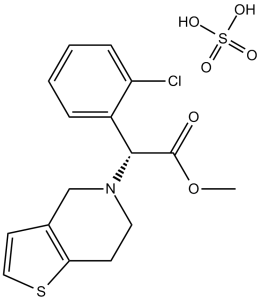
Clopidogrel sulfate (Plavix; Zyllt; Osvix; Plavitor, SR-25990C), the sulfate salt of Clopidogrel, is an orally bioavailable, thienopyridine class of antiplatelet agent acting as an irreversible inhibitor of ADP receptor (P2Y12). Clopidogrel is a prodrug that has to be activated by CYP2C19. It is approved as an antiplatelet and anticoagulant medication used to reduce the risk of heart disease and stroke in those at high risk. Clopidogrel works by irreversibly inhibiting a receptor called P2Y12, an adenosine diphosphate (ADP) chemoreceptor on platelet cell membranes. Clopidogrel acts by inhibiting the ADP receptor on platelet cell membranes.
Physicochemical Properties
| Molecular Formula | C16H16CLNO2S.H2SO4 | |
| Molecular Weight | 419.9 | |
| Exact Mass | 419.026 | |
| Elemental Analysis | C, 45.77; H, 4.32; Cl, 8.44; N, 3.34; O, 22.86; S, 15.27 | |
| CAS # | 120202-66-6 | |
| Related CAS # | Clopidogrel;113665-84-2;Clopidogrel thiolactone;1147350-75-1;Clopidogrel-d3 hydrogen sulfate;1217643-68-9; 90055-48-4 (racemic); 120202-66-6 (sulfate);120202-67-7 (HBr); 120202-65-5 (HCl); 744256-69-7 (besylate); | |
| PubChem CID | 115366 | |
| Appearance | Off-white to light yellow solid powder | |
| Boiling Point | 423.7ºC at 760 mmHg | |
| Melting Point | 184ºC | |
| Flash Point | 210ºC | |
| LogP | 3.965 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 4 | |
| Heavy Atom Count | 26 | |
| Complexity | 463 | |
| Defined Atom Stereocenter Count | 1 | |
| SMILES | COC(=O)[C@H](C1=CC=CC=C1Cl)N2CCC3=C(C2)C=CS3.OS(=O)(=O)O |
|
| InChi Key | FDEODCTUSIWGLK-RSAXXLAASA-N | |
| InChi Code | InChI=1S/C16H16ClNO2S.H2O4S/c1-20-16(19)15(12-4-2-3-5-13(12)17)18-8-6-14-11(10-18)7-9-21-14;1-5(2,3)4/h2-5,7,9,15H,6,8,10H2,1H3;(H2,1,2,3,4)/t15-;/m0./s1 | |
| Chemical Name | methyl (2S)-2-(2-chlorophenyl)-2-(6,7-dihydro-4H-thieno[3,2-c]pyridin-5-yl)acetate sulfate | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
CYP2B6 (IC50 = 18.2 nM); P2Y12 Receptor; CYP2C19 (IC50 = 524 nM) The target of Clopidogrel sulfate is the ADP receptor P2Y12[2] The targets of Clopidogrel sulfate include the ADP receptor P2Y2 [5] |
| ln Vitro |
According to real-time PCR and other methods, clopidogrel hydrogen sulfate (1.5 mM; 12 and 24 hours) increases the expression of TRIB3 and CHOP in a concentration- and time-dependent way. Gene microarray analysis identified 79 genes that were differentially expressed (P<0.05 and fold-change >3) when cells were treated with or without clopidogrel. Gene ontology analysis revealed that response to stress and cell apoptosis dysfunction were ranked in the top 10 cellular events being affected, and that the major components of endoplasmic reticulum stress-mediated apoptosis pathway - CHOP and TRIB3- were up-regulated in a concentration- and time-dependent manner when cells were treated with clopidogrel. Pathway analysis demonstrated that multiple MAPK kinases were phosphorylated in clopidogrel-treated GES-1 cells, but that only SB-203580 (a p38-specific MAPK inhibitor) attenuated cell apoptosis and CHOP over-expression, both of which were induced by clopidogrel. Conclusions: Increased endoplasmic reticulum stress response is involved in clopidogrel-induced gastric mucosal injury, acting through p38 MAPK activation.[3] In vitro experiments showed that Clopidogrel sulfate had no significant inhibitory effect on human cytochrome P450 enzymes (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4), with inhibition rates all below 50% (test concentration: 100 μmol/L) [1] - In in vitro culture experiments of human gastric epithelial cells (GES-1 cells), Clopidogrel sulfate induced cell apoptosis in a concentration-dependent manner, with a significant increase in apoptosis rate at 100 μmol/L. It also upregulated the expression of endoplasmic reticulum stress-related proteins (GRP78, CHOP, caspase-12) and activated the JNK signaling pathway [3] - In in vitro experiments of rabbit middle cerebral artery smooth muscle cells, Clopidogrel sulfate inhibited ADP-mediated P2Y2 receptor activation, thereby blocking the increase in intracellular Ca²⁺ concentration and suppressing vascular smooth muscle contraction [5] |
