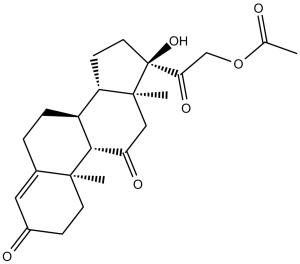
Cortisone acetate (NSC49420; NSC-49420; Cortone acetate; Cortisyl; Incortin), a steroid hormone, is an acetate ester prodrug form of cortisone with glucocoriticoid and mineral corticoid activities. It has been used for the treatment for a number of different diseases, such as severe allergies or skin problems, asthma, or arthritis.
Physicochemical Properties
| Molecular Formula | C23H30O6 | |
| Molecular Weight | 402.48 | |
| Exact Mass | 402.204 | |
| Elemental Analysis | C, 68.64; H, 7.51; O, 23.85 | |
| CAS # | 50-04-4 | |
| Related CAS # | Cortisone;53-06-5 | |
| PubChem CID | 5745 | |
| Appearance | Typically exists as white to off-white solids at room temperature | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 577.3±50.0 °C at 760 mmHg | |
| Melting Point | 237-240 °C(lit.) | |
| Flash Point | 197.3±23.6 °C | |
| Vapour Pressure | 0.0±3.6 mmHg at 25°C | |
| Index of Refraction | 1.566 | |
| LogP | 2.53 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 4 | |
| Heavy Atom Count | 29 | |
| Complexity | 827 | |
| Defined Atom Stereocenter Count | 6 | |
| SMILES | O([H])[C@]1(C(C([H])([H])OC(C([H])([H])[H])=O)=O)C([H])([H])C([H])([H])[C@@]2([H])[C@]3([H])C([H])([H])C([H])([H])C4=C([H])C(C([H])([H])C([H])([H])[C@]4(C([H])([H])[H])[C@@]3([H])C(C([H])([H])[C@@]21C([H])([H])[H])=O)=O |
|
| InChi Key | ITRJWOMZKQRYTA-RFZYENFJSA-N | |
| InChi Code | InChI=1S/C23H30O6/c1-13(24)29-12-19(27)23(28)9-7-17-16-5-4-14-10-15(25)6-8-21(14,2)20(16)18(26)11-22(17,23)3/h10,16-17,20,28H,4-9,11-12H2,1-3H3/t16-,17-,20+,21-,22-,23-/m0/s1 | |
| Chemical Name | 2-((8S,9S,10R,13S,14S,17R)-17-hydroxy-10,13-dimethyl-3,11-dioxo-2,3,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl)-2-oxoethyl acetate | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Glucocorticoid Receptor (GR)[1][2][4] |
| ln Vitro |
In peripheral blood mononuclear cells (PBMC), cortisone (2.8-28,000 nM) dose-dependently reduces cortisol-induced apoptosis [1]. In human peripheral-blood mononuclear cells (PBMCs), cortisol (1 μM) induced apoptosis with an apoptosis rate of 35% (Annexin V/PI staining). Cortisone acetate (10 nM, 100 nM, 1 μM, 10 μM) dose-dependently counteracted this apoptosis-inducing effect. At 100 nM, it reduced the apoptosis rate to 18%; at 1 μM, the apoptosis rate was further decreased to 12%, restoring cell viability to ~90% (MTT assay)[1] |
| ln Vivo |
In rabbits, cortisone (2 mg/kg; intramuscular every other day for two months) decreases tuberculin responses and BCG (Mycobacterium tuberculosis vaccine strain) lesions [2]. In a rodent model of tuberculous lesions, intraperitoneal administration of Cortisone acetate (5 mg/kg daily for 2 weeks) reduced macrophage accumulation in lesions by 40% (histopathological counting). It inhibited macrophage activation, as evidenced by a 35% decrease in TNF-α secretion (ELISA) and reduced phagocytic activity (neutral red uptake assay), and decreased macrophage necrosis rate by 28% compared to vehicle control[2] - In 4-week-old chickens, subcutaneous injection of Cortisone acetate (2 mg/kg twice weekly for 3 weeks) modulated the B cell line. It reduced peripheral blood B cell numbers by 30% (flow cytometry) and inhibited LPS-induced B cell proliferation, with a 45% decrease in proliferative index (thymidine incorporation assay). No significant effect on B cell differentiation was observed[4] |
| Cell Assay |
