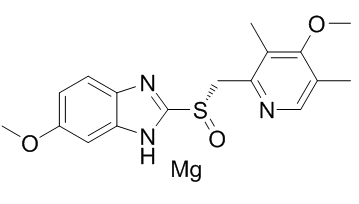
Esomeprazole magnesium, the
magnesium salt of esomeprazole, is a
potent proton pump inhibitor (PPI) with potential usefulness in the treatment
of gastroesophageal reflux disease-GERD. It inhibits H+/K+-ATPase with an IC50 of 0.076 mg/kg.
Physicochemical Properties
| Molecular Formula | C17H19N3O3S-.MG+2 |
| Molecular Weight | 369.721 |
| Exact Mass | 712.198 |
| CAS # | 1198768-91-0 |
| Related CAS # | Esomeprazole;119141-88-7;Esomeprazole magnesium trihydrate;217087-09-7;Esomeprazole sodium;161796-78-7;Esomeprazole magnesium;161973-10-0;Esomeprazole potassium salt;161796-84-5;Esomeprazole hemistrontium;914613-86-8 |
| PubChem CID | 9568613 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 49 |
| Complexity | 453 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | CC1=CN=C(C(=C1OC)C)C[S@](=O)C2=NC3=C([N-]2)C=CC(=C3)OC.CC1=CN=C(C(=C1OC)C)C[S@](=O)C2=NC3=C([N-]2)C=CC(=C3)OC.[Mg+2] |
| InChi Key | KWORUUGOSLYAGD-YPPDDXJESA-N |
| InChi Code | InChI=1S/2C17H18N3O3S.Mg/c2*1-10-8-18-15(11(2)16(10)23-4)9-24(21)17-19-13-6-5-12(22-3)7-14(13)20-17;/h2*5-8H,9H2,1-4H3;/q2*-1;+2/t2*24-;/m00./s1 |
| Chemical Name | magnesium;5-methoxy-2-[(S)-(4-methoxy-3,5-dimethylpyridin-2-yl)methylsulfinyl]benzimidazol-1-ide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Gastric H+/K+ ATPase proton pump. The study demonstrates that esomeprazole‘s effects on triple-negative breast cancer cells are mediated through this target, evidenced by increased expression of the pump in sensitive cells. [1] The study suggests that esomeprazole, beyond its classical target gastric H+/K+ ATPase, can inhibit dimethylarginine dimethylaminohydrolase (DDAH) and downregulate inducible nitric oxide synthase (iNOS) expression and activity, contributing to its anti-inflammatory and anti-fibrotic effects. [2] |
| ln Vitro |
Esomeprazole (25-100 µM; 20 hours; MDA-MB-468 cells) treatment enhances intracellular acidification, which in turn inhibits the growth of triple-negative breast cancer cells in vitro in a dose-dependent manner [1]. Esomeprazole suppressed the growth of triple-negative breast cancer cell line MDA-MB-468 in a dose-dependent manner with an EC50 of approximately 70 µM, as determined by trypan blue exclusion assay after 20 hours of treatment. [1] Esomeprazole (at 50 µM) significantly increased intracellular acidity in MDA-MB-468 cells, reducing the relative fluorescence of the pH-sensitive probe BCECF-AM to 60.2% of control levels. This acidification is linked to its growth-suppressive mechanism. [1] Esomeprazole enhanced the cytotoxic effects of doxorubicin (DOX) on MDA-MB-468 cells. Combination treatment with fixed concentrations of esomeprazole (30 µM) and doxorubicin (30 nM) resulted in a significantly greater reduction in live cell number compared to doxorubicin treatment alone. [1] In contrast to cancer cells, the non-cancerous breast epithelial cell line MCF-10A was significantly less sensitive to esomeprazole. Treatment with 100 µM esomeprazole reduced the number of live MDA-MB-468 cells to 51.88% of control, while the reduction in MCF-10A cells (to 92.31% of control) was not statistically significant. [1] |
| ln Vivo |
The C57BL/6J mice treated with esomeprazole (30–300 mg/kg; oral gavage; daily; for 19 or 11 days) showed a significant reduction in the animals' lung fibrosis progression. Additionally, esomeprazole lowers circulating fibrosis and inflammatory markers [2]. In a mouse model of cotton smoke-induced lung injury over 3 weeks, therapeutic administration of a low dose of esomeprazole (30 mg/kg/day, p.o., starting 10 days post-smoke exposure) significantly increased plasma levels of asymmetric dimethylarginine (ADMA) and reduced plasma nitric oxide (NO) levels compared to vehicle-treated controls, indicating modulation of the DDAH/iNOS pathway. [2] The same therapeutic dose (30 mg/kg/day) significantly reduced circulating levels of pro-inflammatory and pro-fibrotic markers, including tumor necrosis factor-alpha (TNFα) and matrix metalloproteinase-7 (MMP7), compared to vehicle-treated controls exposed to smoke. A decreasing trend was also observed for interleukin-1β (IL1β). [2] Therapeutic administration of low-dose esomeprazole (30 mg/kg/day) significantly inhibited the progression of lung fibrosis. The mean fibrosis score in the treatment group (0.64) was significantly lower than that in the vehicle control group (1.06). Histological analysis showed minimal to no fibrosis in the treatment group, whereas the control group exhibited fibrous thickening of alveolar walls. [2] Treatment with esomeprazole did not show a significant favorable effect on lung inflammation (assessed by H&E staining for inflammatory cell infiltration) in this specific smoke-induced injury model. [2] A high prophylactic dose of esomeprazole (300 mg/kg/day, p.o., starting 2 days post-smoke exposure) was poorly tolerated, leading to increased mortality, bloating, and sluggishness in mice, and showed no meaningful impact on suppressing lung fibrosis. [2] |
