Physicochemical Properties
| Molecular Formula | C22H29FN4O2 |
| Molecular Weight | 400.49 |
| Exact Mass | 400.227 |
| Elemental Analysis | C, 67.26; H, 7.76; F, 4.43; N, 13.07; O, 7.47 |
| CAS # | 2411748-50-8 |
| Related CAS # | EZM0414 TFA;2411759-92-5 |
| PubChem CID | 146395245 |
| Appearance | Off-white to light yellow solid powder |
| LogP | 2.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 29 |
| Complexity | 610 |
| Defined Atom Stereocenter Count | 2 |
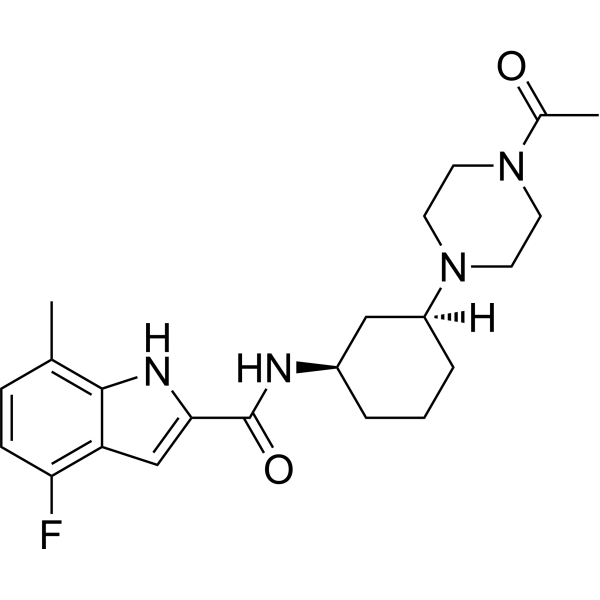
| SMILES | C1C=C(C2NC(=CC=2C=1F)C(N[C@@H]1CCC[C@H](N2CCN(CC2)C(=O)C)C1)=O)C |
| InChi Key | PGNLXEBQMQHFNK-SJORKVTESA-N |
| InChi Code | InChI=1S/C22H29FN4O2/c1-14-6-7-19(23)18-13-20(25-21(14)18)22(29)24-16-4-3-5-17(12-16)27-10-8-26(9-11-27)15(2)28/h6-7,13,16-17,25H,3-5,8-12H2,1-2H3,(H,24,29)/t16-,17+/m1/s1 |
| Chemical Name | N-[(1R,3S)-3-(4-acetylpiperazin-1-yl)cyclohexyl]-4-fluoro-7-methyl-1H-indole-2-carboxamide |
| Synonyms | EZM0414; 2411748-50-8; KCY37T9RXU; EZM-0414; UNII-KCY37T9RXU; N-[(1R,3S)-3-(4-acetylpiperazin-1-yl)cyclohexyl]-4-fluoro-7-methyl-1H-indole-2-carboxamide; N-((1R,3S)-3-(4-Acetylpiperazin-1-yl)cyclohexyl)-4-fluoro-7-methyl-1H-indole-2-carboxamide; 1H-Indole-2-carboxamide, N-((1R,3S)-3-(4-acetyl-1-piperazinyl)cyclohexyl)-4-fluoro-7-methyl-; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | SETD2 (IC50=18 nM) | ||||||||||||||||||||||
| ln Vitro |
EZM0414 suppresses the growth of a panel of MM and DLBCL cell lines with an IC50 of 0.24 μM for t(4;14) cells and 0.023 μM->10 μM for DLBCL cell lines [2]. Inhibition of SETD2 by EZM0414 results in potent anti-proliferative effects in a panel of MM and DLBCL cell lines. EZM0414 inhibited proliferation in both t(4;14) and non-t(4;14) MM cell lines, with higher anti-proliferative activity generally observed in the t(4;14) subset of MM cell lines. The median IC 50value for EZM0414 in t(4;14) cell lines was 0.24 μM as compared to 1.2 μM for non-t(4;14) MM cell lines. Additionally, inhibitory growth effects on DLBCL cell lines demonstrated a wide range of sensitivity with IC 50 values from 0.023 μM to >10 μM. [2] In vitro testing of EZM0414 in a safety panel consisting of 47 targets and a diversity panel of 72 kinases showed IC50 > 25 μM for all targets except D2 (IC50 = 13.0 μM, antagonist) and 5-HT1B (IC50 = 3.2 μM, agonist).[3] |
||||||||||||||||||||||
| ln Vivo |
EZM0414 (15 and 30 mg/kg, po, BID, Daily) suppresses tumor growth in a NOD SCID cell xenograft model of the human KMS-11 and is well tolerated [3]. EZM0414 (50 mg/kg, exhibited bucally) had almost 100% bioavailability in the stent and mouse, with t1/2 of 1.8 hours (mouse) and 3.8 hours (mouse) [3]. EZM0414 resulted in statistically significant potent antitumor activity compared to the vehicle control in three MM and four DLBCL cell line-derived xenograft models. In the t(4;14) MM cell line-derived xenograft model, KMS-11, robust tumor growth regressions were observed at the top two doses with maximal TGI of 95%. In addition, two non-t(4;14) MM (RPMI-8226, MM.1S) and two DLBCL xenograft models (TMD8, KARPAS422) demonstrated > 75% TGI; with two additional DLBCL models (WSU-DLCL2, SU-DHL-10) exhibiting > 50% TGI in response to EZM0414. In all models tested, the antitumor effects observed correlated with reductions in intratumoral H3K36me3 levels demonstrating on-target inhibition of SETD2 methyltransferase activity in vivo. [2] |
||||||||||||||||||||||
| Enzyme Assay |
SETD2 (1434-1711) Assay [3] The biochemical assay monitored the incorporation of the tritiated methyl group from S-adenosyl-methionine (SAM) into a biotinylated histone 3 peptide corresponding to residues 26-40. The sequence of the substrate peptide is biotin-Ahx-RKSAPATGGVKKPHR-NH2 and 3H-SAM was purchased from American Radiolabeled Chemicals, Inc. For the assay, 40 L of enzyme was incubated with 1 L of compound or DMSO for 30 minutes before initiating the reaction with 10 L of substrate solution in a 384- well assay plate. The assay was performed at room temperature in assay buffer composed of 25 mM bicine, pH 8.0, 7.5 mM -mercaptoethanol, 0.002 % Tween-20, and 0.01 % bovine skin gelatin (BSG). The reaction was quenched during the linear portion of product formation with 10 L of 1 mM S-adenosyl-homocysteine (SAH) and 1 mM SAM. From the quenched reaction, 50 L was transferred to a streptavidin-coated Flashplate (Perkin Elmer) and incubated for at least 2h before washing once with 0.1 % Tween-20. Signal from the 3H-labeled peptide captured by the streptavidin-coated plates was counted by a Topcount plate reader. Percent inhibition (%I) and IC50 values were calculated using equations 1 and 2 respectively. % |
|