Physicochemical Properties
| Molecular Formula | C21H27N3O3S |
| Molecular Weight | 401.52 |
| Exact Mass | 401.17731 |
| Elemental Analysis | C, 53.16; H, 6.16; Cl, 14.94; N, 8.86; O, 10.12; S, 6.76 |
| CAS # | 113558-89-7 |
| Related CAS # | E-4031;113559-13-0; 113558-89-7 |
| PubChem CID | 3185 |
| Appearance | Typically exists as solids at room temperature |
| Density | 1.243g/cm3 |
| Boiling Point | 561.7ºC at 760mmHg |
| Flash Point | 293.5ºC |
| Vapour Pressure | 1.21E-12mmHg at 25°C |
| Index of Refraction | 1.596 |
| LogP | 5.466 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 28 |
| Complexity | 603 |
| Defined Atom Stereocenter Count | 0 |
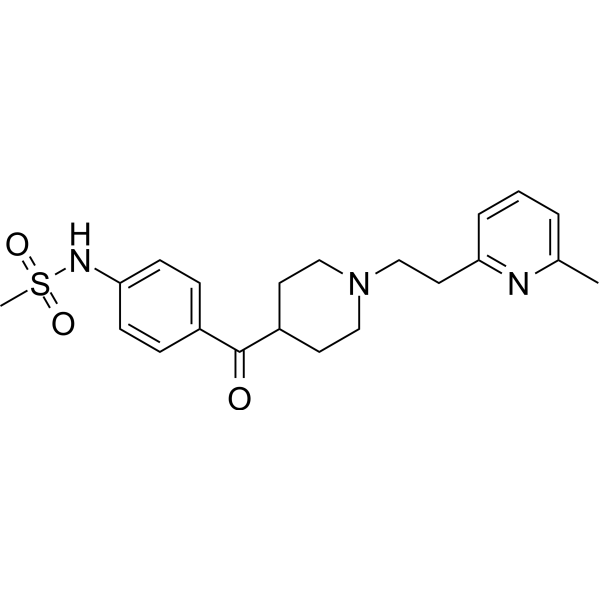
| SMILES | CC1=NC(=CC=C1)CCN2CCC(CC2)C(=O)C3=CC=C(C=C3)NS(=O)(=O)C |
| Synonyms | E-4031; E 4031; E4031; 1L1K8X5DF9; 2-Methyl-6-(2-(4-(4-methylsulfonylamino)benzoylpiperidin-1-yl)ethyl)pyridine; UNII-1L1K8X5DF9; N-(4-((1-(2-(6-Methyl-2-pyridinyl)ethyl)-4-piperidinyl)carbonyl)phenyl)methanesulfonamide; ...; 113558-89-7; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | hERG potassium channel |
| ln Vitro | Maximum diastolic potential (MDP) of single SAN cells in New Zealand white rabbits is significantly de-longitudated by E-4031 (0.1 ~ 10 μM), resulting in the MDP de-longest action scaffold from -58.8±0.9 mV at 1 μM Extended to -24.5±1.8 mV and from -58.2±2.1 to -19.6±1.8 mV at 10 μM[2]. In a dose-dependent manner, E-4031 (0.1 ~ 10 μM) inhibits the depolarization process. In single SAN cells of New Zealand white rabbits, the partial external current and subsequent tail current (ITD) during the complex partial process resulted in an 88% [sup] reduction in ITD [2]. |
| ln Vivo |
Bepridil and E-4031 prolonged QT interval and ARI in all LV layers, though the magnitude of prolongation was greatest in Mid, increasing the transmural ARI dispersion, particularly during bradycardia. Compared with E-4031, bepridil caused mild, reverse use-dependent changes in ventricular repolarization, and less ARI dispersion than E-4031 during slow ventricular pacing. Both drugs increased ARI(max) and cycle length at 50% of ARI(max), though the changes were smaller after bepridil than after E-4031 administration. Bradycardia after the administration of each drug induced no VTA; however, sympathetic stimulation induced sustained polymorphic VTA in two of five dogs treated with E-4031 versus no dog treated with bepridil.
