Physicochemical Properties
| Molecular Formula | C24H30N6O6S2 |
| Molecular Weight | 562.66 |
| Exact Mass | 562.166 |
| CAS # | 2671128-05-3 |
| PubChem CID | 156818030 |
| Appearance | Off-white to light yellow solid powder |
| LogP | 1.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 38 |
| Complexity | 926 |
| Defined Atom Stereocenter Count | 3 |
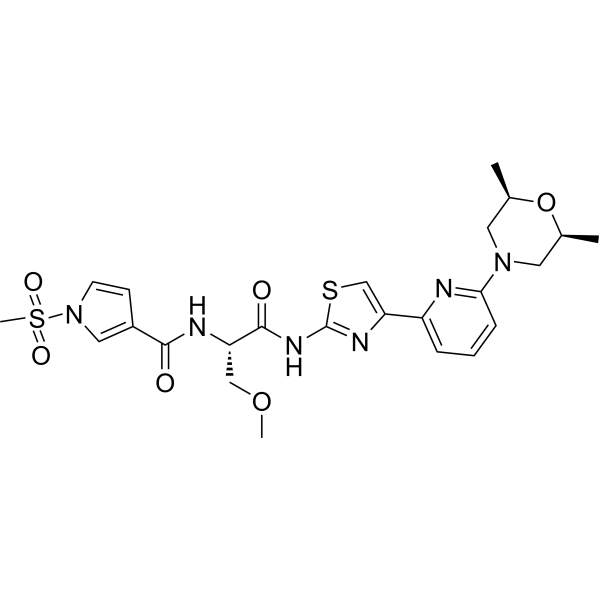
| SMILES | [C@H]1(C)O[C@@H](C)CN(C2=NC(C3N=C(NC(=O)[C@@H](NC(=O)C4=CN(S(=O)(C)=O)C=C4)COC)SC=3)=CC=C2)C1 |
| InChi Key | JBLQNFBXKOAIHG-FCEWJHQRSA-N |
| InChi Code | InChI=1S/C24H30N6O6S2/c1-15-10-29(11-16(2)36-15)21-7-5-6-18(25-21)20-14-37-24(27-20)28-23(32)19(13-35-3)26-22(31)17-8-9-30(12-17)38(4,33)34/h5-9,12,14-16,19H,10-11,13H2,1-4H3,(H,26,31)(H,27,28,32)/t15-,16+,19-/m0/s1 |
| Chemical Name | N-[(2S)-1-[[4-[6-[(2R,6S)-2,6-dimethylmorpholin-4-yl]pyridin-2-yl]-1,3-thiazol-2-yl]amino]-3-methoxy-1-oxopropan-2-yl]-1-methylsulfonylpyrrole-3-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | FHD-286 (10 to 100 nM) causes evaporation of cells for 7 days, ending MLL rearrangement (r), mutation (mt) NPM1 chromosome and 3q26 damage (EVI1 overexpression). FHD-286 also resulted in transcription of ATAC-Seq peaks and genome-wide consensus, as well as site-specific mRNA expression determined by RNA-Seq. These findings were linked to significant reductions in the target genome of MYC, mTORC1, E2F, interferon-γ, IL6 -JAK-STAT3, as well as inflammatory response and oxidative phosphorylation genes. The results of QPCR analysis showed a substantial decrease in the mRNA expression of the SPI1, BCL2, and MYC genes. A log2-fold reduction is observed in [1]. Mass spectrometry investigation of the lumen of FHD-286-treated AML cells revealed c-Myc, SPI1, MEF2C, KMT2C, and CDK4 (in MOLM13) and EVI1, c-Myb, CDK6, and c-Myc (in AML191) cells. |
| Animal Protocol |
Animal/Disease Models: B16F10 tumor-bearing mice [2] Doses: 1.5 mg/kg Route of Administration: oral; FHD-286 (1.5 mg/kg; diaphragm) causes increased levels of FNγ and th1-type chemokine CXCL10 [2]. Results lasting 10 days: increased levels of IFNγ and Th1 chemokine CXCL10. |
| References |
[1]. Abstract 1140: Pre-clinical efficacy of targeting BAF complexes through inhibition of the dual ATPases BRG1 and BRM by FHD-286 in cellular models of AML. Cancer Res (2023) 83 (7_Supplement): 1140. [2]. Synergistic efficacy of the BRM/BRG1 ATPase inhibitor, FHD-286, and anti-PD-1 antibody in mouse syngeneic tumor models. |
| Additional Infomation | Camibirstat is an orally bioavailable, allosteric, small molecule inhibitor of transcription activator BRG1 (SWI/SNF-related matrix-associated actin-dependent regulator of chromatin subfamily A member 4; SMARCA4) and BRM (SWI/SNF-related matrix-associated actin-dependent regulator of chromatin subfamily A member 2; SMARCA2), with potential antineoplastic activity. Upon oral administration, camibirstat targets, binds to, and inhibits the activity of BRG1 and/or BRM, the primary ATPase components and mutually exclusive subunits of the BRG1/BRM-associated factor (BAF) complexes. This may lead to the inhibition of the SWI/SNF chromatin remodeling complex, disrupt chromatin remodeling and gene expression, and result in the downregulation of oncogenic pathways and the inhibition of tumor cell proliferation. BAF is an important regulator of transcriptional programs and gene expression. Mutations in BAF or its transcription factor partners are found in certain diseases including cancers. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~444.32 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.70 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.70 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7773 mL | 8.8864 mL | 17.7727 mL | |
| 5 mM | 0.3555 mL | 1.7773 mL | 3.5545 mL | |
| 10 mM | 0.1777 mL | 0.8886 mL | 1.7773 mL |