| ln Vivo |
When compared to a carrier, clopidogrel hydrogen sulfate (5 mg/kg) effectively prevented thrombosis. In mice, acetylsalicylic acid (0.15 mg/kg) can enhance clopidogrel's antithrombotic efficacy; in mice, acetylsalicylic acid (0.6 mg/kg) can reduce clopidogrel's antithrombotic effect [4]. In a mouse arterial thrombosis model, oral administration of Clopidogrel sulfate (10 mg/kg) significantly prolonged thrombosis time and reduced thrombus weight. When combined with different doses of aspirin (1, 3, 10 mg/kg), the antithrombotic activity showed a synergistic enhancement effect without a significant increase in bleeding risk [4] - In rabbit in vivo experiments, intravenous injection of Clopidogrel sulfate dose-dependently inhibited the contractile response of the middle cerebral artery to ADP induction, with a contraction inhibition rate of over 40% at 10 mg/kg [5] |
| Enzyme Assay |
Clopidogrel is a potent antithrombotic drug that inhibits ADP-induced platelet aggregation. The results of large clinical trials have demonstrated an overall benefit of clopidogrel over aspirin in the prevention of vascular ischemic events (myocardial infarction, stroke, vascular death) in patients with a history of symptomatic atherosclerotic disease. The antiaggregating effect of clopidogrel is attributed to an irreversible inhibition of ADP binding to a purinergic receptor present at the platelet surface. Clopidogrel is not active in vitro and can be considered a precursor of an active metabolite formed in the liver. The chemical structure of this active metabolite and its biological activity have been described recently. Several purinergic receptors have been described on platelets; P2X (1), a calcium channel, and P2Y1 a Gq-coupled seven-transmembrane domain receptor, have been found not to be antagonized by clopidogrel. Another Gi (2)-coupled receptor (named P2Y12) has been recently cloned and stably expressed in CHO cells. These cells displayed a strong affinity for (33)P-2MeS-ADP, a stable analogue of ADP, the binding characteristics of which corresponded in all points to those observed on platelets. The binding of (33)P-2MeS-ADP to these cells was strongly inhibited by the active metabolite of clopidogrel with a potency that was consistent with that observed for this compound on platelets. In these transfected CHO cells, as in platelets, ADP and 2MeS-ADP induced adenylyl cyclase downregulation, an effect that was inhibited by the active metabolite of clopidogrel. These results demonstrate that this receptor corresponds to the previously called "P2t" platelet receptor and show that the active metabolite of clopidogrel binds in a covalent manner to this receptor, thus explaining how it blocks the aggregating effect of ADP on platelets[2]. Cytochrome P450 enzyme inhibition assay: Human liver microsomes were prepared and co-incubated with Clopidogrel sulfate at different concentrations (0.1, 1, 10, 100 μmol/L) and specific substrates. The production of substrate metabolites was detected by high-performance liquid chromatography to calculate the enzyme activity inhibition rate [1] - P2Y2 receptor activity detection assay: Rabbit middle cerebral artery smooth muscle cells were isolated and loaded with Ca²⁺ fluorescent probes. After pretreatment with Clopidogrel sulfate, the cells were stimulated with ADP. The change in intracellular Ca²⁺ fluorescence intensity was detected by fluorescence microscopy to evaluate the inhibitory effect on receptor activation [5] |
| Cell Assay |
