Fostamatinib disodium hexahydrate (formerly R-935788; R 935788; R-788; R788; trade name: Tavalisse) is the disodium salt and hexa-hydrated form of Fostamatinib which is a prodrug of the active metabolite R406. In order to treat thrombocytopenia in adult patients with persistent or chronic immune thrombocytopenia (ITP), the FDA approved the oral bioactive and selective Syk inhibitor fostamatinib in 2018. Using a 41 nM IC50 in a cell-free assay, it inhibits Syk kinase.
Physicochemical Properties
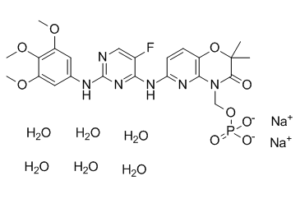
| Molecular Formula | C₂₃H₂₄FN₆NA₂O₉P.₆H₂O | |
| Molecular Weight | 732.51 | |
| Exact Mass | 732.18 | |
| Elemental Analysis | C, 37.71; H, 4.95; F, 2.59; N, 11.47; Na, 6.28; O, 32.76; P, 4.23 | |
| CAS # | 914295-16-2 | |
| Related CAS # | Fostamatinib Disodium;1025687-58-4;Fostamatinib;901119-35-5 | |
| PubChem CID | 24828759 | |
| Appearance | White to gray solid powder | |
| LogP | 3.241 | |
| Hydrogen Bond Donor Count | 8 | |
| Hydrogen Bond Acceptor Count | 21 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 48 | |
| Complexity | 893 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | [Na].O=C1C(C)(C)OC2C(=NC(NC3C(F)=CN=C(NC4C=C(OC)C(OC)=C(OC)C=4)N=3)=CC=2)N1COP(O)(O)=O.O.[Na].O.O.O.O.O |
|
| InChi Key | ZQGJCHHKJNSPMS-UHFFFAOYSA-L | |
| InChi Code | InChI=1S/C23H26FN6O9P.2Na.6H2O/c1-23(2)21(31)30(11-38-40(32,33)34)20-14(39-23)6-7-17(28-20)27-19-13(24)10-25-22(29-19)26-12-8-15(35-3)18(37-5)16(9-12)36-4;;;;;;;;/h6-10H,11H2,1-5H3,(H2,32,33,34)(H2,25,26,27,28,29);;;6*1H2/q;2*+1;;;;;;/p-2 | |
| Chemical Name | disodium;[6-[[5-fluoro-2-(3,4,5-trimethoxyanilino)pyrimidin-4-yl]amino]-2,2-dimethyl-3-oxopyrido[3,2-b][1,4]oxazin-4-yl]methyl phosphate;hexahydrate | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Syk (IC50 = 41 nM) R406, a spleen tyrosine kinase (Syk) inhibitor, is prodrugged by R788. R788 has a Ki of 30 nM and functions as a competitive inhibitor of ATP binding. With an EC50 of 56 nM, R788 dose-dependently prevents anti-IgE-mediated CHMC degranulation. R788 also prevents the release of LTC4 and cytokines and chemokines, such as TNFα, IL-8, and GM-CSF, that are triggered by anti-IgE. All phosphorylation events downstream of Syk signaling are inhibited when R788 inhibits Syk. The most potent inhibitor of IL-4 and IL-2 receptor signaling in CHMC is R788, followed by FcρRI signaling. R788 specifically inhibits human mast cells, neutrophils, and macrophages' FcγR signaling. R788 has the ability to prevent immune complex-mediated local inflammatory injury.R788 causes most of the tested DLBCL cell lines to undergo apoptosis[1]. R788 selectively inhibits both tonic- and ligand-induced BCR signaling (autophosphorylation of SYK525/526 and SYK-dependent phosphorylation of the B-cell linker protein [BLNK]) in R788-sensitive DLBCL cell lines[2]. |
| ln Vitro |
R406, a spleen tyrosine kinase (Syk) inhibitor, is prodrugged by R788. R788 has a Ki of 30 nM and functions as a competitive inhibitor of ATP binding. With an EC50 of 56 nM, R788 dose-dependently prevents anti-IgE-mediated CHMC degranulation. R788 also prevents the release of LTC4 and cytokines and chemokines, such as TNFα, IL-8, and GM-CSF, that are triggered by anti-IgE. All phosphorylation events downstream of Syk signaling are inhibited when R788 inhibits Syk. The most potent inhibitor of IL-4 and IL-2 receptor signaling in CHMC is R788, followed by FcρRI signaling. R788 specifically inhibits human mast cells, neutrophils, and macrophages' FcγR signaling. R788 has the ability to prevent immune complex-mediated local inflammatory injury.R788 causes most of the