Physicochemical Properties
| Molecular Formula | C25H23CLN2O5 |
| Molecular Weight | 466.913525819778 |
| Exact Mass | 466.129 |
| CAS # | 157922-77-5 |
| PubChem CID | 394851 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 4.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 33 |
| Complexity | 712 |
| Defined Atom Stereocenter Count | 1 |
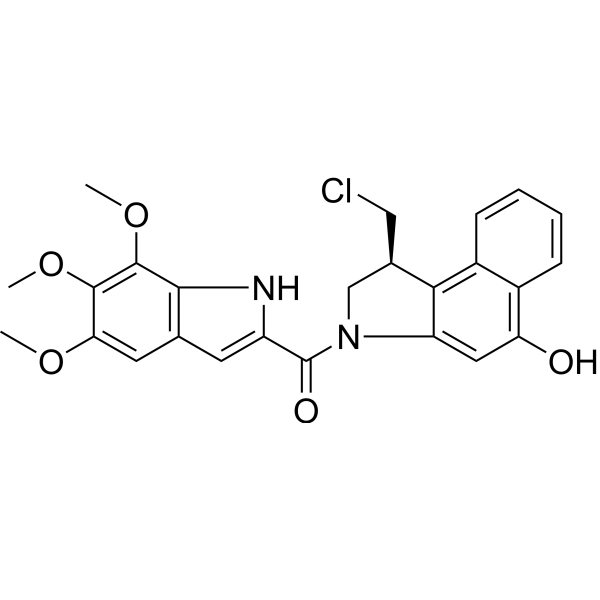
| SMILES | ClC[C@@H]1CN(C(C2=CC3C=C(C(=C(C=3N2)OC)OC)OC)=O)C2C=C(C3C=CC=CC=3C1=2)O |
| InChi Key | NRHDGIYFJJUFKN-CQSZACIVSA-N |
| InChi Code | InChI=1S/C25H23ClN2O5/c1-31-20-9-13-8-17(27-22(13)24(33-3)23(20)32-2)25(30)28-12-14(11-26)21-16-7-5-4-6-15(16)19(29)10-18(21)28/h4-10,14,27,29H,11-12H2,1-3H3/t14-/m1/s1 |
| Chemical Name | [(1S)-1-(chloromethyl)-5-hydroxy-1,2-dihydrobenzo[e]indol-3-yl]-(5,6,7-trimethoxy-1H-indol-2-yl)methanone |
| Synonyms | Duocarmycin TM; 157922-77-5; [(1S)-1-(chloromethyl)-5-hydroxy-1,2-dihydrobenzo[e]indol-3-yl]-(5,6,7-trimethoxy-1H-indol-2-yl)methanone; CBI-TMI; CHEMBL66051; SCHEMBL12596987; (1S)-1-(chloromethyl)-3-(5,6,7-trimethoxy-1H-indole-2-carbonyl)-1H,2H,3H-benzo[e]indol-5-ol; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | DNA synthesis; antitumor antibiotic |
| ln Vitro | Duocarmycin TM (60 μM; 4 d; BJAB and WSU-DLCL2 cells) is a cytotoxic drug that inhibits tumor cell proliferation [1]. |
| Cell Assay |
Cell viability assay [1] Cell Types: BJAB and WSU-DLCL2 Cell Tested Concentrations: 60 μM Incubation Duration: 4 days Experimental Results: Inhibited tumor cell proliferation, the IC50 values of BJAB and WSU-DLCL2 cells were 0.153 μM and 0.079 μM respectively. |
| References |
[1]. Immolation of p-Aminobenzyl Ether Linker and Payload Potency and Stability Determine the Cell-Killing Activity of Antibody-Drug Conjugates with Phenol-Containing Payloads. Bioconjug Chem. 2018 Feb 21;29(2):267-274. |
| Additional Infomation |
The valine-citrulline (Val-Cit) dipeptide and p-aminobenzyl (PAB) spacer have been commonly used as a cleavable self-immolating linker in ADC design including in the clinically approved ADC, brentuximab vedotin (Adcetris). When the same linker was used to connect to the phenol of the cyclopropabenzindolone (CBI) (P1), the resulting ADC1 showed loss of potency in CD22 target-expressing cancer cell lines (e.g., BJAB, WSU-DLCL2). In comparison, the conjugate (ADC2) of a cyclopropapyrroloindolone (CPI) (P2) was potent despite the two corresponding free drugs having similar picomolar cell-killing activity. Although the corresponding spirocyclization products of P1 and P2, responsible for DNA alkylation, are a prominent component in buffer, the linker immolation was slow when the PAB was connected as an ether (PABE) to the phenol in P1 compared to that in P2. Additional immolation studies with two other PABE-linked substituted phenol compounds showed that electron-withdrawing groups accelerated the immolation to release an acidic phenol-containing payload (to delocalize the negative charge on the anticipated anionic phenol oxygen during immolation). In contrast, efficient immolation of LD4 did not result in an active ADC4 because the payload (P4) had a low potency to kill cells. In addition, nonimmolation of LD5 did not affect the cell-killing potency of its ADC5 since immolation is not required for DNA alkylation by the center-linked pyrrolobenzodiazepine. Therefore, careful evaluation needs to be conducted when the Val-Cit-PAB linker is used to connect antibodies to a phenol-containing drug as the linker immolation, as well as payload potency and stability, affects the cell-killing activity of an ADC.[1] Solid-phase synthesis allowed the rapid generation of a peptide–drug conjugate. A peptide targeting the Thomsen-Friedenreich antigen (TFα) was conjugated to the alkylating subunit of the potent cytotoxin duocarmycin SA. The compound, containing a cathepsin B cleavable linker, was shown to be active and selective against TFα expressing tumor cell lines.https://pubs.acs.org/doi/10.1021/acs.bioconjchem.0c00282 |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~107.09 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.35 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.35 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1417 mL | 10.7087 mL | 21.4174 mL | |
| 5 mM | 0.4283 mL | 2.1417 mL | 4.2835 mL | |
| 10 mM | 0.2142 mL | 1.0709 mL | 2.1417 mL |