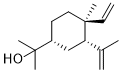
Elemol is a naturally occurring fragrance material isolated from olive oil and citronella oil. It is a yellow to brown yellow viscous liquid. It has been widely used as fixative or dressing agent in fine fragrance, beauty care, laundry care and household etc. Also used as raw material and intermediate for organic synthesis and pharmaceutical daily flavoring
Physicochemical Properties
| Molecular Formula | C15H26O |
| Molecular Weight | 222.36634 |
| Exact Mass | 222.198 |
| CAS # | 639-99-6 |
| Related CAS # | 639-99-6; |
| PubChem CID | 92138 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.942 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 16 |
| Complexity | 290 |
| Defined Atom Stereocenter Count | 3 |
| SMILES | CC(=C)[C@@H]1C[C@@H](CC[C@@]1(C)C=C)C(C)(C)O |
| InChi Key | GFJIQNADMLPFOW-VNHYZAJKSA-N |
| InChi Code | InChI=1S/C15H26O/c1-7-15(6)9-8-12(14(4,5)16)10-13(15)11(2)3/h7,12-13,16H,1-2,8-10H2,3-6H3/t12-,13+,15-/m1/s1 |
| Chemical Name | (1S,2S,4R)-(-)-alpha,alpha-Dimethyl-1-vinyl-o-menth-8-ene-4-methanol |
| Synonyms | AI3 00210; Elemol; AI3-00210; Elemol; 639-99-6; ALPHA-ELEMOL; 2-[(1r,3s,4s)-4-ethenyl-4-methyl-3-(prop-1-en-2-yl)cyclohexyl]propan-2-ol; 2-[(1R,3S,4S)-4-ethenyl-4-methyl-3-prop-1-en-2-ylcyclohexyl]propan-2-ol; L92AJ7G06I; .alpha.-Elemol; .beta.-Elemol; AI300210 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural product; Flavoring Agents |
| ln Vitro | Chamaecyparis obtusa has been traditionally used as an antibiotic agent and in cosmetics for the prevention of microorganism infection and skin troubles. Atopic dermatitis (AD) is a chronic inflammatory skin disease that encompasses immunologic responses, susceptibility factors and compromised skin-barrier function. Use of plant medicines in therapeutic treatment of AD has recently been suggested as an alternative therapeutic option. The present study examined the effect of elemol, an active component of Chamaecyparis obtusa, on AD using in vivo and in vitro models. RBL-2H3 cells were stimulated with concanavalin A and dinitrophenyl human serum albumin, and atopic dermatitis was induced in BALB/c mice by topical application of 2,4-dinitrochlorobenzene (DNCB) prior to elemol treatment. The mRNA expression was evaluated by reverse transcription quantitative polymerase chain reaction, and the levels of β-hexosaminidase and serum immunoglobulin E (IgE) were examined by ELISA. Histological changes were also performed by microscopy. Elemol attenuated the onset of AD-like skin lesions, reduced serum IgE levels and decreased mast cell infiltration into the dermis and hypodermis[1]. |
| ln Vivo | In addition, elemol downregulated the transcriptional expression of several pro-inflammatory cytokines, including TNF-α, IL-1β, IL-6 and IκBα, in the skin of the DNCB-induced animal models of AD. In the RBL-2H3 mast cell line, elemol significantly inhibited the mRNA expression of IL-4 and IL-13, and further attenuated the release of β-hexosaminidase from mast cells. Histological examination revealed that elemol significantly ameliorated the DNCB-induced dermal destruction in mice. The results of the present study suggested that elemol may have therapeutic potential in the treatment of AD due to its immunosuppressive effects[1]. |
| References |
[1]. Int J Mol Med. 2015 Aug;36(2):463-72. [2]. Chemotype diversity of Psidium guajava L. Phytochemistry. 2018 Jun 15;153:129-137. |
| Additional Infomation |
Elemol is a sesquiterpenoid that is isopropanol which is substituted at position 2 by a (3S,4S)-3-isopropenyl-4-methyl-4-vinylcyclohexyl group. It has a role as a fragrance and a plant metabolite. It is a sesquiterpenoid, a tertiary alcohol and an olefinic compound. Elemol has been reported in Magnolia officinalis, Heracleum dissectum, and other organisms with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.4970 mL | 22.4850 mL | 44.9701 mL | |
| 5 mM | 0.8994 mL | 4.4970 mL | 8.9940 mL | |
| 10 mM | 0.4497 mL | 2.2485 mL | 4.4970 mL |