCR nude mice at a dose of 1 mg/kg in a solution of 60% PEG400/ 10% ethanol and orally at a dose of 25 mg/kg in a solution of 0.5% methylcellulose/ 0.2% Tween 80 (MCT). Rats: 1 mg/kg of GDC-0326 prepared in 60% PEG400/10% ethanol is administered intravenously to male Sprague-Dawley rats. Male Sprague-Dawley rats are administered 5 mg/kg of GDC-0326 orally (PO) in a 0.5% methylcellulose/0.2% Tween 80 (MCT) solution. Therapeutic efficacy study in RIP1-Tag2 mice: RIP1-Tag2 mice at 12 weeks of age were treated daily by oral gavage with GDC-0326 at a dose of 12 mg/kg/day. The compound was formulated in a vehicle consisting of 0.5% (w/v) methylcellulose and 0.2% (w/v) polysorbate 80 in de-ionized water. Treatment continued until the mice reached 14 weeks of age. Mice were then euthanized for analysis of tumor burden, metastasis, and tissue biomarkers[2] Pharmacodynamic study: RIP1-Tag2 mice bearing tumors were treated with a single dose of GDC-0326 (12 mg/kg, po) or vehicle. Mice were euthanized 3 hours post-dose, and individual tumors were harvested for western blot analysis to assess inhibition of AKT and S6 phosphorylation[2] |
| Toxicity/Toxicokinetics |
In the described in vivo study, daily oral administration of GDC-0326 at 12 mg/kg/day for 2 weeks to RIP1-Tag2 mice was tolerated, as evidenced by a significant improvement in lifespan compared to controls[2] The article does not report specific toxicity endpoints (e.g., body weight loss, clinical observations, histopathology of major organs) or detailed toxicokinetic data for GDC-0326[2] |
| References |
[1]. J Med Chem. 2016 Feb 11;59(3):985-1002. [2]. Clin Cancer Res. 2016 Dec 1;22(23):5805-5817. |
| Additional Infomation |
(2s)-2-({2-[1-(Propan-2-Yl)-1h-1,2,4-Triazol-5-Yl]-5,6-Dihydroimidazo[1,2-D][1,4]benzoxazepin-9-Yl}oxy)propanamide is an organic molecular entity. GDC-0326 is a newly developed small molecule inhibitor with selectivity for the p110α isoform of PI3K[2] Its anti-tumor activity in the RIP1-Tag2 pancreatic neuroendocrine tumor model is attributed to a dual mechanism: 1) a direct pro-apoptotic effect on tumor cells (cell-intrinsic), and 2) a prominent anti-angiogenic effect mediated through inhibition of endothelial cell function, leading to reduced tumor vascularity and subsequent tumor cell death[2] A key finding is that GDC-0326 blocks tumor cell dissemination and metastasis in this model[2] The study provides a rationale for the selective targeting of p110α in pancreatic neuroendocrine tumors and suggests it may be an alternative or complementary strategy to mTOR inhibition[2] |
Solubility Data
| Solubility (In Vitro) |
DMSO: 76~100 mg/mL (198.7~261.5mM) Ethanol: ~19 mg/mL (~49.7 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6149 mL | 13.0746 mL | 26.1493 mL | |
| 5 mM | 0.5230 mL | 2.6149 mL | 5.2299 mL | |
| 10 mM | 0.2615 mL | 1.3075 mL | 2.6149 mL |