Dotanoc is a ligand to make gallium Ga 68-DOTANOC, which is a gallium Ga 68-radiolabeled analogue of somatostatin that may be used in conjunction with positron emission tomography (PET) to image neuroendocrine tumors and metastases.
Physicochemical Properties
| Molecular Formula | C69H92N14O18S2 |
| Molecular Weight | 1469.68179416656 |
| Exact Mass | 1454.64 |
| Elemental Analysis | C, 56.93; H, 6.51; N, 13.47; O, 18.68; S, 4.40 |
| CAS # | 619300-53-7 |
| Related CAS # | 619300-53-7;1027785-95-0 (Ga68 complex);1040397-47-4 (Ga68 complex); |
| PubChem CID | 15518147 |
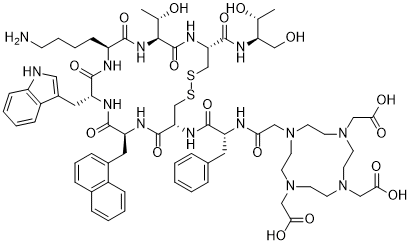
| Sequence | DOTA-{d-Phe}-Cys-{Nal}-{d-Trp}-Lys-Thr-Cys-Thr-ol (Disulfide bridge: Cys2-Cys7) |
| SequenceShortening | DOTA-{d-Phe}-C-{Nal}-{d-Trp}-KTCT-ol (Disulfide bridge: Cys2-Cys7) |
| Appearance | Solid powder |
| LogP | -5.6 |
| Hydrogen Bond Donor Count | 16 |
| Hydrogen Bond Acceptor Count | 24 |
| Rotatable Bond Count | 26 |
| Heavy Atom Count | 102 |
| Complexity | 2730 |
| Defined Atom Stereocenter Count | 10 |
| SMILES | C[C@H]([C@H]1C(=O)N[C@@H](CSSC[C@@H](C(=O)N[C@H](C(=O)N[C@@H](C(=O)N[C@H](C(=O)N1)CCCCN)CC2=CNC3=CC=CC=C32)CC4=CC=CC5=CC=CC=C54)NC(=O)[C@@H](CC6=CC=CC=C6)NC(=O)CN7CCN(CCN(CCN(CC7)CC(=O)O)CC(=O)O)CC(=O)O)C(=O)N[C@H](CO)[C@@H](C)O)O |
| InChi Key | PZCJTYVWTGPGOH-OKVMNLLFSA-N |
| InChi Code | InChI=1S/C69H94N14O17S2/c1-42(85)55(39-84)76-68(99)57-41-102-101-40-56(77-64(95)52(31-44-13-4-3-5-14-44)72-58(87)35-80-23-25-81(36-59(88)89)27-29-83(38-61(92)93)30-28-82(26-24-80)37-60(90)91)67(98)75-53(32-46-17-12-16-45-15-6-7-18-48(45)46)66(97)74-54(33-47-34-71-50-20-9-8-19-49(47)50)65(96)73-51(21-10-11-22-70)63(94)79-62(43(2)86)69(100)78-57/h3-9,12-20,34,42-43,51-57,62,71,84-86H,10-11,21-33,35-41,70H2,1-2H3,(H,72,87)(H,73,96)(H,74,97)(H,75,98)(H,76,99)(H,77,95)(H,78,100)(H,79,94)(H,88,89)(H,90,91)(H,92,93)/t42-,43-,51+,52-,53+,54-,55-,56+,57+,62+/m1/s1 |
| Chemical Name | Dotanoc DOTA-NOC DOTA-Nal3-octreotide InChi Key |
| Synonyms | Dotanoc; DOTA-NOC; Dotanoc; DOTA-NOC; DOTA-nal3-octreotide; 1IIY9GZY9U; UNII-1IIY9GZY9U; 619300-53-7; L-Cysteinamide, N-(2-(4,7,10-tris(carboxymethyl)-1,4,7,10-tetraazacyclododec-1-yl)acetyl)-D-phenylalanyl-L-cysteinyl-3-(1-naphthalenyl)-L-alanyl-D-tryptophyl-L-lysyl-L-threonyl-N-((1R,2R)-2-hydroxy-1-(hydroxymethyl)propyl)-, cyclic (2->7)-disulfide; CHEMBL4297373; DOTA-Nal3-octreotide; DOTA-(1-Nal)3-Octreotide; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Somatostatin receptor ; RDC Peptide |
| ln Vitro |
IC50 (μM) values for binding affinity to human somatostatin adsorbed to SST 1–5 [1]. Hsst1 hsst2 hsst3 hsst4 hsst5 compound InIII-DOTA-NOC >10000 2.9 8 227 11.2 YIII-DOTA-NOC >10000 3.3 26 >10000 10.4
Receptor binding and affinity profiles [1] Table 2 shows the IC50 values of the three radiopeptides studied in this work as their YIII (InIII) complexed versions and of YIII-DOTA-LAN for the five somatostatin receptor subtypes. The values were obtained by performing complete displacement experiments with the universal somatostatin radioligand [125I][Leu8, D-Trp22, Tyr25]-somatostatin-28 on membranes from cells expressing the receptor subtypes and were compared with the data for somatostatin-28. All compounds bound specifically to sstr2 with IC50 values between 3 and 23 nM. High specific binding affinities to sstr3 were also found for InIII-DOTA-NOC (IC50=8±2 nM), YIII-DOTA-NOC (IC50=26±1.9 nM) and YIII-DOTA-OC (IC50=27±8 nM). YIII-DOTA-TOC and YIII-DOTA-LAN showed only very low affinities to sstr3 (IC50 ≥300 nM). All metallopeptides also showed specific binding to sstr5; YIII-DOTA-TOC bound with IC50=204±92 nM and YIII-DOTA-OC with IC50=58±22 nM whereas YIII-DOTA-LAN (IC50=16.3±3.4 nM) and YIII-DOTA-NOC (IC50=10.4±1.6 nM) showed rather high