Physicochemical Properties
| Molecular Formula | C24H24FNO3 |
| Molecular Weight | 393.45067024231 |
| Exact Mass | 393.174 |
| Elemental Analysis | C, 73.26; H, 6.15; F, 4.83; N, 3.56; O, 12.20 |
| CAS # | 405225-21-0 |
| Related CAS # | 405225-21-0;571147-18-7; |
| PubChem CID | 5311283 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 5.019 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 29 |
| Complexity | 481 |
| Defined Atom Stereocenter Count | 0 |
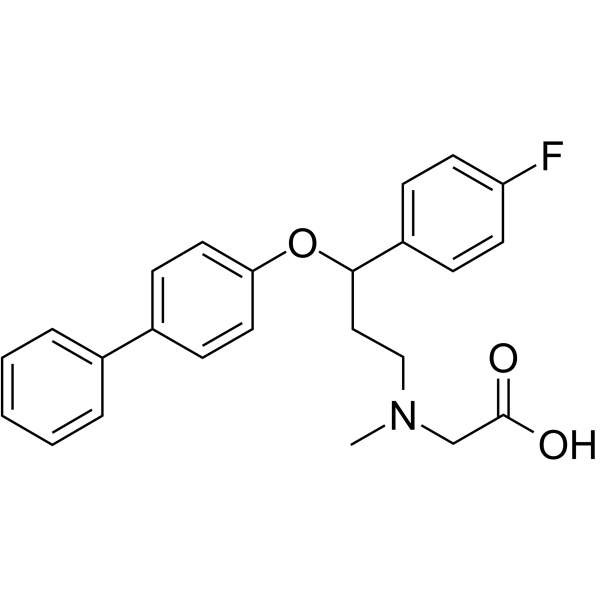
| SMILES | CN(CC(O)=O)CCC(C1=CC=C(F)C=C1)OC1=CC=C(C2=CC=CC=C2)C=C1 |
| InChi Key | FDORQEIHOKEJNX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H24FNO3/c1-26(17-24(27)28)16-15-23(20-7-11-21(25)12-8-20)29-22-13-9-19(10-14-22)18-5-3-2-4-6-18/h2-14,23H,15-17H2,1H3,(H,27,28) |
| Chemical Name | 2-[[3-(4-fluorophenyl)-3-(4-phenylphenoxy)propyl]-methylamino]acetic acid |
| Synonyms | NFPS; 405225-21-0; 2-[[3-(4-fluorophenyl)-3-(4-phenylphenoxy)propyl]-methylamino]acetic acid; CHEMBL26512; 571147-18-7; N-[3-(4'-fluorophenyl)-3-(4'-phenylphenoxy)propyl]sarcosine; [3H]NFPS; [3H]-NFPS; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | ALX-5407 (NFPS) selectively inhibits glycine transporter type 1 (GlyT1) with a binding affinity Kd =7.1±1.3 nMKd=7.1±1.3nM in rat forebrain membranes and Kd =21±3 nMKd=21±3nM for recombinant rat GlyT1a. It shows no activity against GlyT2 (IC50>100μM)[1] |
| ln Vitro |
[³H]NFPS binds rapidly to rat forebrain membranes (\( t_{1/2} = 13 \pm 4 \, \text{min} \)) and dissociates slowly (\( t_{1/2} = 28 \pm 5 \, \text{min} \)). Saturation analysis reveals a single binding site (\( B_{\text{max}} = 3.14 \pm 0.26 \, \text{pmol/mg protein} \)). Glycine and sarcosine weakly displace bound [³H]NFPS (\( IC_{50} = 1008 \, \mu\text{M} \) and \( 190 \, \mu\text{M} \), respectively), acting as non-competitive antagonists[1] Functional uptake assays in HEK-293 cells expressing GlyT1a show ALX-5407 potently inhibits [¹⁴C]glycine uptake (\( IC_{50} = 9.8 \pm 0.1 \, \text{nM} \)). Kinetic studies confirm non-competitive inhibition, reducing \( V_{\text{max}} \) without altering \( K_m \) for glycine, Na⁺, or Cl⁻[1] Regional binding density varies: cerebellum (\( 12.5 \pm 1.2 \, \text{pmol/mg protein} \)), striatum (\( 7.1 \pm 1.6 \)), cortex (\( 5.9 \pm 0.7 \)), hippocampus (\( 4.5 \pm 1.4 \))[1] |
| ln Vivo |
ALX-5407 increases extracellular glycine concentration by 40% in rat prefrontal cortex after systemic administration, enhancing NMDA receptor activity[1] In rat dentate gyrus, it potentiates NMDA receptor-dependent long-term potentiation (LTP), supporting its role in modulating excitatory neurotransmission[1] |
| Enzyme Assay |
Radioligand binding assays: Rat forebrain membranes (50 µl) incubated with [³H]NFPS (0.25–65 nM) in TB1 buffer (120 mM NaCl, 2 mM KCl, 1 mM CaCl₂, 1 mM MgCl₂, 10 mM HEPES, pH 7.5). Non-specific binding defined with 10 µM unlabeled NFPS. Filtration through GF/B filters after 30-min incubation, followed by scintillation counting[1] |
| Cell Assay |
Glycine uptake assays: JAR cells or GlyT1-expressing HEK-293 cells incubated with [¹⁴C]glycine (10 µM) in TB1A buffer ± inhibitors. Uptake terminated by rapid filtration after 3 h; radioactivity quantified via scintillation[1] Ion dependency tests: Na⁺/Cl⁻ replaced with Li⁺/acetate to assess ion binding site interactions[1] |
| Animal Protocol |
In vivo glycine modulation: Rats administered ALX-5407 systemically (dose unspecified). Extracellular glycine in prefrontal cortex measured via microdialysis[1] LTP studies: Drug delivered to evaluate NMDA receptor potentiation in dentate gyrus. Synaptic responses recorded electrophysiologically[1] |
| ADME/Pharmacokinetics | Systemic administration elevates cortical glycine by 40%, indicating blood-brain barrier penetration[1] |
| References |
[1]. Pharmacology and expression analysis of glycine transporter GlyT1 with [3H]-(N-[3-(4'-fluorophenyl)-3-(4'phenylphenoxy)propyl])sarcosine. Neuropharmacology. 2003 Oct;45(5):585-93. [2]. GlyT1 Inhibitor NFPS Exerts Neuroprotection via GlyR Alpha1 Subunit in the Rat Model of Transient Focal Cerebral Ischaemia and Reperfusion. Cell Physiol Biochem. 2016;38(5):1952-62. |
| Additional Infomation |
strong>ALX-5407 is a non-competitive inhibitor binding to an extracellular site on GlyT1 distinct from glycine/ion translocation sites. It modulates NMDA receptors by elevating synaptic glycine, with implications for treating schizophrenia (improves negative/cognitive symptoms)[1] Its racemic form was tritiated ([³H]NFPS; 19 Ci/mmol) for binding studies. The (+) enantiomer shows higher potency (\( IC_{50} = 0.67 \pm 0.1 \, \text{nM} \)) than (−) enantiomer (\( IC_{50} = 30 \pm 11 \, \text{nM} \))[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5416 mL | 12.7081 mL | 25.4162 mL | |
| 5 mM | 0.5083 mL | 2.5416 mL | 5.0832 mL | |
| 10 mM | 0.2542 mL | 1.2708 mL | 2.5416 mL |