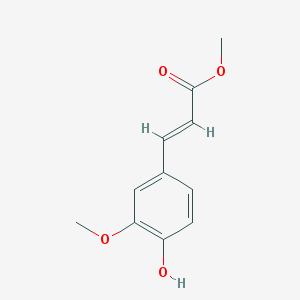
Ferulic acid methyl ester (Methyl ferulate) is a naturally occurring ferulic acid analog isolated from Stemona tuberosa with anti-inflammatory and antioxidant activity.
Physicochemical Properties
| Molecular Formula | C₁₁H₁₂O₄ |
| Molecular Weight | 208.21 |
| Exact Mass | 208.073 |
| CAS # | 2309-07-1 |
| Related CAS # | (E)-Ferulic acid methyl ester;22329-76-6 |
| PubChem CID | 5357283 |
| Appearance | Off-white to pink solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 338.1±27.0 °C at 760 mmHg |
| Melting Point | 62-65°C |
| Flash Point | 130.4±17.2 °C |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.575 |
| LogP | 1.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 15 |
| Complexity | 237 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | COC1=C(C=CC(=C1)/C=C/C(=O)OC)O |
| InChi Key | AUJXJFHANFIVKH-GQCTYLIASA-N |
| InChi Code | InChI=1S/C11H12O4/c1-14-10-7-8(3-5-9(10)12)4-6-11(13)15-2/h3-7,12H,1-2H3/b6-4+ |
| Chemical Name | (E)-3-(4-hydroxy-3-methoxyphenyl)acrylic acid |
| Synonyms | Methyl ferulateConiferic acid trans-Ferulic acid NSC-2821 NSC2821NSC 2821Ferulic Acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Following treatment for six, eighteen, and forty-eight hours, BMDMs do not exhibit any cytotoxic effects from methyl ferulate (25 μg/mL) [2]. Methyl ferulate (Methyl ferulate; 5, 10, 25 µg/mL) suppresses primary bone marrow-derived macrophages at 10 and 25 µg/mL and inhibits TNFα, IL6, IFNγ, but not IL10. NO generation in BMDM cells [2]. At 25 µg/mL, methyl ferulate suppresses the expression of COX-2 and blocks p-p38 and p-JNK[2]. - Anti-inflammatory activity: Ferulic acid methyl ester inhibited LPS-induced production of pro-inflammatory cytokines TNF-α and IL-1β in RAW 264.7 macrophages in a dose-dependent manner. At concentrations of 25 μM, 50 μM, and 100 μM, it reduced TNF-α levels by 32%, 58%, and 75% respectively, and IL-1β levels by 28%, 53%, and 71% respectively, compared to LPS-treated control cells [2] - Antioxidant activity: Ferulic acid methyl ester scavenged DPPH radicals with an IC50 of 12.5 μM, and inhibited LPS-induced reactive oxygen species (ROS) generation in RAW 264.7 cells (inhibition rate = 62% at 100 μM) [2] |
| Enzyme Assay |
- DPPH radical scavenging assay: Ferulic acid methyl ester (5-50 μM) was mixed with DPPH solution (0.1 mM) in ethanol, incubated at room temperature for 30 minutes. Absorbance was measured at 517 nm to calculate the scavenging rate, with ascorbic acid as the positive control [2] - ROS inhibition assay: A ROS-sensitive fluorescent probe was mixed with Ferulic acid methyl ester (25-100 μM) and RAW 264.7 cell suspension. After pre-incubation for 30 minutes, LPS (1 μg/mL) was added to induce ROS generation. Fluorescence intensity was measured at excitation 488 nm/emission 525 nm after 24 hours of incubation [2] |
| Cell Assay |
Cell viability assay [2] Cell Types: primary bone marrow-derived macrophages (BMDM) Tested Concentrations: 25 µg/mL Incubation Duration: 6 hrs (hours), 18 hrs (hours), 48 hrs (hours) Experimental Results: No cytotoxic effect on BMDM. Western Blot Analysis[2] Cell Types: RAW 246.7 Cell Tested Concentrations: 10 µg/mL and 25 µg/mL Incubation Duration: 1 hour before LPS stimulation Experimental Results: Significant inhibition of COX-2 expression at 25 µg/mL concentration. - Anti-inflammatory cell assay: RAW 264.7 macrophages were seeded into 24-well plates at a density of 5×10⁴ cells/well and incubated overnight. Cells were pre-treated with Ferulic acid methyl ester (25-100 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. The cell supernatant was collected, and concentrations of TNF-α and IL-1β were detected by enzyme-linked immunosorbent assay (ELISA) [2] |
| References |
[1]. Sultana R. Ferulic acid ethyl ester as a potential therapy in neurodegenerative disorders. Biochim Biophys Acta. 2012 May;1822(5):748-52. [2]. Anti-inflammatory activity of methyl ferulate isolated from Stemona tuberosa Lour. Asian Pac J Trop Med. 2014 Sep;7S1:S327-31. |
| Additional Infomation |
Trans-methylferulate is a cinnamate ester that is the methyl ester of ferulic acid. It has been isolated from Pisonia aculeata. It has a role as a plant metabolite. It is a cinnamate ester, a methyl ester and a member of guaiacols. It is functionally related to a ferulic acid. Methyl ferulate has been reported in Meum athamanticum, Iris milesii, and other organisms with data available. See also: Black Cohosh (part of). - Natural source: Ferulic acid methyl ester (methyl ferulate) is a natural phenolic ester isolated from the tubers of Stemona tuberosa Lour. [2] - Chemical classification: It belongs to the ferulic acid ester family, with a structure consisting of ferulic acid linked to a methyl group via an ester bond [2] - Mechanism of action: Ferulic acid methyl ester exerts anti-inflammatory effects by suppressing LPS-induced production of pro-inflammatory cytokines (TNF-α, IL-1β) and reducing oxidative stress through ROS scavenging [2] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 100 mg/mL (~480.28 mM) H2O : ~2 mg/mL (~9.61 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (12.01 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (12.01 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (12.01 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.8028 mL | 24.0142 mL | 48.0284 mL | |
| 5 mM | 0.9606 mL | 4.8028 mL | 9.6057 mL | |
| 10 mM | 0.4803 mL | 2.4014 mL | 4.8028 mL |