Western Blot Analysis[3] Cell Types: GES-1 cells Tested Concentrations: 1.5 mM Incubation Duration: 12 and 24 hrs (hours) Experimental Results: The mRNA expression levels of both CHOP and TRIB3 were up-regulated in a concentration- and time-dependent manner. The protein expression levels of both CHOP and TRIB3 were up-regulated in a concentration- and time-dependent manner. Apoptosis Analysis[3] Cell Types: Gastric epithelial cell (GES-1) cells Tested Concentrations: 1.5 mM Incubation Duration: 24 hrs (hours) Experimental Results: Induced apoptosis of gastric epithelial cells. The GES-1 cells were used as a model system, the effects of clopidogrel on the whole gene expression profile were evaluated by human gene expression microarray and gene ontology analysis, changes of the mRNA and protein expression were determined by real-time PCR and Western blot analysis, and cell viability and apoptosis were measured by MTT assay and flow cytometry analysis, respectively.[3] Gastric epithelial cell apoptosis assay: GES-1 cells were seeded in culture plates and cultured to the logarithmic growth phase. Different concentrations of Clopidogrel sulfate (10, 50, 100 μmol/L) were added, and the culture was continued for 24 hours. Flow cytometry was used to detect the cell apoptosis rate, and Western blot was used to detect the expression levels of endoplasmic reticulum stress-related proteins and JNK pathway proteins [3] - Vascular smooth muscle cell contraction-related assay: Rabbit middle cerebral artery smooth muscle cells were isolated, seeded, and treated with Clopidogrel sulfate for 12 hours. After stimulation with ADP, the cell contraction amplitude was detected by a cell contraction analyzer, and real-time quantitative PCR was used to detect the mRNA expression level of the P2Y2 receptor [5] |
| Animal Protocol |
Animal/Disease Models: Age-matched C57BL/6J male mice weighing at least 25 g were used at 8 to 12 weeks of age[4] Doses: 5 mg/kg Route of Administration: Gavage treatment Experimental Results: Clopidogrel Dramatically inhibited thrombus formation compared with vehicle. When Clopidogrel was given in combination with 0.6 mg/kg ASA, thrombi were Dramatically larger than those measured with Clopidogrel alone. In contrast, when Clopidogrel was given in combination with 0.15 mg/kg ASA, thrombi were Dramatically smaller than those in mice treated with Clopidogrel and 0.6 mg/kg ASA, and smaller than those in mice given Clopidogrel alone. The purpose of this study was to examine this possibility in vivo. Approach and Results- Mice were given oral acetylsalicylic acid at varying doses, oral clopidogrel (5 mg/kg body weight), or both. At doses of 0.15 and 0.6 mg/kg, acetylsalicylic acid inhibited arachidonic acid-induced platelet aggregation, but only 0.6 mg/kg acetylsalicylic acid, or higher, decreased the plasma levels of 6-keto-prostaglandin-F1α, the stable metabolite of prostacyclin. When given with clopidogrel, laser injury-induced arterial thrombi were significantly larger with the 0.6 mg/kg dose of acetylsalicylic acid than with the 0.15 mg/kg dose. Thrombi in mice treated with clopidogrel and the 0.15 mg/kg dose of acetylsalicylic acid were smaller than in mice treated with clopidogrel alone, suggesting that acetylsalicylic acid can add to the antithrombotic effect of clopidogrel but that higher doses of acetylsalicylic acid blunt the antithrombotic effect of clopidogrel. Conclusions- These findings support the use of lower, prostacyclin-preserving, doses of acetylsalicylic acid in conjunction with clopidogrel.[4] New Zealand white rabbits (1.9-2.7 kg) were treated orally with vehicle or clopidogrel (3 or 10 mg/kg) for three days. On the fourth day, the rabbits were anesthetized for blood collection and then euthanized. The brain was collected, and the middle cerebral arteries were isolated. We used light transmission aggregometry and pressure myography to elucidate the mechanisms of the off-target effects associated with clopidogrel treatment. We confirmed that inhibition of P2Y12 activation by clopidogrel inhibited ADP-induced platelet aggregation but had no impact on P2Y12-independent arachidonic acid- or collagen-induced platelet aggregation. Analysis of middle cerebral arteries from clopidogrel treated rabbits showed that clopidogrel did not affect P2Y4, P2Y6, and P2Y14 receptor-mediated contraction but attenuated the contractile response after P2Y2 receptor activation. Further analysis determined P2Y2-mediated constriction was endothelium-dependent. Vasoconstriction is a primary component of hemostasis, and impaired vasoconstriction can prolong bleeding. These results suggest clopidogrel inhibits the endothelial P2Y2 receptor in the middle cerebral artery, which provides a mechanistic explanation for the adverse cerebral bleeding associated with the drug.