affinities to sstr5. In vitro internalization studies in AR4-2J cells [1] Figure 3 shows the results in respect of the time-dependent internalization of [111In/115In]DOTA-NOC, [111In/115In]DOTA-TOC and [111In/115In]DOTA-OC into AR4-2J rat pancreatic tumour cells during a 240-min incubation period at 37°C. About 85%–95% of totally internalized ligand was specifically internalized. At 30 min and 1.67 nM concentration, [111In]DOTA-NOC showed 7.2%±1.0% specific cell uptake of the total activity administered which increased to 24.8%±1.6% uptake at 4 h. A tendency to reach a plateau was found at 24 h for all three peptides (data not shown). The uptake of [111In]DOTA-OC at 30 min was only 1.0%±0.2% and increased to 7.5%±0.3% at 4 h, whereas [111In]DOTA-TOC showed 2.0%±0.7% internalization at 30 min, rising to 11.6%±0.8% at 4 h. The percentage of internalized peptide measured at 30 min as a function of concentration is shown in Fig. 4. The rate of externalization is shown in Fig. 5. In these experiments, 111In labelled peptides were allowed to internalize for 120 min; cells were then washed twice with PBS before removing the receptor-bound ligand with the glycine buffer. Medium was then added and removed after 10 min, 30 min, 60 min, 120 min and 240 min and measured for radioactivity. Up to 60 min, the three peptides showed insignificant differences in the externalization rate. Thereafter the extent of externalized ligand was highest for [111In]DOTA-OC, with about 60% at 4 h compared with 47%±4% for [111In]DOTA-NOC and 43%±2% for [111In]DOTA-TOC. At 4 h the externalization curve of all peptides showed a plateau. Study of the chemical structure of the externalized [111In]DOTA-NOC and [111In]DOTA-TOC by HPLC gave no indication of metabolites. The peptides externalized were intact. |
| ln Vivo |
Biodistribution studies in rats [1] The 4-h, 24-h and 48-h uptake values of [111In]DOTA-NOC and [111In]DOTA-TOC in sst receptor-positive organs, including pancreas, adrenals, pituitary, stomach and CA 20948 rat pancreatic tumour, as well as the kidneys, liver, spleen, femur and blood are shown in Table 3. Both radiopeptides displayed rapid blood clearance with less than 0.02% ID/g remaining in the blood at 4 h. There was also fast clearance from all sstr-negative tissues except the kidneys. The excretion of both peptides was mainly by the kidneys. [111In]DOTA-NOC had a significantly higher uptake than [111In]DOTA-TOC in all sstr-positive tissues at all time points, e.g. at 4 h (tumour, 3.9%±0.6% ID/g vs 1.15%±0.16% ID/g; pancreas, 7.75%±1.4% ID/g vs 2.57%±0.08% ID/g; adrenals, 7.43%±0.42% ID/g vs 3.62%±0.14% ID/g; pituitary, 1.66%±0.20% ID/g vs 1.48%±0.07% ID/g). To estimate the uptake in sstr-positive organs which may be due to receptor subtype expression other than sstr2, in vivo blocking studies were performed in normal rats using different blocking agents like [DTPA0-Tyr3-Thr8]-octreotide (DTPA-TATE), an sstr2-specific ligand (IC50=3.9±1 nM), and InIII-DOTA-NOC (IC50: sstr2=2.9±0.1 nM, sstr3=8±2 nM, sstr5=11.2±3.5 nM). In the adrenals, blocking with only 25 μg of InIII-DOTA-NOC showed a very efficient reduction of about 95% whereas 25 μg of the sstr2-selective ligand DTPA-TATE resulted in only about 75% blocking. A significantly higher blocking effect was also found with InIII-DOTA-NOC in the pancreas, pituitary and stomach (data not shown). |
| Cell Assay |