tested DLBCL cell lines to undergo apoptosis[1]. R788 selectively inhibits both tonic- and ligand-induced BCR signaling (autophosphorylation of SYK525/526 and SYK-dependent phosphorylation of the B-cell linker protein [BLNK]) in R788-sensitive DLBCL cell lines[2]. R406 dose-dependently inhibited anti-IgE-mediated degranulation (tryptase release) in cultured human mast cells (CHMC) with an EC₅₀ of 0.056 ± 0.02 µM, but showed no activity on ionomycin-triggered degranulation, indicating specificity for FcεRI signaling. [1] R406 inhibited anti-IgE-induced production and release of LTC₄ (EC₅₀ = 0.093 µM), TNFα (EC₅₀ = 0.118 µM), IL-8 (EC₅₀ = 0.140 µM), and GM-CSF (EC₅₀ = 0.158 µM) in CHMC. [1] R406 also inhibited anti-IgG-induced degranulation (EC₅₀ = 0.064 µM) and mediator release (LTC₄, TNFα, IL-8, GM-CSF) in CHMC, indicating inhibition of FcγR signaling. [1] R406 inhibited TNFα production induced by FcγR-cross-linking in human monocyte-derived macrophages (EC₅₀ = 0.111 µM) and the monocytic cell line THP-1 (EC₅₀ = 0.171 µM). [1] R406 inhibited oxidative burst in TNFα-primed human neutrophils stimulated with anti-IgG (EC₅₀ = 0.033 µM). [1] R406 inhibited CD69 up-regulation in primary human B cells stimulated with anti-IgM (EC₅₀ = 0.048 µM), indicating inhibition of B-cell receptor (BCR) signaling. [1] R406 did not inhibit phosphorylation of Syk tyrosine 352 or the FcRγ chain in CHMC, but inhibited phosphorylation of linker for activation of T cells (LAT) tyrosine 191 and downstream signaling molecules (PLCγ1, Akt, ERK, p38, JNK), confirming selective Syk inhibition. [1] In cellular selectivity assessments, R406 inhibited Flt3 autophosphorylation with approximately 5-fold less potency than Syk, and showed weaker inhibition of Jak and Lck-dependent pathways. [1] R406 (up to 50 µM) had negligible effects on phagocytosis of FITC-labeled opsonized bacteria by monocytes and granulocytes in whole blood. [1] R406 did not significantly inhibit oxidative burst activity of human leukocytes induced by phagocytosis of opsonized E. coli, nor did it affect granulocyte migration toward fMLP at 10 µM. [1] R406 (up to 20 µM) did not inhibit microbicidal activity of purified primary neutrophils against opsonized S. aureus. [1] |
| ln Vivo |
R788 administered orally to mice decreases immune complex-mediated inflammation in two antibody-induced arthritis models and a reverse-passive Arthus reaction.[1] Another study found that R788 significantly increases the survival of treated animals while also effectively inhibiting BCR signaling in vivo, which reduces the proliferation and survival of malignant B cells. [3] In rheumatoid arthritis models, R788 shows a marked decrease in key inflammatory mediators like TNFalpha, IL-1, IL-6, and IL-18, which results in less inflammation and bone degradation.[4] Oral administration of R406 (1 and 5 mg/kg, 1 hour before challenge) reduced cutaneous reverse passive Arthus reaction in mice by approximately 72% and 86%, respectively, as measured by Evans blue dye extravasation. [1] In the collagen antibody-induced arthritis (CAIA) mouse model, oral administration of R406 (twice daily starting 4 hours after antibody challenge) reduced paw inflammation and swelling, slowed disease progression, and improved joint histopathology (reduced synovitis, pannus formation, leukocyte infiltration). [1] In the K/BxN serum transfer mouse model, oral administration of R406 (10 mg/kg twice daily) delayed onset and reduced severity of clinical arthritis, with paw thickening and clinical scores reduced by approximately 50%. [1] R406 dosed orally up to 100 mg/kg (resulting in systemic exposures up to 25 µM) did not extend bleeding time in mice after tail-tip amputation. [1] |
