Physicochemical Properties
| Molecular Formula | C30H31N3O3 |
| Molecular Weight | 481.5854 |
| Exact Mass | 597.247 |
| CAS # | 153653-30-6 |
| Related CAS # | Dofequidar;129716-58-1 |
| PubChem CID | 9960287 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 44 |
| Complexity | 771 |
| Defined Atom Stereocenter Count | 0 |
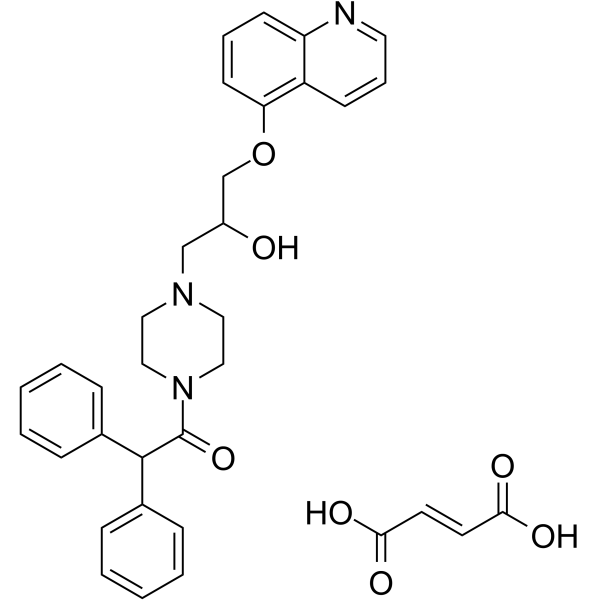
| SMILES | C1CN(CCN1CC(COC2=CC=CC3=C2C=CC=N3)O)C(=O)C(C4=CC=CC=C4)C5=CC=CC=C5.C(=C/C(=O)O)\C(=O)O |
| InChi Key | QIAVTDQTRFYXSD-WLHGVMLRSA-N |
| InChi Code | InChI=1S/C30H31N3O3.C4H4O4/c34-25(22-36-28-15-7-14-27-26(28)13-8-16-31-27)21-32-17-19-33(20-18-32)30(35)29(23-9-3-1-4-10-23)24-11-5-2-6-12-24;5-3(6)1-2-4(7)8/h1-16,25,29,34H,17-22H2;1-2H,(H,5,6)(H,7,8)/b;2-1+ |
| Chemical Name | (E)-but-2-enedioic acid;1-[4-(2-hydroxy-3-quinolin-5-yloxypropyl)piperazin-1-yl]-2,2-diphenylethanone |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Treatment with Dofequidar fumarate dose-dependently reduced the proportion of Hoechst33342-effluxing side population (SP) cells, a cancer stem-like cell population, in various cancer cell lines (HeLa, BSY-1, KM12). For example, in HeLa cells, 2.5 µM dofequidar significantly decreased the SP fraction. [4] In a vesicle transport assay using membrane vesicles overexpressing ABCG2/BCRP, Dofequidar fumarate inhibited the ATP-dependent uptake of [³H]methotrexate ([³H]MTX) in a dose-dependent manner, similar to the known ABCG2 inhibitor fumitremorgin C (FTC). Verapamil (an ABCB1 inhibitor) did not show inhibition. [4] Dofequidar fumarate increased the intracellular accumulation of mitoxantrone (MXR) in K562 cells stably overexpressing ABCG2/BCRP (K562/BCRP), as measured by flow cytometry, indicating inhibition of ABCG2-mediated drug efflux. [4] Dofequidar fumarate sensitized ABCG2/BCRP-overexpressing KB/BCRP cells to mitoxantrone (MXR), reversing their chemoresistance. Treatment with 10 µM dofequidar reduced the GI₅₀ of MXR in KB/BCRP cells from 30.46 nM to 3.84 nM, similar to the effect of 1 µM FTC. [4] Dofequidar fumarate sensitized SP cells isolated from HeLa, HBC-5, and BSY-1 cell lines to chemotherapeutic drugs (topotecan, mitoxantrone). For instance, in HeLa SP cells, dofequidar (3 µM) reduced the GI₅₀ for topotecan from 77.0 nM to 45.6 nM, making it comparable to non-SP (NSP) cells (55.6 nM). [4] |
| ln Vivo |
In nude mice bearing xenograft tumors derived from HeLa SP cells (which are chemoresistant), oral administration of Dofequidar fumarate (200 mg/kg) prior to intravenous irinotecan (CPT-11, 67 mg/kg) significantly enhanced the antitumor efficacy of CPT-11. The combination therapy (CPT-11 + dofequidar) drastically reduced tumor volume compared to CPT-11 alone, which only arrested tumor growth without reduction. The combination therapy was as effective against SP-derived tumors as CPT-11 alone was against NSP-derived tumors. [4] Dofequidar fumarate alone (200 mg/kg, orally) had almost no effect on the growth of SP- or NSP-derived tumors in vivo. [4] The combination therapy (CPT-11 + dofequidar) appeared well-tolerated in mice, with very small changes in body weight observed. [4] |
| Enzyme Assay |
An in vitro vesicle transport assay was performed to assess inhibition of ABCG2/BCRP function. Membrane vesicles prepared from insect cells overexpressing human ABCG2/BCRP protein were used. The assay mixture contained 50 mM MOPS-Tris (pH 7.0), 7.5 mM MgCl₂, 70 mM KCl, 160 nM unlabeled methotrexate (MTX), the test inhibitor (dofequidar, FTC, or verapamil), 1 mCi/mL [³H]MTX, and membrane vesicles (25 µg protein) in a 30 µL volume. The mixture was kept on ice for 5 minutes, then the transport reaction was initiated by adding 20 µL of 10 mM ATP (or AMP as a negative control) and incubating at 37°C for 5 minutes. The reaction was terminated by adding an ice-cold stop solution (40 mM MOPS-Tris pH 7.0, 70 mM KCl). The membrane vesicles were collected on a filter plate, washed, and the retained radioactivity was measured with a liquid scintillation counter to quantify ATP-dependent [³H]MTX uptake. [4] |
| Cell Assay |
