Physicochemical Properties
| Molecular Formula | C15H13NO2 |
| Molecular Weight | 239.2692 |
| Exact Mass | 239.094 |
| CAS # | 1428-67-7 |
| Related CAS # | (R)-DPN;524047-78-7 |
| PubChem CID | 102614 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 449.4±35.0 °C at 760 mmHg |
| Melting Point | 203°C |
| Flash Point | 225.6±25.9 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.640 |
| LogP | 2.06 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 18 |
| Complexity | 293 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | GHZHWDWADLAOIQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C15H13NO2/c16-10-13(12-3-7-15(18)8-4-12)9-11-1-5-14(17)6-2-11/h1-8,13,17-18H,9H2 |
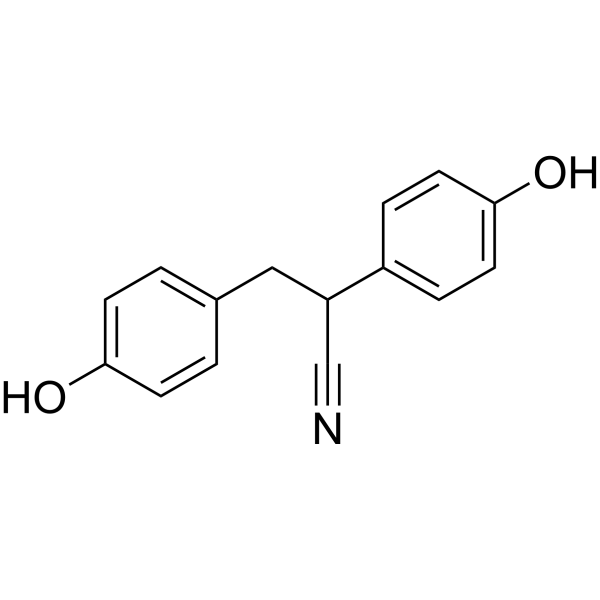
| Chemical Name | 2,3-bis(4-hydroxyphenyl)propanenitrile |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Estrogen receptor alpha (ERα) (Relative Binding Affinity (RBA) = 0.25% compared to estradiol) Estrogen receptor beta (ERβ) (RBA = 18% compared to estradiol; β/α selectivity = 72-fold; Transcriptional EC50 for ERβ = 0.85 nM, for ERα = 66 nM; β/α potency selectivity = 78-fold) [1] |
| ln Vitro |
DPN is a full ERα agonist with 78-fold selectivity for ERα potency (EC50 for ERβ=0.85 nM; EC50 for ERα=66 nM) and a 70-fold relative binding affinity selectivity for ERα [1]. In cultured cortical neurons, Aβ1-42 (10 μM)-induced toxicity is prevented from causing morphological changes by DPN (10 nM) [2]. ROS levels are lowered by DPN (0.1–100 nM) in a non-dose-responsive way [2]. In a dose-independent manner, DPN (0.1-100 nM) significantly reduces Aβ1-42-stimulated Bax expression [2]. In cultured cortical neurons, DPN (0.1-100 nM) lowers activated IL-1 levels induced by Aβ1-42 treatment [2]. Aβ1-42-upregulated JNK and p38 phosphorylation are inhibited by DPN (0.1-100 nM) [2]. DPN acts as a full agonist on both ERα and ERβ but shows strong selectivity for ERβ, with a 70-fold higher relative binding affinity and 170-fold higher relative potency in transcription assays compared to ERα. It demonstrates higher affinity and potency for ERβ than the phytoestrogen genistein. [1] |
| ln Vivo | In an 11-day period, DPN (10 μg) administered subcutaneously improved swimming capacity, decreased FST immobility, and elevated TPH protein expression in the dorsal raphe nucleus (DR) in a rat model [3]. |
| Enzyme Assay |
A competitive radiometric binding assay was used to determine the binding affinities of DPN for purified, recombinant full-length human ERα and ERβ. The assay was performed using 10 nM [³H]estradiol as the tracer. Incubations were carried out at 0°C for 18–24 hours, and hydroxyapatite was used to absorb the purified receptor-ligand complexes. Binding affinities are expressed as relative binding affinity (RBA) values, with the RBA of estradiol set at 100%. [1] |
| Cell Assay |
Transcriptional activation was assessed in human endometrial cancer (HEC-1) cells transfected with expression plasmids for ERα and ERβ and an estrogen-responsive reporter gene (chloramphenicol acetyltransferase (CAT) or luciferase (Luc)). For the CAT assay, cells in 60 mm dishes were transfected with a calcium phosphate precipitate containing the reporter plasmid, ER expression vector, and internal control (pCMVβGal). After 24 hours of ligand treatment, CAT activity was measured and normalized to β-galactosidase activity. For the Luc assay, HEC-1 cells in 24-well plates were transfected using lipofectin-transferrin with the reporter plasmid, ER expression vector, and internal control (pRL-CMV). After 6 hours, the medium was replaced with media containing ligands, and incubation continued for 24 hours. Luciferase activity was measured using a dual-luciferase reporter assay system, normalized to transfection efficiency, and expressed as a percentage of the activity induced by 10⁻⁸ M estradiol. [1] |
| Animal Protocol |
Animal/Disease Models: Adult SD (SD (Sprague-Dawley)) female rat (220-250 g), ovariectomized animal model [3] Doses: 10 μg/rat Route of Administration: daily subcutaneous injection for 11 days Experimental Results: increased number of swimming times in FST, diminished immobility. |
| References |
[1]. Estrogen Receptor-β Potency-Selective Ligands: Structure−Activity Relationship Studies of Diarylpropionitriles and Their Acetylene and Polar Analogues. Journal of Medicinal Chemistry, 2001. 44(24), 4230–4251. [2]. Neuroprotective effects of diarylpropionitrile against β-amyloid peptide-induced neurotoxicity in rat cultured cortical neurons. Neurosci Lett. 2014 Aug 22;578:44-9. [3]. Physiological dosages of estradiol and diarylpropionitrile decrease depressive behavior and increase tryptophan hydroxylase expression in the dorsal raphe nucleus of rats subjected to the forced swim test. Neuroreport. 2019 Jan 16;30(2):66-70. [4]. Effects of long-term dietary administration of estrogen receptor-beta agonist diarylpropionitrile on ovariectomized female ICR (CD-1) mice. GeroScience. 2018 Aug; 40(4): 393–403. |
| Additional Infomation |
2,3-bis(4-hydroxyphenyl)propionitrile is a nitrile that is acetonitrile in which one of the hydrogens is replaced by a 4-hydroxyphenyl group while a second hydrogen is replaced by a 4-hydroxybenzyl group. It is a specific agonist for estrogen receptor beta (ERbeta). It has a role as an estrogen receptor agonist. It is a member of phenols and a nitrile. DPN (2,3-bis(4-hydroxyphenyl)propionitrile) is a diarylpropionitrile identified as a potent and selective ERβ agonist. Its nitrile functionality is critical for ERβ selectivity, providing an optimal combination of linear geometry and polarity. Structural modifications, such as adding a second nitrile group or an ortho-methyl group on the β-aromatic ring, can enhance affinity and selectivity for ERβ. DPN and its analogues are useful tools for studying the structural and functional differences between ERα and ERβ. [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~417.94 mM) H2O : ~0.67 mg/mL (~2.80 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (10.45 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (10.45 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (10.45 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.1794 mL | 20.8969 mL | 41.7938 mL | |
| 5 mM | 0.8359 mL | 4.1794 mL | 8.3588 mL | |
| 10 mM | 0.4179 mL | 2.0897 mL | 4.1794 mL |