[5] Mouse arterial thrombosis model experiment: Male C57BL/6 mice were selected and randomly divided into a control group, Clopidogrel sulfate alone group (10 mg/kg), aspirin alone groups (1, 3, 10 mg/kg), and combined administration groups. All drugs were dissolved in normal saline and administered orally once a day for 7 consecutive days. Two hours after the last administration, carotid artery thrombosis was induced by FeCl₃, and the thrombosis time was recorded and the thrombus was weighed [4] - Rabbit cerebral vascular contraction experiment: Adult New Zealand white rabbits were selected and randomly grouped. Different doses of Clopidogrel sulfate (3, 10 mg/kg) were injected via the ear vein, and the control group was injected with an equal volume of normal saline. Thirty minutes after administration, the middle cerebral artery was isolated and placed in a vascular perfusion device. After stimulation with ADP, the vascular contraction amplitude and contraction speed were recorded [5] |
| ADME/Pharmacokinetics |
Metabolism: Clopidogrel sulfate is a prodrug, mainly metabolically activated by the hepatic cytochrome P450 enzyme system. Its metabolic process does not depend on the strong catalysis of specific CYP enzymes, and it has no significant inhibitory effect on major CYP enzymes, resulting in a low risk of drug metabolic interactions [1] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No published information is available on the use of clopidogrel during breastfeeding. The manufacturer reports that no adverse effects have been observed in breastfed infants with maternal clopidogrel use during lactation in a small number of postmarketing cases. Since no published information is available on the use of clopidogrel during breastfeeding, an alternate drug may be preferred, especially while nursing a newborn or preterm infant. If it is used by a nursing mother, monitor the infant for bruising and bleeding. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. In vitro toxicity: Clopidogrel sulfate can induce apoptosis of human gastric epithelial cells and activate the endoplasmic reticulum stress pathway, suggesting a potential risk of gastrointestinal toxicity [3] - Drug-drug interactions: In vitro experiments showed that Clopidogrel sulfate does not inhibit major human cytochrome P450 enzymes, and the possibility of metabolic interactions when combined with drugs metabolized by these enzymes is low [1] - In vivo toxicity: In mouse experiments, Clopidogrel sulfate (10 mg/kg) alone or in combination with aspirin did not cause a significant prolongation of bleeding time or organ damage [4] |
| References |
[1]. Comparison of human cytochrome P450 inhibition by the thienopyridines prasugrel, Clopidogrel, and ticlopidine. Drug Metab Pharmacokinet. 2008;23(6):412-20. [2]. P2Y12, a new platelet ADP receptor, target of Clopidogrel.Semin Vasc Med. 2003 May;3(2):113-22. [3]. Increased endoplasmic reticulum stress response is involved in Clopidogrel-induced apoptosis of gastric epithelial cells. PLoS One. 2013 Sep 13;8(9):e74381. [4]. Effect of Different Doses of Acetylsalicylic Acid on the Antithrombotic Activity of Clopidogrel in a Mouse Arterial Thrombosis Model.Arterioscler Thromb Vasc Biol. 2018 Oct;38(10):2338-2344. [5]. Clopidogrel treatment inhibits P2Y2-Mediated constriction in the rabbit middle cerebral artery [published online ahead of print, 2021 Oct 1]. Eur J Pharmacol. 2021;174545. |
| Additional Infomation |