| Enzyme Assay |
There are fluorescence polarization reactions. In order to determine Ki, eight distinct ATP concentrations are set up in duplicate 200-μL reactions, starting at 200 μM (2-fold serial dilutions), with or without DMSO or R788 at 125, 62.5, 31.25, 15.5, or 7.8 nM. Twenty microliters of each reaction are taken out and quenched at various times to bring the reaction to an end. Plotting the rate of reaction against the ATP concentration yields the apparent Km and Vmax for each concentration of R788. To calculate the Ki, the apparent Km (or apparent Ki/Vmax) is finally plotted against the inhibitor concentration. Fluorescence Polarization Kinase Assay: Duplicate 200-µl reactions were set up at eight different ATP concentrations (serial dilutions from 200 µM) in the presence of either DMSO or R406 at concentrations of 125, 62.5, 31.25, 15.5, or 7.8 nM. At different time points, 20 µl of each reaction was removed and quenched. The reaction rate at each ATP concentration was determined and plotted against ATP concentration to determine apparent Km and Vmax. The apparent Km was then plotted against inhibitor concentration to determine the Ki. Data analysis was performed using enzyme kinetics software. [1] Crystallography: The kinase domain of Syk (Ile365 to Asn635 with mutation R440Q) was crystallized. Diffraction data were collected to a 2.3-Å resolution. The structure was solved by molecular replacement using a previously determined Syk kinase structure as the search model. Soaking crystals with R406 revealed electron density consistent with R406 binding competitively in the ATP-binding pocket. [1] |
| Cell Assay |
Complementary data shows the growth, priming, and stimulation of cultured human mast cells (CHMC), which are derived from cord blood CD34+ progenitor cells. Cells are cultivated in DMSO or R788 for half an hour prior to stimulation. The next step involves stimulating the cells using either 2 μM ionomycin or 0.25 to 2 mg/mL anti-IgE or anti-IgG. A modified Tyrode's buffer is used to stimulate approximately 1500 cells per well for 30 minutes in order to measure tryptase. Ten thousand cells per well are stimulated for one hour for LTC4 production and seven hours for cytokine production. LTC4 and cytokines are detected using Luminex multiplex technology, while tryptase activity is determined by luminescence readout of a peptide substrate. Human Mast Cell Degranulation and Mediator Release: Cultured human mast cells (CHMC) derived from cord blood CD34⁺ progenitors were primed and pre-incubated with R406 or DMSO for 30 minutes. Cells were stimulated with anti-IgE or anti-IgG. Tryptase release was measured after 30 minutes using a luminescent peptide substrate assay. For LTC₄ and cytokine (TNFα, IL-8, GM-CSF) production, cells were stimulated for 1 or 7 hours, and mediators were measured using multiplex technology. [1] Western Blot for Signaling Analysis: CHMC or other cell types were pre-incubated with R406 or control inhibitors (e.g., PP2) for 40 minutes and stimulated with anti-IgE for 5 minutes. Cells were lysed in SDS sample buffer. Proteins were separated by SDS-PAGE, transferred to membranes, and probed with phospho-specific antibodies against Syk (Tyr352), LAT (Tyr191), PLCγ1 (Tyr783), Akt (Ser473), ERK (Thr202/Tyr204), p38 (Thr180/Tyr182), and JNK (Thr183/Tyr185). Membranes were reprobed for total protein to verify equal loading. For immunoprecipitation, cells were stimulated and lysed, followed by standard immunoprecipitation