Glucocorticoids (GCs) have been considered to regulate immune cell systems through induction of apoptosis in thymocytes and mature peripheral-blood lymphocytes. Here we report that apoptosis induced by cortisol in mitogen-activated peripheral-blood mononuclear cells (PBMC) is suppressed by cortisone, an oxidized metabolite of cortisol. Apoptosis in PBMCs is quantified by a cell death ELISA procedure, which can specifically detect fragmented DNA. Cortisol induced PBMC-apoptosis at concentrations more than 10 ng/ml (28 nM) in concanavalin A-stimulated PBMCs and cortisone suppressed this apoptosis at a concentration range of 1-10,000 ng/ml (2.8-28,000 nM) dose-dependently. Prednisone, a synthetic oxidized-GC, also suppressed the apoptosis-inducing effect of cortisol in a dose-dependent manner. Suppression of cortisol-induced apoptosis by cortisone was consistently observed in PBMCs derived from 16 healthy subjects. Examination for inhibitory activities of the steroids against [3H]dexamethasone binding to PBMCs suggested that cortisone can bind cellular GC-receptors (GC-Rs), but the affinity of cortisone to GCRs is 1/30 or less than that of cortisol. The results raised a possible role of cortisone in cortisol-mediated regulation of apoptosis in activated human PBMCs. The counteracting action of cortisone against cortisol-induced apoptosis may take place partially through intervention of GC-receptors (GC-Rs), but may also be due to unknown pathway(s) different from those mediated by cellular GC-Rs[1]. PBMC apoptosis counteraction assay: Human peripheral-blood mononuclear cells (PBMCs) were isolated and seeded in 96-well plates. Cortisone acetate (10 nM, 100 nM, 1 μM, 10 μM) was added for 1-hour pre-treatment, followed by incubation with cortisol (1 μM) for 48 hours. Apoptosis was detected by Annexin V-FITC/PI staining and flow cytometry. MTT assay was performed to assess cell viability and rule out non-specific cytotoxicity of Cortisone acetate alone[1] |
| Animal Protocol |
Animal/Disease Models: Male New Zealand white rabbits (2.1-2.4 kg) were injected with BCG six days after the first dose [2] Doses: 2 mg/kg Route of Administration: intramuscularinjection every other day for 2 months Experimental Results: BCG lesions and tuberculosis bacteria were diminished factor reaction. diminished the number of infiltrating mononuclear cells (MN), the number of caseous necrosis and ulcers, and the percentage of β-galactosidase-positive NMs. Tuberculous lesion rodent model: Rodents were infected with Mycobacterium tuberculosis to induce tuberculous lesions. One week post-infection, Cortisone acetate was administered via intraperitoneal injection at 5 mg/kg once daily for 2 weeks; vehicle control received equal volume of normal saline. Animals were euthanized after treatment, and tuberculous lesions were collected for histopathological macrophage counting, TNF-α quantification (ELISA), and phagocytic activity assay (neutral red uptake)[2] - Chicken B cell modulation model: 4-week-old chickens were randomly grouped. Cortisone acetate was injected subcutaneously at 2 mg/kg twice weekly for 3 weeks; control chickens received saline. Peripheral blood was collected weekly to isolate B cells. Flow cytometry counted B cell numbers, and thymidine incorporation assay measured LPS-induced B cell proliferation[4] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Corticosteroids are eliminated predominantly in the urine. Data regarding the clearance of cortisone acetate is not readily available. |
| Toxicity/Toxicokinetics |
Protein Binding Corticosteroids are generally bound to corticosteroid binding globulin and serum albumin in plasma. |
| References |
[1]. Cortisone counteracts apoptosis-inducing effect of cortisol in human peripheral-blood mononuclear cells. Int Immunopharmacol. 2001 Nov;1(12):2109-15. [2]. The effect of cortisone on the accumulation, activation, and necrosis of macrophages in tuberculous lesions. Inflammation. 1978 Jun;3(2):159-76. [3]. In Vivo Antifungal Activity of Monolaurin against Candida albicans Biofilms. Biol Pharm Bull. 2018;41(8):1299-1302. [4]. In vivo effects of cortisone on the B cell line in chickens. J Immunol. 1975 Nov;115(5):1370-4. |