Clopidogrel Bisulfate is a thienopyridine with antiplatelet activity. Clopidogrel bisulfate irreversibly alters the platelet receptor for adenosine diphosphate (ADP), thereby blocking the binding of ADP to its receptor, inhibiting ADP-mediated activation of the glycoprotein complex GPIIb/IIIa, and inhibiting fibrinogen binding to platelets and platelet adhesion and aggregation. (NCI04) A ticlopidine analog and platelet purinergic P2Y receptor antagonist that inhibits adenosine diphosphate-mediated PLATELET AGGREGATION. It is used to prevent THROMBOEMBOLISM in patients with ARTERIAL OCCLUSIVE DISEASES; MYOCARDIAL INFARCTION; STROKE; or ATRIAL FIBRILLATION. See also: Clopidogrel (has active moiety). Drug Indication Secondary prevention of atherothrombotic eventsClopidogrel is indicated in: adult patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from seven days until less than six months) or established peripheral arterial disease; adult patients suffering from acute coronary syndrome: non-ST-segment-elevation acute coronary syndrome (unstable angina or non-Q-wave myocardial infarction), including patients undergoing a stent placement following percutaneous coronary intervention, in combination with acetylsalicylic acid (ASA); ST-segment-elevation acute myocardial infarction, in combination with ASA in patients undergoing percutaneous coronary intervention (including patients undergoing a stent replacement) or medically treated patients eligible for thrombolytic/fibrinolytic therapy. In patients with moderate to high-risk Transient Ischemic Attack (TIA) or minor Ischemic Stroke (IS)Clopidogrel in combination with ASA is indicated in: Adult patients with moderate to high-risk TIA (ABCD2 score â¥4) or minor IS (NIHSS â¤3) within 24 hours of either the TIA or IS event. Prevention of atherothrombotic and thromboembolic events in atrial fibrillationIn adult patients with atrial fibrillation who have at least one risk factor for vascular events, are not suitable for treatment with vitamin-K antagonists and who have a low bleeding risk, clopidogrel is indicated in combination with ASA for the prevention of atherothrombotic and thromboembolic events, including stroke. Secondary prevention of atherothrombotic eventsClopidogrel is indicated in: Adult patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease. Adult patients suffering from acute coronary syndrome: Non-ST segment elevation acute coronary syndrome (unstable angina or non-Q-wave myocardial infarction), including patients undergoing a stent placement following percutaneous coronary intervention, in combination with acetylsalicylic acid (ASA). ST segment elevation acute myocardial infarction, in combination with ASA in patients undergoing percutaneous coronary intervention (including patients undergoing a stent placement) or medically treated patients eligible for thrombolytic/fibrinolytic therapy. In patients with moderate to high-risk Transient Ischemic Attack (TIA) or minor Ischemic Stroke (IS)Clopidogrel in combination with ASA is indicated in: Adult patients with moderate to high-risk TIA (ABCD2 score â¥4) or minor IS (NIHSS â¤3) within 24 hours of either the TIA or IS event. Prevention of atherothrombotic and thromboembolic events in atrial fibrillationIn adult patients with atrial fibrillation who have at least one risk factor for vascular events, are not suitable for treatment with Vitamin K antagonists (VKA) and who have a low bleeding risk, clopidogrel is indicated in combination with ASA for the prevention of atherothrombotic and thromboembolic events, including stroke. Secondary prevention of atherothrombotic events Clopidogrel is indicated in: Adult patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease. Adult patients suffering from acute coronary syndrome: Non-ST segment elevation acute coronary syndrome (unstable angina or non-Q-wave myocardial infarction), including patients undergoing a stent placement following percutaneous coronary intervention, in combination with acetylsalicylic acid (ASA). ST segment elevation acute myocardial infarction, in combination with ASA in medically treated patients eligible for thrombolytic therapy. Prevention of atherothrombotic and thromboembolic events in atrial fibrillationIn adult patients with atrial fibrillation who have at least one risk factor for vascular events, are not suitable for treatment with Vitamin K antagonists (VKA) and who have a low bleeding risk, clopidogrel is indicated in combination with ASA for the prevention of atherothrombotic and thromboembolic events, including stroke. Secondary prevention of atherothrombotic eventsClopidogrel is indicated in: Adult patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease. Adult patients suffering from acute coronary