protocols using specific antibodies. [1] Macrophage TNFα Production: Human primary macrophages derived from CD14⁺ monocytes (differentiated with GM-CSF for 5 days) or THP-1 cells (primed with IFN-γ) were added to IgG-coated wells in the presence of R406. After 16-20 hours incubation, TNFα in supernatants was measured by multiplex assay. LPS stimulation in uncoated wells served as a Syk-independent control. [1] Neutrophil Oxidative Burst: Primary human neutrophils were isolated, pre-incubated with R406 in buffer, primed with TNFα, and stimulated with anti-IgG or PMA. Respiratory burst was measured by adding dihydrorhodamine 123 and analyzing intracellular fluorescence by flow cytometry. [1] B Cell Activation: Primary human B cells isolated from peripheral blood were pre-incubated with R406 in medium for 60 minutes, stimulated with anti-IgM for 6 hours, stained with anti-CD69-APC, and analyzed by flow cytometry. [1] |
| Animal Protocol |
Balb/c mice with arthritis 1 mg/kg or 5 mg/kg o.g. Reverse Passive Arthus Reaction: Female C57BL/6 mice were challenged intravenously with 1% ovalbumin (OVA) containing 1% Evans blue dye (10 mg/kg). Ten minutes later, rabbit anti-OVA IgG (50 µg in 25 µl) was injected intradermally at three sites on one side of the back; control rabbit IgG was injected on the opposite side. R406 or vehicle (67% PEG 400) was administered orally 60 minutes before challenge. Four hours later, mice were euthanized, and 8-mm punch biopsies of injection sites were incubated in formamide at 80°C overnight. Extravasated dye concentration was measured spectrophotometrically at OD610. [1] Collagen Antibody-Induced Arthritis (CAIA): Female Balb/c mice were passively sensitized by intravenous administration of a collagen-induced arthritis monoclonal antibody blend on day 0, followed by intraperitoneal LPS (25 µg) on day 2. R406 or vehicle (35% TPGS, 60% PEG 400, 5% propylene glycol) was administered orally twice daily for 14 days, starting 4 hours after antibody challenge on day 0. Clinical scores were assessed regularly. [1] K/BxN Serum Transfer Model: Arthritis was induced in C57BL/6 mice by intraperitoneal injection of 150 µl of pooled sera from K/BxN mice on days 0 and 2. R406 or vehicle (35% TPGS, 60% PEG 400, 5% propylene glycol) was administered orally 1 hour before serum injection and then twice daily for 13 days. Ankle thickness and clinical arthritis scores were assessed daily. [1] Bleeding Time Assay: Mice were dosed orally with R406 (up to 100 mg/kg) or aspirin (100 mg/kg). Bleeding time was measured after tail-tip amputation. [1] |
| ADME/Pharmacokinetics |
In a clinical study in healthy human volunteers, R406 was highly orally bioavailable. Plasma concentration increased dose-proportionally up to 400 mg, then reached a plateau. [1] The maximal plasma concentration was generally reached between 1.2 and 1.3 hours after dosing. [1] The terminal half-life was approximately 15 hours. [1] R406 exhibited high plasma protein binding (>98%) in humans. [1] The plasma concentration producing 50% reduction of basophil activation (CD63 expression) ex vivo was 496 ± 42 ng/ml, corresponding to an EC₅₀ of 1.06 µM. [1] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of fostamatinib during breastfeeding. Because the active metabolite of fostamatinib (R406) is 98.3% bound to plasma proteins, the amount in milk is likely to be low. However, the active metabolites has a half-life of 15 hours, and might accumulate in the infant. The manufacturer recommends that breastfeeding be discontinued during fostamatinib therapy and for at least 1 month after the final dose. