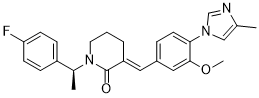
E2012 (E-2012) is a novel and potent γ-secretase modulator (GSM) γ-secretase modulator (GSM). It acts by inhibiting 3β-hydroxysterol Δ24-reductase (DHCR24) at the final step of the cholesterol biosynthesis. E2012 induces cataract in the rat by inhibiting DHCR24 at the final step of cholesterol synthesis with associated elevation in desmosterol within the lens, preceded by desmosterol changes that would serve as a predictive safety biomarker for lenticular opacity.
Physicochemical Properties
| Molecular Formula | C25H26FN3O2 |
| Molecular Weight | 419.5004 |
| Exact Mass | 419.201 |
| CAS # | 870843-42-8 |
| PubChem CID | 11560787 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.195g/cm3 |
| Boiling Point | 649.168ºC at 760 mmHg |
| Flash Point | 346.405ºC |
| Index of Refraction | 1.6 |
| LogP | 5.033 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 31 |
| Complexity | 649 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | CC1=CN(C=N1)C2=C(C=C(C=C2)/C=C/3\CCCN(C3=O)[C@@H](C)C4=CC=C(C=C4)F)OC |
| InChi Key | PUOAETJYKQITMO-LANLRWRYSA-N |
| InChi Code | InChI=1S/C25H26FN3O2/c1-17-15-28(16-27-17)23-11-6-19(14-24(23)31-3)13-21-5-4-12-29(25(21)30)18(2)20-7-9-22(26)10-8-20/h6-11,13-16,18H,4-5,12H2,1-3H3/b21-13+/t18-/m0/s1 |
| Chemical Name | (3E)-1-[(1S)-1-(4-fluorophenyl)ethyl]-3-[[3-methoxy-4-(4-methylimidazol-1-yl)phenyl]methylidene]piperidin-2-one |
| Synonyms | E 2012 E-2012 E2012 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | At IC50 values of 11.0 and 15.1 nM, respectively, E2012 has a concentration-dependent inhibitory effect on cholesterol production in primary cultured rat hepatocytes and HepG2 cells [1]. |
| ln Vivo | The in vivo concentrations of E 2012 in the lenses of cataract patients were significantly higher than the in vitro inhibitory effects following 13 weeks of repeated dosage. By blocking DHCR24 at the final stage of cholesterol synthesis, E 2012 causes cataracts in rats. This increases desmosterol in the lens and causes subsequent alterations in desmosterol, which can be used as a predictive safety biomarker for lens opacification [2]. |
| References |
[1]. E2012-induced cataract and its predictive biomarkers. Toxicol Sci. 2014 Jan;137(1):249-58. [2]. Portelius E, Van Broeck B, Andreasson U, Gustavsson MK, Mercken M, Zetterberg H, Borghys H, Blennow K.Acute effect on the Aβ isoform pattern in CSF in response to γ-secretase modulator and inhibitor treatment in dogs.J Alzheimers Dis. 2010;21(3):1005-12. |
| Additional Infomation |
E-2012 is a gamma secretase modulator that is being evaluated as a potential new treatment for Alzheimer's disease. Drug Indication Investigated for use/treatment in alzheimer's disease. Mechanism of Action Gamma secretase plays a role in the production of beta-amyloid, a major component of plaque in the brain, which is thought to be a cause of Alzheimer's disease. E2012 is a new chemical entity discovered by Eisai and, in preclinical (laboratory) research, has shown some potential to reduce the production of beta-amyloid by modulating the function of gamma secretase. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~119.19 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (5.96 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (5.96 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.96 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3838 mL | 11.9190 mL | 23.8379 mL | |
| 5 mM | 0.4768 mL | 2.3838 mL | 4.7676 mL | |
| 10 mM | 0.2384 mL | 1.1919 mL | 2.3838 mL |