| Cell Assay |
Cell Viability Assay[1] Cell Types: MDA-MB-468 Cell Tested Concentrations: 25 µM, 50 µM, 75 µM, 100 µM Incubation Duration: 20 hrs (hours) Experimental Results: Inhibition of triple negative breast cancer cells in a dose-dependent manner in vitro. Cell Viability/Growth Suppression Assay: Triple-negative breast cancer MDA-MB-468 cells and non-cancerous breast epithelial MCF-10A cells were cultured in their respective growth media. Cells were treated with varying concentrations of esomeprazole, doxorubicin, or their combination in 6-well plates for 20 hours in RPMI medium (chosen for its low buffering capacity) containing fetal bovine serum. After treatment, cells were washed, detached with trypsin, and stained with trypan blue. The number of live (unstained) cells was counted using an automated cell counter. Data were expressed relative to untreated control cells. [1] Intracellular pH Measurement: MDA-MB-468 cells were treated with esomeprazole (50 µM) or acidified medium (pH 6.0) for 20 hours. After treatment, cells were washed and incubated with 3 µM BCECF-AM, a pH-sensitive fluorescent probe, for 30 minutes at 37°C. Following another wash, intracellular fluorescence was detected and imaged using confocal microscopy. The fluorescence intensity at 530 nm, which is inversely correlated with intracellular acidity (lower pH = lower fluorescence), was quantified via digital image analysis and expressed as relative fluorescence per cell. [1] Immunofluorescence for Target Detection: MDA-MB-468 and MCF-10A cells were grown on glass coverslips. Cells were fixed with paraformaldehyde, permeabilized, and incubated with a primary mouse monoclonal antibody against the human H+/K+ ATPase β subunit. After washing, cells were incubated with a fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse IgG secondary antibody. Fluorescence was visualized and detected using confocal microscopy to assess the expression level of the gastric proton pump. [1] Western Blotting for Target Confirmation: Cell lysates from MDA-MB-468 and MCF-10A cells were prepared. Proteins were separated by electrophoresis, transferred to a membrane, and probed with the same primary mouse monoclonal antibody against human H+/K+ ATPase β subunit used in immunofluorescence. A horseradish peroxidase-conjugated goat anti-mouse IgG was used as the secondary antibody for detection. To confirm equal protein loading, the membrane was also probed with a mouse monoclonal antibody against human α-tubulin. [1] |
| Animal Protocol |
Animal/Disease Models: C57BL/6J mice (8 weeks old, 25-30 g) cotton smoke-induced lung injury [2] Doses: 30 mg/kg, 300 mg/kg Route of Administration: po (oral gavage); daily; continued for 19 Or 11-day Experimental Results: Dramatically inhibited the progression of lung fibrosis in animals. Cotton Smoke-Induced Lung Injury Model in Mice: Eight-week-old C57BL/6J mice were exposed to cotton smoke for 21 days to induce lung injury. Animals were randomized into groups: a no-exposure (sham) group, a smoke-exposed vehicle control group, a prophylactic esomeprazole group, and a therapeutic esomeprazole group. The vehicle and prophylactic groups received daily oral gavage of either 10% ethanol (vehicle) or esomeprazole (300 mg/kg dissolved in 10% ethanol) starting 2 days after the initiation of smoke exposure. The therapeutic group received daily oral esomeprazole at a lower dose of 30 mg/kg (in 10% ethanol) starting 10 days after smoke exposure began and continuing until the end of the study (necropsy). At necropsy, blood was collected via cardiac puncture for plasma analysis, and lungs and other organs were harvested for weight measurement and histopathological examination (H&E and Masson's Trichrome staining). [2] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion After oral administration, peak plasma levels (Cmax) occur