Conclusions: Unlike the pure I(kr) blocker, E-4031, bepridil exhibited weak properties of reverse use-dependency and protected against sympathetic stimulation-induced VTA. It may be an effective supplemental treatment for recipients of implantable cardioverter defibrillator.[2] The role of the delayed rectifier current (IK) in impulse generation was studied in single sinoatrial nodal myocytes of the rabbit. We used the class III antiarrhythmic drug E-4031, which blocks IK in rabbit ventricular myocytes. In single sinoatrial nodal cells, E-4031 (0.1 mumol/L) significantly prolonged cycle length and action potential duration, depolarized maximum diastolic potential, and reduced both the upstroke velocity of the action potential and the diastolic depolarization rate. Half of the cells were arrested completely. At higher concentrations (1 and 10 mumol/L), spontaneous activity ceased in all cells. Three ionic currents fundamental for pacemaking, ie, IK, the long-lasting inward calcium current (ICa,L), and the hyperpolarization-activated current (I(f)), were studied by using the whole-cell and amphotericin-perforated patch technique. E-4031 blocked part of the outward current during depolarizing steps as well as the tail current upon subsequent repolarization (ITD) in a dose-dependent manner. E-4031 (10 mumol/L) depressed ITD (88 +/- 4%) (n = 6), reduced peak ICa,L at 0 mV (29 +/- 15%) (n = 4), but did not affect I(f). Lower concentrations did not affect ICa,L. Additional use of 5 mumol/L nifedipine demonstrated that ITD is carried in part by a calcium-sensitive current. Interestingly, complete blockade of IK and ICa,L unmasked the presence of a background current component with a reversal potential of -32 +/- 5.4 mV (n = 8) and a conductance of 39.5 +/- 5.6 pS/pF, which therefore can contribute both to the initial part of repolarization and to full diastolic depolarization[2]. |
| Cell Assay | Membrane potentials and currents were recorded by using both whole-cell21 and amphotericin–perforated patch techniques. The whole-cell method was used in only 5 of 10 cells in which the effects of 0.1 μmol/L E-4031 was studied. The amphotericin–perforated patch technique was used in all other experiments to reduce dilution of intracellular components, a possible cause of rundown of membrane currents. Electrodes were pulled from borosilicate glass (outer diameter, 1 mm; with a glass fiber inside the lumen) by using a vertical one-stage patch-electrode puller and were fire-polished. Electrode resistance varied between 3 and 5 MΩ[2]. |
| Animal Protocol |
Drug Administration[1] Bepridil, 4 mg/kg, was dissolved in sterile saline and administered intravenously in a single bolus.19 E-4013 was dissolved in sterile saline and administered intravenously in a bolus of 50 μg/kg, followed by a maintenance dose titrated between 5 and 20 μg/kg/min, to match the ARI prolongation induced by bepridil (280–360 ms) during pacing at 140 beats per minute (bpm).20 The data were collected during steady state, beginning 5 minutes after the administration of bepridil, or 5 minutes after the onset of a stable maintenance infusion of E-4031. Study Protocol and Data Collection[1] After the creation of complete AV block, the heart was paced at 100 bpm from the RV throughout the experiment. Completion of the following protocols of (1) and (2) was within 30 minutes. 1) Before the administration of bepridil or E-4031, the surface electrocardiogram, the transmural distribution of ARI, and ARI dispersion were recorded at a pacing cycle length of 428 ms (140 bpm), 500 ms (120 bpm), 600 ms (100 bpm), 750 ms (80 bpm), and 1,000 ms (60 bpm). The measurements were made at steady state, 30 seconds after the onset of each pacing rate. 2) To study the inducibility of VTA, (a) the back-up RV pacing rate was slowed to 40 bpm to allow lengthening of the cardiac cycle to 800–1,500 ms for 60 seconds, and (b) the left stellate ganglion was stimulated for 20 seconds during demand pacing at a cycle length of 750 ms. Completion of the following protocols of (3) was within 30 minutes. 3) These same protocols (1 and 2) were repeated after the administration of bepridil in five dogs and E-4031 in five other dogs. |
| References |
[1]. Effects of bepridil versus E-4031 on transmural ventricular repolarization and inducibility of ventricular tachyarrhythmias in the dog. Pacing Clin Electrophysiol. 2010 Aug;33(8):950-9. [2]. Effects of delayed rectifier current blockade by E-4031 on impulse generation in single sinoatrial nodal myocytes of the rabbit. Circ Res. 1995 Apr;76(4):607-15. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4905 mL | 12.4527 mL | 24.9054 mL | |
| 5 mM | 0.4981 mL | 2.4905 mL | 4.9811 mL | |
| 10 mM | 0.2491 mL | 1.2453 mL | 2.4905 mL |