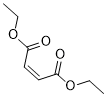
Diethylmaleate is the diethyl ester of maleic acid and a glutathione-depleting compound that inhibits NFkB. DEM induces upregulation of GSH(L-c-glutamyl-L-cysteinyl-glycine) metabolism, and the downregulation of pathways of cancer, chemokine signaling, cytokine-cytokine receptor, and focal adhesion in transformed cells. DEM appears to modify microenvironment of transformed cells thereby restraining tumor cell growth.
Physicochemical Properties
| Molecular Formula | C8H12O4 | |
| Molecular Weight | 172.18 | |
| Exact Mass | 172.073 | |
| CAS # | 141-05-9 | |
| Related CAS # |
|
|
| PubChem CID | 5271566 | |
| Appearance | Colorless to light yellow liquid | |
| Density | 1.1±0.1 g/cm3 | |
| Boiling Point | 214.0±0.0 °C at 760 mmHg | |
| Melting Point | −10 °C(lit.) | |
| Flash Point | 93.3±0.0 °C | |
| Vapour Pressure | 0.2±0.4 mmHg at 25°C | |
| Index of Refraction | 1.443 | |
| LogP | 1.68 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 12 | |
| Complexity | 164 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | CCOC(=O)/C=C\C(=O)OCC |
|
| InChi Key | IEPRKVQEAMIZSS-WAYWQWQTSA-N | |
| InChi Code | InChI=1S/C8H12O4/c1-3-11-7(9)5-6-8(10)12-4-2/h5-6H,3-4H2,1-2H3/b6-5- | |
| Chemical Name | 2-Butenedioic acid (2Z)-, diethyl ester | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
|
||
| ln Vivo |
|
||
| Animal Protocol |
|
||
| References | Chem Biol Interact.2014 Aug 5;219:37-47;Biochem Pharmacol.1992 Feb 4;43(3):451-6. | ||
| Additional Infomation |
Diethyl maleate is a maleate ester resulting from the formal condensation of both carboxy groups of maleic acid with ethanol. A colourless liquid at room temperature (m.p. -10℃) with boiling point 220℃ at 1 atm., it is commonly used as a dienophile for Diels-Alder-type cycloaddition reactions in organic synthesis. It has a role as a glutathione depleting agent. It is a maleate ester and an ethyl ester. It is functionally related to an ethanol. See also: 2-Butenedioic acid (2Z)-, di-C8-15-alkyl esters (annotation moved to). |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 9.09 mg/mL (52.79 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 90.9 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.8079 mL | 29.0394 mL | 58.0788 mL | |
| 5 mM | 1.1616 mL | 5.8079 mL | 11.6158 mL | |
| 10 mM | 0.5808 mL | 2.9039 mL | 5.8079 mL |