Physicochemical Properties
| Molecular Formula | C23H15CL4FN2O3S2 |
| Molecular Weight | 592.3 |
| Exact Mass | 589.926 |
| CAS # | 861238-35-9 |
| PubChem CID | 11296282 |
| Appearance | Light yellow to green yellow solid powder |
| LogP | 9.251 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 35 |
| Complexity | 908 |
| Defined Atom Stereocenter Count | 0 |
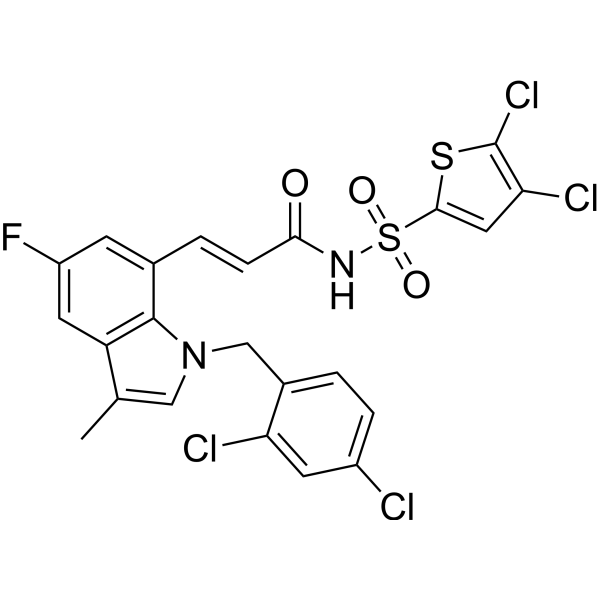
| SMILES | CC1=CN(C2=C(C=C(C=C12)F)/C=C/C(=O)NS(=O)(=O)C3=CC(=C(S3)Cl)Cl)CC4=C(C=C(C=C4)Cl)Cl |
| InChi Key | BFBTVZNKWXWKNZ-HWKANZROSA-N |
| InChi Code | InChI=1S/C23H15Cl4FN2O3S2/c1-12-10-30(11-14-2-4-15(24)7-18(14)25)22-13(6-16(28)8-17(12)22)3-5-20(31)29-35(32,33)21-9-19(26)23(27)34-21/h2-10H,11H2,1H3,(H,29,31)/b5-3+ |
| Chemical Name | (E)-3-[1-[(2,4-dichlorophenyl)methyl]-5-fluoro-3-methylindol-7-yl]-N-(4,5-dichlorothiophen-2-yl)sulfonylprop-2-enamide |
| Synonyms | DG041; DG 041; DG-041 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | TP pins (IC50=742 nM), EP1 (IC50=486 nM), and DP1 (IC50=131 nM) make up DG-041[1]. |
| ln Vivo | DG-041, administered intravenously at 1.78 mg/kg or bucally at 9.62 mg/kg, had a t1/2 of 2.7 hours and a Cmax of 2.74 μM and 9.46 μM, respectively. 1250 mL/h/kg is the intravenous injection CL [ |
| Animal Protocol |
Animal/Disease Models: Male Sprague Dawley rat [1] Doses: 1.78 mg/kg (intravenous) or 9.62 mg/kg (oral) Route of Administration: intravenous (iv) (iv)or oral Experimental Results: t1/2 is 2.7 hrs (hrs (hours)), 4.06 hrs (hrs (hours)) and Cmax is 9.46 1]. intravenous (iv) (iv)injection and oral administration are μM and 2.74 μM respectively. |
| References |
[1]. Antagonists of the EP3 receptor for prostaglandin E2 are novel antiplatelet agents that do not prolong bleeding. ACS Chem Biol. 2009 Feb 20;4(2):115-26. [2]. Heterocyclic 1,7-disubstituted indole sulfonamides are potent and selective human EP3 receptorantagonists. Bioorg Med Chem Lett. 2009 Dec 1;19(23):6797-800. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~422.07 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.51 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6883 mL | 8.4417 mL | 16.8833 mL | |
| 5 mM | 0.3377 mL | 1.6883 mL | 3.3767 mL | |
| 10 mM | 0.1688 mL | 0.8442 mL | 1.6883 mL |