Physicochemical Properties
| Molecular Formula | C48H73N13O17S3 |
| Molecular Weight | 1200.36512732506 |
| Exact Mass | 1199.44 |
| CAS # | 1203586-72-4 |
| Related CAS # | Cyclotraxin B TFA |
| PubChem CID | 90489002 |
| Appearance | White to off-white solid powder |
| LogP | -9 |
| Hydrogen Bond Donor Count | 16 |
| Hydrogen Bond Acceptor Count | 22 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 81 |
| Complexity | 2230 |
| Defined Atom Stereocenter Count | 10 |
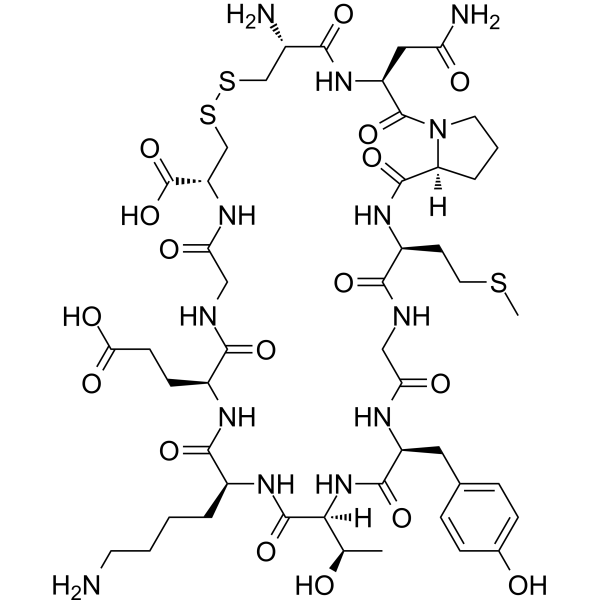
| SMILES | S(C)CC[C@H]1C(NCC(N[C@H](C(N[C@@H]([C@@H](C)O)C(N[C@H](C(N[C@H](C(NCC(N[C@H](C(=O)O)CSSC[C@@H](C(N[C@@H](CC(N)=O)C(N2CCC[C@H]2C(N1)=O)=O)=O)N)=O)=O)CCC(=O)O)=O)CCCCN)=O)=O)CC1C=CC(=CC=1)O)=O)=O |
| InChi Key | JLBMMJHZUYBFGX-ZHTCEXBHSA-N |
| InChi Code | InChI=1S/C48H73N13O17S3/c1-24(62)39-46(75)58-28(6-3-4-15-49)43(72)56-29(12-13-38(67)68)41(70)52-21-37(66)55-33(48(77)78)23-81-80-22-27(50)40(69)59-32(19-35(51)64)47(76)61-16-5-7-34(61)45(74)57-30(14-17-79-2)42(71)53-20-36(65)54-31(44(73)60-39)18-25-8-10-26(63)11-9-25/h8-11,24,27-34,39,62-63H,3-7,12-23,49-50H2,1-2H3,(H2,51,64)(H,52,70)(H,53,71)(H,54,65)(H,55,66)(H,56,72)(H,57,74)(H,58,75)(H,59,69)(H,60,73)(H,67,68)(H,77,78)/t24-,27+,28+,29+,30+,31+,32+,33+,34+,39+/m1/s1 |
| Chemical Name | (3S,6R,11R,17S,20S,23S,26S,32S,35S)-6-amino-20-(4-aminobutyl)-3-(2-amino-2-oxoethyl)-17-(2-carboxyethyl)-23-[(1R)-1-hydroxyethyl]-26-[(4-hydroxyphenyl)methyl]-32-(2-methylsulfanylethyl)-2,5,13,16,19,22,25,28,31,34-decaoxo-8,9-dithia-1,4,12,15,18,21,24,27,30,33-decazabicyclo[33.3.0]octatriacontane-11-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | TrkB |
| References |
[1]. Cyclotraxin-B, the first highly potent and selective TrkB inhibitor, has anxiolytic properties in mice. PLoS One. 2010 Mar 19;5(3):e9777. [2]. Cyclotraxin-B, a new TrkB antagonist, and glial blockade by propentofylline, equally prevent and reverse cold allodynia induced by BDNF or partial infraorbital nerve constriction in mice. J Pain. 2012 Jun;13(6):579-89. [3]. Systemic Delivery of a Brain-Penetrant TrkB Antagonist Reduces Cocaine Self-Administration and Normalizes TrkB Signaling in the Nucleus Accumbens and Prefrontal Cortex. J Neurosci. 2016 Aug 3;36(31):8149-59. |
Solubility Data
| Solubility (In Vitro) | H2O: 100 mg/mL (83.31 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 100 mg/mL (83.31 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8331 mL | 4.1654 mL | 8.3308 mL | |
| 5 mM | 0.1666 mL | 0.8331 mL | 1.6662 mL | |
| 10 mM | 0.0833 mL | 0.4165 mL | 0.8331 mL |