at approximately 1.5 hours (Tmax). The Cmax increases proportionally when the dose is increased, and there is a three-fold increase in the area under the plasma concentration-time curve (AUC) from 20 to 40 mg. At repeated once-daily dosing with 40 mg, the systemic bioavailability is approximately 90% compared to 64% after a single dose of 40 mg. The mean exposure (AUC) to esomeprazole increases from 4.32 μmolhr/L on Day 1 to 11.2 μmolhr/L on Day 5 after 40 mg once daily dosing. The AUC after administration of a single 40 mg dose of Esomeprazole is decreased by 43% to 53% after food intake compared to fasting conditions. Esomeprazole should be taken at least one hour before meals. _Combination Therapy with Antimicrobials:_ Esomeprazole magnesium 40 mg once daily was given in combination with [DB01211] 500 mg twice daily and [DB01060] 1000 mg twice daily for 7 days to 17 healthy male and female subjects. The mean steady state AUC and Cmax of esomeprazole increased by 70% and 18%, respectively during triple combination therapy compared to treatment with esomeprazole alone. The observed increase in esomeprazole exposure during co-administration with clarithromycin and amoxicillin is not expected to produce significant safety concerns. The plasma elimination half-life of esomeprazole is approximately 1 to 1.5 hours. Less than 1% of parent drug is excreted in the urine. Approximately 80% of an oral dose of esomeprazole is excreted as inactive metabolites in the urine, and the remainder is found as inactive metabolites in the feces. The apparent volume of distribution at steady state in healthy volunteers is approximately 16 L. The plasma elimination half-life of esomeprazole is approximately 1 to 1.5 hours. Less than 1% of parent drug is excreted in the urine. Approximately 80% of an oral dose of esomeprazole is excreted as inactive metabolites in the urine, and the remainder is found as inactive metabolites in the feces. Esomeprazole is 97% bound to plasma proteins. Plasma protein binding is constant over the concentration range of 2 to 20 umol/L. The apparent volume of distribution at steady state in healthy volunteers is approximately 16 L. NEXIUM Delayed-Release Capsules and NEXIUM For Delayed-Release Oral Suspension contain a bioequivalent enteric-coated granule formulation of esomeprazole magnesium. Bioequivalency is based on a single dose (40 mg) study in 94 healthy male and female volunteers under fasting condition. After oral administration peak plasma levels (Cmax) occur at approximately 1.5 hours (Tmax). The Cmax increases proportionally when the dose is increased, and there is a three-fold increase in the area under the plasma concentration-time curve (AUC) from 20 to 40 mg. At repeated once-daily dosing with 40 mg, the systemic bioavailability is approximately 90% compared to 64% after a single dose of 40 mg. The mean exposure (AUC) to esomeprazole increases from 4.32 umolhr/L on Day 1 to 11.2 umolhr/L on Day 5 after 40 mg once daily dosing. Metabolism / Metabolites Esomeprazole is extensively metabolized in the liver by the cytochrome P450 (CYP) enzyme system. The metabolites of esomeprazole lack antisecretory activity. The major part of esomeprazole’s metabolism is dependent upon the CYP2C19 isoenzyme, which forms the hydroxy and desmethyl metabolites. The remaining amount is dependent on CYP3A4 which forms the sulphone metabolite. CYP2C19 isoenzyme exhibits polymorphism in the metabolism of esomeprazole, since some 3% of Caucasians and 15 to 20% of Asians lack CYP2C19 and are termed Poor Metabolizers. However, the influence of CYP 2C19 polymorphism is less pronounced for esomeprazole than for omeprazole. At steady state, the ratio of AUC in Poor Metabolizers to AUC in the rest of the population (Extensive Metabolizers) is approximately 2. Following administration of equimolar doses, the S- and R-isomers are metabolized differently by the liver, resulting in higher plasma levels of the S- than of the R-isomer. Nine major urinary metabolites have been detected. The two main metabolites have been identified as hydroxyesomeprazole and the corresponding carboxylic acid. Three major metabolites have been identified in plasma: the 5-O-desmethyl- and sulphone derivatives and hydroxyesomeprazole. The major metabolites of esomeprazole have no effect on gastric acid secretion. Esomeprazole is extensively metabolized in the liver by the cytochrome P450 (CYP) enzyme system. The