Side Population (SP) cell analysis and sorting: Cells were trypsinized, resuspended in ice-cold HBSS with 2% FBS, and stained with 2.5-15 µg/mL Hoechst33342 dye for 60 minutes at 37°C in the presence or absence of transporter inhibitors (e.g., dofequidar, reserpine, FTC). After washing, cells were analyzed or sorted using a flow cytometer (FACS Vantage). SP cells were identified as the low-fluorescence population in the Hoechst Blue vs. Hoechst Red dot plot. [4] Intracellular drug accumulation assay: K562/BCRP cells (5 x 10⁵ cells) were incubated with 3 µM mitoxantrone (MXR) for 30 minutes at 37°C with or without dofequidar or FTC. Cells were then washed with ice-cold PBS and subjected to fluorescence analysis by flow cytometry (excitation 630 nm) to measure intracellular MXR fluorescence. [4] Cell viability assay (MTS assay): Cells (e.g., sorted SP/NSP cells, KB/BCRP cells) were cultured in medium containing serial dilutions of anticancer drugs (e.g., mitoxantrone, topotecan) with or without dofequidar for 3 days. Cell viability was then assessed by adding MTS reagent, incubating for 1 hour, and measuring the absorbance with a microplate spectrophotometer. The 50% growth inhibition (GI₅₀) values were determined. [4] Gene silencing: HeLa cells were transfected with ABCG2-specific siRNA or control siRNA using a lipid-based transfection reagent. After 24-48 hours, cells were harvested for analysis of ABCG2 protein expression by immunoblotting and for SP analysis by Hoechst33342 staining. [4] |
| Animal Protocol |
Tumor xenograft model: Sorted HeLa SP or NSP cells (100 to 10,000 cells) were suspended in DMEM growth medium, mixed 1:1 with Matrigel, and subcutaneously injected into 5-6 week-old female BALB/c nude mice. Tumor formation was monitored. For secondary transplantation, established tumors were resected, diced into pieces, and transplanted into new mice. [4] Drug treatment: When secondary tumor volume reached approximately 100 mm³, mice were sorted into groups. Dofequidar fumarate was administered orally at a dose of 200 mg/kg (vehicle/control not specified in method details, but water was used as control in results) 30 minutes before the intravenous administration of irinotecan (CPT-11) at 67 mg/kg. This treatment schedule (dofequidar + CPT-11) was repeated on days 0, 4, and 8. Tumor size and body weight were measured every 2-3 days. [4] |
| References |
[1]. Potentiation of the antitumor activity by a novel quinoline compound, MS-209, in multidrug-resistant solid tumor cell lines. Oncol Res. 1997;9(2):61-9. [2]. MS-209, a quinoline-type reversal agent, potentiates antitumor efficacy of docetaxel in multidrug-resistant solid tumor xenograft models. Clin Cancer Res. 2002 Feb;8(2):582-8. [3]. A new quinoline derivative MS-209 reverses multidrug resistance and inhibits multiorgan metastases by P-glycoprotein-expressing human small cell lung cancer cells. Jpn J Cancer Res. 2001 Jul;92(7):785-92. [4]. Dofequidar fumarate sensitizes cancer stem-like side population cells to chemotherapeutic drugs by inhibiting ABCG2/BCRP-mediated drug export. Cancer Sci. 2009 Nov;100(11):2060-8. |
| Additional Infomation |
See also: Dofequidar Fumarate (annotation moved to). Dofequidar fumarate (MS-209) is an orally active quinoline-derivative compound initially developed as a multidrug resistance (MDR) reversing agent targeting ABCB1/P-gp and ABCC1/MRP1. [4] This study provides evidence that Dofequidar fumarate also potently inhibits the ABCG2/BCRP transporter, which is highly expressed in cancer stem-like side population (SP) cells. By inhibiting ABCG2, it sensitizes these resistant cells to chemotherapeutic drugs like mitoxantrone, topotecan, and irinotecan (via its active metabolite SN-38). [4] The mechanism of action involves inhibiting the drug-efflux function of ABC transporters, thereby increasing intracellular concentration of chemotherapeutic agents and overcoming chemoresistance, particularly in stem-like cancer cells. [4] The study suggests that combining Dofequidar fumarate with conventional chemotherapy (e.g., CPT-11) could be a strategy to target chemotherapy-resistant cancer stem cells, potentially improving treatment outcomes and reducing recurrence. [4] Phase III clinical trials for breast cancer indicated that dofequidar combined with CAF (cyclophosphamide, doxorubicin, fluorouracil) showed improved progression-free and overall survival in patients with no prior therapy, aligning with the concept of targeting pre-existing resistant stem-like cells. [4] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~167.32 mM) H2O : ~1 mg/mL (~1.67 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.18 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), suspension solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0765 mL | 10.3823 mL | 20.7646 mL | |
| 5 mM | 0.4153 mL | 2.0765 mL | 4.1529 mL | |
| 10 mM | 0.2076 mL | 1.0382 mL | 2.0765 mL |