Medium was removed from the six-well plates and cells were washed once with 2 ml of internalization buffer (DMEM, 1% fetal bovine serum, amino acids and vitamins, pH 7.4). Furthermore, 1.5 ml internalization buffer was added to each well and the plates were incubated at 37°C for about 1 h. Thereafter approximately 500,000 cpm or 0.02 MBq/well 111In/115In-labelled peptides (2.5 pmol/well) to a final concentration of 1.67 nM were added to the medium and the cells were incubated at 37°C for the indicated time periods in triplicate. Internalization was also studied using three different concentrations of [111In/115In]DOTA-NOC (0.15 pmol/well or 0.1 nM, 2.5 pmol/well or 1.67 nM, and 10 pmol/well or 6.67 nM). To determine non-specific membrane binding and internalization, cells were incubated with radioligand in the presence of 1 μM octreotide. Cellular uptake was stopped by removing medium from the cells and by washing twice with 1 ml of ice-cold phosphate-buffered saline (PBS). Acid wash for 10 min with a 0.1 M glycine buffer pH 2.8 on ice was also performed twice. This was shown previously to be sufficient to remove >90% of receptor-bound radioligand. This procedure was performed to distinguish between membrane-bound (acid-releasable) and internalized (acid-resistant) radioligand. Finally, the cells were treated with 1 N NaOH. The culture medium, the receptor-bound and the internalized fraction were measured radiometrically in a gamma-counter.[1] Radioligand externalization studies: AR4-2J cells (1 million) were incubated with 2.5 pmol/well or 1.67 nM [111In/115In]-labelled DOTA-NOC, DOTA-TOC or DOTA-OC for 120 min, then the medium was removed and the wells were washed twice with 1 ml ice-cold PBS. In each experiment an acid wash for 10 min on ice with a glycine buffer of pH 2.8 was performed to remove the receptor-bound ligand. Cells were then incubated again at 37°C with fresh externalization buffer (DMEM containing 1% fetal bovine serum pH 7.4). After different time points the external medium was removed for quantification of radioactivity in a gamma-counter and replaced with fresh 37°C externalization medium. Internalized ligand was extracted in 1 N NaOH, removed and quantified in a gamma-counter. The recycled fraction was expressed as a percentage of the total internalized amount per 1 million cells, and the stability of the externalized peptides was determined using HPLC after removal of the solvent by a centrifugal evaporator [1]. |
| Animal Protocol |
Biodistribution [1] Animal experiments were performed in compliance with the regulations of our institutions and with generally accepted guidelines governing such work. Male Lewis rats (200–250 g) bearing the CA20948 pancreatic tumour (0.4–3.5 g) were used in the experiments. Rats were injected under ether anaesthesia with 2–3 MBq of 0.34 nmol (0.5 μg total peptide mass) [111In]DOTA-NOC in 0.5 ml saline into the dorsal vein of the penis. At several time points, rats were sacrificed under ether anaesthesia. Organs and blood were collected and the radioactivity in these samples was determined using a gamma-counter. In order to determine the non-specific uptake of the radiopeptides, rats were injected with 0.5 mg octreotide in 0.5 ml saline as a co-injection with the radioligand. To study the sstr2-, 3- and 5-related specific uptake of [111In]DOTA-NOC in the SRIF receptor-positive tissues, blocking studies were designed with two different somatostatin analogues: DTPA-TATE (sstr2-selective ligand) and InIII-DOTA-NOC (sstr2, 3, and 5 affinity). Twenty-five micrograms of these peptides was co-injected with 2–3 MBq [111In]DOTA-NOC (0.34 nmol in 0.5 ml saline) into the dorsal vein of the penis of non-tumour-bearing male Lewis rats. Rats were sacrificed at 24 h and the organs of interest collected and counted for radioactivity. |
| ADME/Pharmacokinetics |