metabolites of esomeprazole lack antisecretory activity. The major part of esomeprazole's metabolism is dependent upon the CYP 2C19 isoenzyme, which forms the hydroxy and desmethyl metabolites. The remaining amount is dependent on CYP 3A4 which forms the sulphone metabolite. CYP 2C19 isoenzyme exhibits polymorphism in the metabolism of esomeprazole, since some 3% of Caucasians and 15 to 20% of Asians lack CYP 2C19 and are termed Poor Metabolizers. At steady state, the ratio of AUC in Poor Metabolizers to AUC in the rest of the population (Extensive Metabolizers) is approximately 2. Following administration of equimolar doses, the S- and R-isomers are metabolized differently by the liver, resulting in higher plasma levels of the S- than of the R-isomer. Biological Half-Life 1-1.5 hours |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Esomeprazole is the S-enantiomer of the proton-pump inhibitor, omeprazole. Limited information indicates that maternal doses of 10 mg daily produce low levels in milk and would not be expected to cause any adverse effects in breastfed infants. ◉ Effects in Breastfed Infants One mother taking omeprazole 20 mg daily orally pumped and discarded her milk once each day 4 hours after her morning dose. She breastfed her infant the remainder of the day for 3 months before weaning. The infant remained well at 12 months of age. A woman with rheumatoid arthritis was treated with oral esomeprazole 10 mg, prednisone 2.5 mg and sulfasalazine 1 gram once daily as well as injections of certolizumab pegol 200 mg every 2 weeks. Her infant was about 50% breastfed and 50% formula fed. The infant had no detectable drug-related adverse effects. ◉ Effects on Lactation and Breastmilk Omeprazole (the racemic form) has been reported to cause gynecomastia in several men and a retrospective claims database study in the United States found that users of proton pump inhibitors had an increased risk of gynecomastia. A review article reported that a search of database from the European Pharmacovigilance Centre found 45 cases of gynecomastia, 9 cases of galactorrhea, 19 cases of breast pain and 12 cases of breast enlargement associated with esomeprazole. A search of the WHO global pharmacovigilance database found 114 cases of gynecomastia, 38 cases of galactorrhea, 56 cases of breast pain and 28 cases of breast enlargement associated with esomeprazole. One woman developed elevated serum prolactin and estradiol with bilateral galactorrhea one week after starting esomeprazole 40 mg once daily for reflux esophagitis. The galactorrhea disappeared 3 days after discontinuing esomeprazole and prolactin and estradiol returned to normal 7 days after discontinuation. One month later, the patient restarted esomeprazole and again developed bilateral galactorrhea. She was switched to lansoprazole with no galactorrhea developing. The prolactin level in a mother with established lactation may not affect her ability to breastfeed. Protein Binding Esomeprazole is 97% bound to plasma proteins. Plasma protein binding is constant over the concentration range of 2 to 20 µmol/L. The discussion mentions that proton pump inhibitors (PPIs) as a class are generally well-tolerated with mild and transient side effects like headache and diarrhea, and that high intravenous doses (up to 240 mg daily) have been used in Zollinger-Ellison syndrome with good tolerability. However, potential problems with long-term high-dose use are noted as unknown due to a lack of studies. [1] In this mouse study, a high dose of esomeprazole (300 mg/kg/day, p.o.) used in the prophylactic regimen was not well-tolerated. It caused adverse effects including bloating, sluggishness, and increased mortality. The Kaplan-Meier survival plot showed decreased survival in the high-dose group compared to controls. [2] A lower therapeutic dose of esomeprazole (30 mg/kg/day, p.o.) was well-tolerated without reported significant adverse effects in the mice. [2] Organ weight analysis at necropsy showed no significant differences in lung, heart, or kidney weights between the esomeprazole-treated groups and the vehicle control. A slight, but statistically insignificant, increase in liver weight was observed in the esomeprazole-treated groups. [2] |
| References |
[1]. Use of proton pump inhibitors as adjunct treatment for triple-negative breast cancers. An introductory study. J Pharm Pharm Sci. 2014;17(3):439-46. [2]. Therapeutic Efficacy of Esomeprazole in Cotton Smoke-Induced Lung Injury Model. Front Pharmacol. 2017 Jan 26;8:16. [3]. Esomeprazole: a clinical review. Am J Health Syst Pharm. 2002 Jul 15;59(14):1333-9. |