| Additional Infomation |
Cortisone acetate is a corticosteroid hormone. Cortisone acetate was first isolate in 1935 and became more widely researched in 1949. Since then, glucocorticoids such as cortisone acetate have been used to treat a number of inflammatory conditions such as endocrine, rheumatic, collagen, dermatologic, allergic, ophthalmic, respiratory, hematologic, neoplastic, edematous, and gastrointestinal diseases and disorders. Cortisone acetate was granted FDA approval on 13 June 1950. Cortisone Acetate is the acetate salt form of cortisone, a synthetic or semisynthetic analog of the naturally occurring cortisone hormone produced by the adrenal glands with anti-inflammatory and immunomodulating properties. Cortisone acetate diffuses through the cell membrane and binds to nuclear glucocorticoid receptors. The receptor-ligand complex binds to promotor regions of certain genes and initiates RNA transcription. This results in an induction of synthesis of certain anti-inflammatory proteins while inhibiting the synthesis of certain inflammatory mediators. A naturally occurring glucocorticoid that has been used in replacement therapy for ADRENAL INSUFFICIENCY and as an anti-inflammatory agent. Cortisone itself is inactive; it is converted in the liver to the active metabolite HYDROCORTISONE. (From Martindale, The Extra Pharmacopoeia, 30th ed, p726) See also: Cortisone (has active moiety) ... View More ... Drug Indication Cortisone acetate is indicated to treat a wide variety of endocrine, rheumatic, collagen, dermatologic, allergic, ophthalmic, respiratory, hematologic, neoplastic, edematous, and gastrointestinal diseases and disorders. Mechanism of Action The short term effects of corticosteroids are decreased vasodilation and permeability of capillaries, as well as decreased leukocyte migration to sites of inflammation. Corticosteroids binding to the glucocorticoid receptor mediates changes in gene expression that lead to multiple downstream effects over hours to days. Glucocorticoids inhibit neutrophil apoptosis and demargination; they inhibit phospholipase A2, which decreases the formation of arachidonic acid derivatives; they inhibit NF-Kappa B and other inflammatory transcription factors; they promote anti-inflammatory genes like interleukin-10. Lower doses of corticosteroids provide an anti-inflammatory effect, while higher doses are immunosuppressive. High doses of glucocorticoids for an extended period bind to the mineralocorticoid receptor, raising sodium levels and decreasing potassium levels. Cortisone acetate is a synthetic glucocorticoid prodrug that exerts biological effects after metabolic activation to cortisone[1][2][4] - Its core mechanism involves binding to the glucocorticoid receptor (GR) to regulate immune cell function, including counteracting PBMC apoptosis, modulating macrophage accumulation and activation, and suppressing B cell proliferation[1][2][4] - It exhibits immunomodulatory and anti-inflammatory activities, targeting multiple immune cell populations (PBMCs, macrophages, B cells)[1][2][4] - It is clinically relevant for managing inflammatory and immune-related disorders, leveraging its ability to regulate in vivo immune responses[2][4] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.5 mg/mL (1.24 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.5 mg/mL (1.24 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 0.5 mg/mL (1.24 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 25 mg/mL (62.11 mM) in 0.1% Tween-80 in PBS (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4846 mL | 12.4230 mL | 24.8460 mL | |
| 5 mM | 0.4969 mL | 2.4846 mL | 4.9692 mL | |
| 10 mM | 0.2485 mL | 1.2423 mL | 2.4846 mL |