syndrome: - Non-ST segment elevation acute coronary syndrome (unstable angina or non-Q-wave myocardial infarction), including patients undergoing a stent placement following percutaneous coronary intervention, in combination with acetylsalicylic acid (ASA). - ST segment elevation acute myocardial infarction, in combination with ASA in medically treated patients eligible for thrombolytic therapy. Prevention of atherothrombotic and thromboembolic events in atrial fibrillationIn adult patients with atrial fibrillation who have at least one risk factor for vascular events, are not suitable for treatment with Vitamin K antagonists (VKA) and who have a low bleeding risk, clopidogrel is indicated in combination with ASA for the prevention of atherothrombotic and thromboembolic events, including stroke. Prevention Secondary prevention of atherothrombotic events Clopidogrel is indicated in: Adult patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease. Adult patients suffering from acute coronary syndrome: - Non-ST segment elevation acute coronary syndrome (unstable angina or non-Q-wave myocardial infarction), including patients undergoing a stent placement following percutaneous coronary intervention, in combination with acetylsalicylic acid (ASA). - ST segment elevation acute myocardial infarction, in combination with ASA in medically treated patients eligible for thrombolytic therapy. Prevention of atherothrombotic and thromboembolic events in atrial fibrillation: - In adult patients with atrial fibrillation who have at least one risk factor for vascular events, are not suitable for treatment with Vitamin K antagonists (VKA) and who have a low bleeding risk, clopidogrel is indicated in combination with ASA for the prevention of atherothrombotic and thromboembolic events, including stroke. Prevention of atherothrombotic events Clopidogrel is indicated in: Adult patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease. Clopidogrel is indicated in adults for the prevention of atherothrombotic events in: patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease. For further information please refer to section 5. 1. Clopidogrel is indicated in adults for the prevention of atherothrombotic events in: - Patients suffering from myocardial infarction (from a few days until less than 35 days), ischaemic stroke (from 7 days until less than 6 months) or established peripheral arterial disease. - Patients suffering from acute coronary syndrome: Non-ST segment elevation acute coronary syndrome (unstable angina or non-Q-wave myocardial infarction), including patients undergoing a stent placement following percutaneous coronary intervention, in combination with acetylsalicylic acid (ASA). ST segment elevation acute myocardial infarction, in combination with ASA in medically treated patients eligible for thrombolytic therapy. Clopidogrel sulfate is a clinically commonly used antiplatelet drug. It inhibits ADP-mediated platelet activation and aggregation by irreversibly binding to the platelet P2Y12 receptor, and is used for the prevention and treatment of thrombotic cardio-cerebrovascular diseases [2] - Its mechanism of action relies on hepatic metabolic activation to generate active metabolites that exert pharmacological effects. The binding to the P2Y12 receptor is irreversible, and platelet function recovery depends on the production of new platelets [2] - In the arterial thrombosis model, the combination of Clopidogrel sulfate and aspirin can enhance antithrombotic effects without significantly increasing the bleeding risk, providing experimental evidence for clinical combined medication [4] - Clopidogrel sulfate can inhibit P2Y2 receptor-mediated vascular contraction, which may have a certain improvement effect on cerebral vasospasm [5] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.95 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.95 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: 50 mg/mL (119.08 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3815 mL | 11.9076 mL | 23.8152 mL | |
| 5 mM | 0.4763 mL | 2.3815 mL | 4.7630 mL | |
| 10 mM | 0.2382 mL | 1.1908 mL | 2.3815 mL |