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. In cellular selectivity profiling, R406 showed some inhibition of adenosine A₃ receptor (IC₅₀ ≈ 0.081 µM), adenosine transporter (IC₅₀ ≈ 1.84 µM), and monoamine transporter (IC₅₀ ≈ 2.74 µM) in biochemical binding assays. [1] In human volunteers, even at the highest doses (up to 600 mg), R406 did not inhibit collagen- or ADP-induced platelet aggregation in platelet-rich plasma. [1] Safety assessment in human volunteers indicated no effects on hematologic or chemistry safety parameters at doses that inhibited basophil activation. [1] The most frequent adverse event in the 600 mg dose group was postural dizziness (5 out of 6 subjects). [1] R406 treatment caused a dose-dependent reduction of circulating CD45+CD14+ mononuclear cells 4 hours after a single dose in humans, which was reversed by 20 hours. [1] |
| References |
[1]. J Pharmacol Exp Ther . 2006 Dec;319(3):998-1008. [2]. Blood . 2008 Feb 15;111(4):2230-7. [3]. Blood . 2010 Dec 2;116(23):4894-905. [4].IDrugs . 2009 Mar;12(3):174-85. [5]. J Pharmacol Exp Ther . 2006 Dec;319(3):998-1008. |
| Additional Infomation |
Fostamatinib Disodium is an orally available disodium salt of the Syk kinase inhibitor fostamatinib with potential anti-inflammatory and immunomodulating activities. Fostamatinib inhibits Syk kinase-mediated IgG Fc gamma receptor signaling, resulting in inhibition of the activation of mast cells, macrophages, and B-cells and related inflammatory responses and tissue damage. Syk kinase, widely expressed in hematopoietic cells, is a nonreceptor tyrosine kinase that is involved in coupling activated immunoreceptors to signal downstream events that mediate diverse cellular responses, including proliferation, differentiation, and phagocytosis. See also: Fostamatinib Disodium (annotation moved to). Drug Indication Tavlesse is indicated for the treatment of chronic immune thrombocytopenia (ITP) in adult patients who are refractory to other treatments. R406 is a small molecule with the chemical name N4-(2,2-dimethyl-3-oxo-4H-pyrid[1,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine. [1] It was identified through cell-based structure-activity relationship screening using human mast cells to find inhibitors of FcεRI signaling. [1] R406 binds the ATP-binding pocket of Syk in a U-shaped conformation, with key interactions at the hinge region via the pyrimidine N1 and linker N2, and additional hydrogen bonds and hydrophobic interactions with the methoxylated phenyl and heterocyclic rings. [1] R406 inhibits Syk-dependent signaling in immune cells (mast cells, macrophages, neutrophils, B cells) without markedly interfering with Syk-independent innate immune responses (phagocytosis, microbicidal activity) or hemostasis in mice. [1] The drug has shown therapeutic potential in animal models of immune complex-mediated inflammatory diseases such as rheumatoid arthritis. [1] In humans, oral R406 achieves exposures capable of inhibiting Syk-dependent IgE-mediated basophil activation, suggesting potential for modulating Syk activity in human allergic and autoimmune disorders. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: 8.33 mg/mL (11.37 mM) in 0.5% CMC-Na/saline water (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 10 mg/mL (13.65 mM) in Cremophor EL (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Solubility in Formulation 3: 0.5% CMC+0.25% Tween 80,pH6.5: 30 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3652 mL | 6.8258 mL | 13.6517 mL | |
| 5 mM | 0.2730 mL | 1.3652 mL | 2.7303 mL | |
| 10 mM | 0.1365 mL | 0.6826 mL | 1.3652 mL |