| Additional Infomation |
Esomeprazole magnesium is a magnesium salt resulting from the formal reaction of magnesium hydroxide with 2 mol eq. of esomeprazole. An inhibitor of gastric acid secretion, it is used for the treatment of gastro-oesophageal reflux disease, dyspepsia, peptic ulcer disease, and Zollinger-Ellison syndrome. It has a role as an EC 3.6.3.10 (H(+)/K(+)-exchanging ATPase) inhibitor and an anti-ulcer drug. It contains an esomeprazole(1-). Esomeprazole, sold under the brand name Nexium, is a proton pump inhibitor (PPI) medication used for the management of gastroesophageal reflux disease (GERD), for gastric protection to prevent recurrence of stomach ulcers or gastric damage from chronic use of NSAIDs, and for the treatment of pathological hypersecretory conditions including Zollinger-Ellison (ZE) Syndrome. It can also be found in quadruple regimens for the treatment of H. pylori infections along with other antibiotics including [DB01060], [DB01211], and [DB00916], for example. Its efficacy is considered similar to other medications within the PPI class including [DB00338], [DB00213], [DB00448], [DB05351], and [DB01129]. Esomeprazole is the s-isomer of [DB00338], which is a racemate of the S- and R-enantiomer. Esomeprazole has been shown to inhibit acid secretion to a similar extent as [DB00338], without any significant differences between the two compounds in vitro. Esomeprazole exerts its stomach acid-suppressing effects by preventing the final step in gastric acid production by covalently binding to sulfhydryl groups of cysteines found on the (H+, K+)-ATPase enzyme at the secretory surface of gastric parietal cells. This effect leads to inhibition of both basal and stimulated gastric acid secretion, irrespective of the stimulus. As the binding of esomeprazole to the (H+, K+)-ATPase enzyme is irreversible and new enzyme needs to be expressed in order to resume acid secretion, esomeprazole's duration of antisecretory effect persists longer than 24 hours. PPIs such as esomeprazole have also been shown to inhibit the activity of dimethylarginine dimethylaminohydrolase (DDAH), an enzyme necessary for cardiovascular health. DDAH inhibition causes a consequent accumulation of the nitric oxide synthase inhibitor asymmetric dimethylarginie (ADMA), which is thought to cause the association of PPIs with increased risk of cardiovascular events in patients with unstable coronary syndromes. Due to their good safety profile and as several PPIs are available over the counter without a prescription, their current use in North America is widespread. Long term use of PPIs such as esomeprazole has been associated with possible adverse effects, however, including increased susceptibility to bacterial infections (including gastrointestinal C. difficile), reduced absorption of micronutrients such as iron and B12, and an increased risk of developing hypomagnesemia and hypocalcemia which may contribute to osteoporosis and bone fractures later in life. Rapid discontinuation of PPIs such as esomeprazole may cause a rebound effect and a short term increase in hypersecretion. Esomeprazole doses should be slowly lowered, or tapered, before discontinuing to prevent this rebound effect. Esomeprazole Magnesium is the magnesium salt of esomeprazole, the S-isomer of omeprazole, with gastric proton pump inhibitor activity. In the acidic compartment of parietal cells, esomeprazole is protonated and converted into the active achiral sulphenamide; the active sulphenamide forms one or more covalent disulfide bonds with the proton pump hydrogen-potassium adenosine triphosphatase (H+/K+ ATPase), thereby inhibiting its activity and the parietal cell secretion of H+ ions into the gastric lumen, the final step in gastric acid production. H+/K+ ATPase is an integral membrane protein of the gastric parietal cell. Esomeprazole is the S-isomer of omeprazole, with gastric proton pump inhibitor activity. In the acidic compartment of parietal cells, esomeprazole is protonated and converted into the active achiral sulfenamide; the active sulfenamide forms one or more covalent disulfide bonds with the proton pump hydrogen-potassium adenosine triphosphatase (H+/K+ ATPase), thereby inhibiting its activity and the parietal cell secretion of H+ ions into