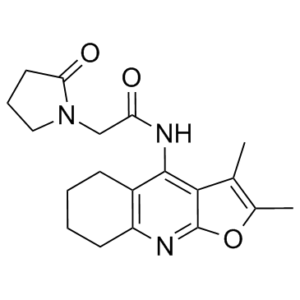
Coluracetam (also known as MKC-231 and BCI-540) is a novel choline uptake enhancer. MKC-231(10(-10)-10(-6) moll) significantly increased high affinity choline uptake (HACU) when it was incubated with the hippocampal synaptosomes of ethylcholine mustard aziridinium ion (AF64A) treated rats, but not of normal rats. MKC-231 did not affect the AChE activity, [3H]- quinuclidinyl benzilate binding, and [3H]-pirenzepine binding. Oral administration of MKC-231 (1-10 mg/kg) significantly improved the learning deficits in the Morris' water maze of AF64A-treated rats, but it did not produce any significant side effects, like tremor, salivation or hypothermia, which were observed in rats treated with high doses of tacrine.
Physicochemical Properties
| Molecular Formula | C19H23N3O3 | |
| Molecular Weight | 341.41 | |
| Exact Mass | 341.173 | |
| CAS # | 135463-81-9 | |
| Related CAS # |
|
|
| PubChem CID | 214346 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 634.1±55.0 °C at 760 mmHg | |
| Melting Point | 225 °C | |
| Flash Point | 337.3±31.5 °C | |
| Vapour Pressure | 0.0±1.9 mmHg at 25°C | |
| Index of Refraction | 1.641 | |
| LogP | 3.27 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 25 | |
| Complexity | 540 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | PSPGQHXMUKWNDI-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C19H23N3O3/c1-11-12(2)25-19-17(11)18(13-6-3-4-7-14(13)20-19)21-15(23)10-22-9-5-8-16(22)24/h3-10H2,1-2H3,(H,20,21,23) | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
A referenced neurochemical study (Bessho et al., 1994, cited within [1]) demonstrated that MKC-231 increases high-affinity choline uptake (HACU) in vitro in the hippocampus of AF64A-treated rats. [1] |
| ln Vivo |
MKC-231(10(-10)-10(-6) moll) significantly increased high affinity choline uptake (HACU) when it was incubated with the hippocampal synaptosomes of ethylcholine mustard aziridinium ion (AF64A) treated rats, but not of normal rats. MKC-231 did not affect the AChE activity, [3H]- quinuclidinyl benzilate binding, and [3H]-pirenzepine binding. Oral administration of MKC-231 (1-10 mg/kg) significantly improved the learning deficits in the Morris' water maze of AF64A-treated rats, but it did not produce any significant side effects, like tremor, salivation or hypothermia, which were observed in rats treated with high doses of tacrine.[1] Chronic oral administration of MKC-231 (0.3, 1.0, or 3.0 mg/kg, once daily for 11 days) significantly improved working memory deficits induced by ethylcholine aziridinium ion (AF64A) in mice, as assessed by a delayed non-matching to sample task in a T-maze. The correct response rates improved from 61.1% (AF64A+vehicle) to 86.1%, 85.0%, and 83.3% at doses of 0.3, 1.0, and 3.0 mg/kg, respectively. Acute oral administration of MKC-231 (0.3, 1.0, or 3.0 mg/kg, single dose) did not significantly improve the AF64A-induced working memory deficit. [1] Chronic oral administration of MKC-231 (0.3 and 1.0 mg/kg, once daily for 12 days) significantly reversed the depletion of hippocampal ACh content induced by AF64A in mice. ACh levels increased from 60.8% of control (AF64A+vehicle) to 84.4% and 75.5% of control at doses of 0.3 and 1.0 mg/kg, respectively. The effect at 3.0 mg/kg (65.0% of control) was not significant, indicating a bell-shaped dose-response curve. Acute oral administration of MKC-231 did not reverse the AF64A-induced hippocampal ACh depletion. [1] A referenced neurochemical study (Bessho et al., 1994, cited within [1]) demonstrated that MKC-231 increases ACh release in vivo in the hippocampus of AF64A-treated rats. [1] |
| Animal Protocol |
Amnesia Model Induction: Male ddY mice (27-28 g) were anesthetized and infused bilaterally with ethylcholine aziridinium ion (AF64A; 1.75 nmol/2.0 µl per side) into the lateral cerebral ventricle using a stereotaxic apparatus and an infusion pump. Control (sham) mice received an equivalent volume of isotonic saline. [1] Behavioral Training: Four days after surgery, mice were habituated and trained for 4 days in a T-maze apparatus connected to their home cage, where they learned to navigate for food and water. [1] Acute Drug Treatment (Behavioral/Neurochemical): Mice were treated with a single oral dose of MKC-231 (suspended in 0.5% Tween 80 solution) at 0.3, 1.0, or 3.0 mg/kg. Behavioral testing in the T-maze task or brain collection for neurochemical analysis was performed 60 minutes after drug administration. [1] Chronic Drug Treatment (Behavioral): Mice were treated orally with MKC-231 (suspended in 0.5% Tween 80 solution) at 0.3, 1.0, or 3.0 mg/kg, once daily for 11 days. Behavioral testing (performance trials) was conducted twice, approximately 20-24 hours after the 10th and 11th administrations. [1] Chronic Drug Treatment (Neurochemical): Mice from the chronic behavioral study received a 12th administration of MKC-231 immediately after the second behavioral trial. They were killed for brain ACh assay the next day (approximately 24 hours after the final dose). [1] |
| References |
[1]. Effect of the novel high affinity choline uptake enhancer 2-(2-oxopyrrolidin-1-yl)-N-(2,3-dimethyl-5,6,7,8-tetrahydrofuro[2,3-b] quinolin-4-yl)acetoamide on deficits of water maze learning in rats. Arzneimittelforschung. 1996 Apr;46(4):369-73. [2]. MKC-231, a choline uptake enhancer, ameliorates working memory deficits and decreased hippocampal acetylcholine induced by ethylcholine aziridinium ion in mice. J Neural Transm Gen Sect. 1994;98(1):1-13. |
| Additional Infomation |
MKC-231 is a newly synthesized tetrahydrofuroquinoline analogue and a member of a novel class of nootropic agents. Its chemical name is [2-(2-oxypyrrolidin-1-yl)-N-(2,3-dimethyl-5,6,7,8-tetrahydrofuro[2,3-b]quinolin-4-yl)acetamide]. Its proposed mechanism of action is enhancing high-affinity choline uptake (HACU), which is rate-limiting for ACh synthesis, particularly under conditions of cholinergic deficit. This distinguishes it from acetylcholinesterase inhibitors or direct cholinergic agonists. The study suggests that the ameliorative effects of chronic MKC-231 on memory and hippocampal ACh levels, observed 20-24 hours after the last dose, may be due to a persistent repair of cholinergic function rather than a direct acute drug action. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9290 mL | 14.6451 mL | 29.2903 mL | |
| 5 mM | 0.5858 mL | 2.9290 mL | 5.8581 mL | |
| 10 mM | 0.2929 mL | 1.4645 mL | 2.9290 mL |