Cell uptake and release [1] In order to obtain a defined and homogeneous metallopeptide, [111In]DOTA-NOC was complexed with "cold" InIII to yield [111In, 115In]DOTA-NOC. At 4 h of internalization, [111In/115In]DOTA-NOC showed a factor of 2 higher specific cell uptake than [111In/115In]DOTA-TOC and a factor of about 3 higher than [111In/115In]DOTA-OC at 1.67 nM peptide concentration per 1 million cells. The difference was even more pronounced at 0.1 nM concentration (Fig. 4). This order follows the receptor affinity of the three radiopeptides, indicating that receptor affinity is the major factor determining the rate of internalization. As the addition of excess cold octreotide inhibits 90% of the uptake, it can be considered as specific and receptor mediated. In the time interval of the study, no steady state was reached, but the distinct leveling off of [111In/115In]DOTA-NOC uptake indicates that steady state was closely approached; we explain this by the onset of efflux of radiopeptides that were shown to be structurally intact. If, upon internalization of the radioligand for 2 h, the cells were exposed to the culture medium, a time-dependent efflux of the radiopeptides could again be observed, indicating rapid recycling to the extracellular medium. A steady state was reached already after 2–3 h of release (Fig. 5). This is in agreement with data that we have published previously on [67Ga]NODAGA-TOC [49]. We interpret this as beginning reactivation of the receptors by the intact externalized peptides and concomitant re-endocytosis. The finding that the weakest binder [111In]DOTA-OC apparently shows the most efficient externalization fits with this explanation. The fact that the externalized peptides are still intact upon release is another indication that this is the correct interpretation. It is also in keeping with the conclusion drawn from data obtained by Koenig et al. Biodistribution studies [1] The biodistribution studies in CA20948-bearing tumour rats demonstrated superior uptake of [111In]DOTA-NOC compared with [111In]DOTA-TOC at 4 h, 24 h and 48 h in receptor-positive normal tissues (except the pituitary) and the tumour. An estimated area under the curve showed an improvement of approximately 2.5-fold in the tumour. This improvement is likely due to the improved sst2, sst3 and sst5 receptor affinity and the significantly faster rate of internalization, as exemplified in the AR4-2J cell line. The octreotide co-injection experiment demonstrated that the uptake is specific and receptor mediated. Radiometal labelled radiopeptides show high and persistent kidney uptake, limiting their therapeutic potential. One of the goals in the design of new somatostatin-based radioligands is to reduce their uptake in the kidney. Indeed, the tumour-to-kidney ratio of [111In]DOTA-NOC is improved 2.5-fold compared with [111In]DOTA-TOC. Despite the distinctly higher lipophilicity of [111In]DOTA-NOC over [111In]DOTA-TOC, the uptake in the liver and the intestines is surprisingly low. In addition, the long residence time of the new radiopeptide in the tumour indicates that it is not only suitable for imaging but also efficacious in targeted radiotherapy when labelled with 90Y and/or 177Lu provided that there is no significant difference among these MIII radiometals. To understand the contribution of the high uptake values in SRIF receptor-positive organs due to the different subtype affinities of [111In]DOTA-NOC, the uptake in these tissues was studied using different blocking agents, namely DTPA-TATE, an sstr2-specific ligand with IC50=3.9±1 nM, and InIII-DOTA-NOC, which has a high affinity to sstr2, sstr3 and sstr5. The higher blocking efficiency of InIII-DOTA-NOC in the adrenals, pancreas, stomach and pituitary may indicate that part of the radioligand uptake is due to the improved receptor subtype profile. These organs have previously been shown to express different receptor subtypes, at least at the mRNA level: sstr2: adrenals, pituitary and pancreas; sstr3: pituitary, pancreas and stomach; sstr5: adrenals, pituitary, pancreas and stomach. In conclusion, we have developed a new radiopeptide based on somatostatin which promises to target a broader range of somatostatin receptors and concomitantly a larger spectrum of tumours. These preclinical data indicate that [111In]DOTA-NOC is superior to existing and well-studied radiolabelled somatostatin analogues. Indeed, the predictions from these preclinical studies have been confirmed in initial clinical studies in which excellent images of thyroid cancer patients have been obtained. We assume that [90Y]/[177Lu]DOTA-NOC will have similar favourable properties. |
| References |
[1]. DOTA-NOC, a high-affinity ligand of somatostatin receptor subtypes 2, 3 and 5 for labelling with various radiometals. Eur J Nucl Med Mol Imaging. 2003 Oct;30(10):1338-47. |
| Additional Infomation | Earlier studies have shown that modification of the octapeptide octreotide in positions 3 and 8 may result in compounds with increased somatostatin receptor affinity that, if radiolabelled, display improved uptake in somatostatin receptor-positive tumours. The aim of a recent research study in our laboratory was to employ the parallel peptide synthesis approach by further exchanging the amino acid in position 3 of octreotide and coupling the macrocyclic chelator DOTA(1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid) to these peptides for labelling with radiometals like gallium-67 or -68, indium-111, yttrium-90 and lutetium-177. The purpose was to find radiopeptides with an improved somatostatin receptor binding profile in order to extend the spectrum of targeted tumours. A first peptide, [111In,90Y-DOTA]-1-Nal3-octreotide (111In,90Y-DOTA-NOC), was isolated which showed an improved profile. InIII-DOTA-NOC exhibited the following IC50 values (nM) when studied in competition with [125I][Leu8, d-Trp22, Tyr25]somatostatin-28 (values for YIII-DOTA-NOC are shown in parentheses): sstr2, 2.9 +/- 0.1 (3.3 +/- 0.2); sstr3, 8 +/- 2 (26 +/- 1.9); sstr5, 11.2 +/- 3.5 (10.4 +/- 1.6). Affinity towards sstr1 and 4 was very low or absent. InIII-DOTA-NOC is superior to all somatostatin-based radiopeptides having this particular type of binding profile, including DOTA-lanreotide, and has three to four times higher binding affinity to sstr2 than InIII,YIII-DOTA-Tyr3-octreotide (InIII,YIII-DOTA-TOC). In addition, [111In]DOTA-NOC showed a specific and high rate of internalization into AR4-2J rat pancreatic tumour cells which, after 4 h, was about two times higher than that of [111In]DOTA-TOC and three times higher than that of [111In]DOTA-octreotide ([111In]DOTA-OC). The internalized radiopeptides were externalized intact upon 2 h of internalization followed by an acid wash. After 2-3 h of externalization a plateau is reached, indicating a steady-state situation explained by reactivation of the receptors followed by re-endocytosis. Biodistribution studies in CA 20948 tumour-bearing rats showed rapid clearance from all sstr-negative tissues except the kidneys. At 4 h the uptake of [111In]DOTA-NOC in the tumour and sstr-positive tissues, such as adrenals, stomach and pancreas, was three to four times higher than that of [111In]DOTA-TOC. Differential blocking studies indicate that this is at least partially due to the uptake mediated by sstr3 and sstr5. These very promising preclinical data justify the use of this new radiopeptide for imaging and potentially internal radiotherapy studies in patients. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~50 mg/mL (34.4 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.6804 mL | 3.4021 mL | 6.8042 mL | |
| 5 mM | 0.1361 mL | 0.6804 mL | 1.3608 mL | |
| 10 mM | 0.0680 mL | 0.3402 mL | 0.6804 mL |