the gastric lumen, the final step in gastric acid production. H+/K+ ATPase is an integral membrane protein of the gastric parietal cell. The S-isomer of omeprazole. See also: Esomeprazole (has active moiety); Esomeprazole Magnesium; Naproxen (component of). Drug Indication Esomeprazole is indicated for the treatment of acid-reflux disorders including healing and maintenance of erosive esophagitis, and symptomatic gastroesophageal reflux disease (GERD), peptic ulcer disease, H. pylori eradication, prevention of gastrointestinal bleeds with NSAID use, and for the long-term treatment of pathological hypersecretory conditions including Zollinger-Ellison Syndrome. FDA Label Nexium Control is indicated for the short-term treatment of reflux symptoms (e. g. heartburn and acid regurgitation) in adults. Mechanism of Action Esomeprazole exerts its stomach acid-suppressing effects by preventing the final step in gastric acid production by covalently binding to sulfhydryl groups of cysteines found on the (H+, K+)-ATPase enzyme at the secretory surface of gastric parietal cells. This effect leads to inhibition of both basal and stimulated gastric acid secretion, irrespective of the stimulus. As the binding of esomeprazole to the (H+, K+)-ATPase enzyme is irreversible and new enzyme needs to be expressed in order to resume acid secretion, esomeprazole's duration of antisecretory effect that persists longer than 24 hours. Esomeprazole is a proton pump inhibitor that suppresses gastric acid secretion by specific inhibition of the H+/K+-ATPase in the gastric parietal cell. The S- and R-isomers of omeprazole are protonated and converted in the acidic compartment of the parietal cell forming the active inhibitor, the achiral sulphenamide. By acting specifically on the proton pump, esomeprazole blocks the final step in acid production, thus reducing gastric acidity. This effect is dose-related up to a daily dose of 20 to 40 mg and leads to inhibition of gastric acid secretion. Esomeprazole is a proton pump inhibitor (PPI), a substituted benzimidazole drug developed to suppress gastric acid production by irreversibly inhibiting the H+/K+ ATPase in gastric parietal cells. It is clinically used for gastroesophageal reflux disease, peptic ulcers, and functional dyspepsia. [1] This study explores its potential repurposing as an adjunct in cancer therapy. The proposed mechanism is that esomeprazole inhibits proton pumps (specifically the gastric H+/K+ ATPase found to be expressed in breast cancer cells), leading to intracellular acidification, which in turn suppresses cancer cell growth and increases their sensitivity to chemotherapy drugs like doxorubicin. [1] The study provides the first evidence that the gastric H+/K+ ATPase is expressed in triple-negative breast cancer cells (MDA-MB-468), which mediates their sensitivity to esomeprazole. This finding is novel, as previous belief held that this pump's expression was restricted to gastric parietal cells. [1] Esomeprazole is a proton pump inhibitor (PPI) clinically used to treat gastric acid-related disorders like GERD. This study explores its potential repurposing for treating lung injury and fibrosis. [2] The proposed mechanism involves the inhibition of DDAH, leading to increased levels of its substrate ADMA, which is an endogenous inhibitor of iNOS. This results in reduced iNOS activity and NO production, attenuating nitrosative stress, inflammation, and fibrosis. PPIs are also noted to upregulate heme oxygenase-1 (HO-1) and may activate the Keap1/Nrf2 pathway, contributing to cytoprotective and antioxidant effects. [2] The study distinguishes between the drug's action in the acidic stomach (where it is activated to inhibit H+/K+ ATPase) and its potential action at neutral pH in extra-gastric tissues (where the prodrug form may be active). The beneficial effects observed in the lung injury model, in mice which do not physiologically experience reflux, support a direct pharmacological effect beyond gastric acid suppression. [2] Retrospective clinical studies cited in the introduction and discussion suggest that PPI use is associated with beneficial outcomes in chronic lung diseases like idiopathic pulmonary fibrosis (IPF) and chronic obstructive pulmonary disease (COPD). [2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7047 mL | 13.5237 mL | 27.0475 mL | |
| 5 mM | 0.5409 mL | 2.7047 mL | 5.4095 mL | |
| 10 mM | 0.2705 mL | 1.3524 mL | 2.7047 mL |