Physicochemical Properties
| Molecular Formula | C45H78O2 |
| Molecular Weight | 651.09962 |
| Exact Mass | 650.6 |
| CAS # | 303-43-5 |
| Related CAS # | Cholesteryl oleate-d7;1416275-35-8;Cholesteryl oleate-d7-1;2260669-48-3 |
| PubChem CID | 5283632 |
| Appearance | White to off-white solid powder |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 675.4±44.0 °C at 760 mmHg |
| Melting Point | 44-47 °C(lit.) |
| Flash Point | 362.9±16.0 °C |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C |
| Index of Refraction | 1.511 |
| LogP | 18.69 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 22 |
| Heavy Atom Count | 47 |
| Complexity | 977 |
| Defined Atom Stereocenter Count | 8 |
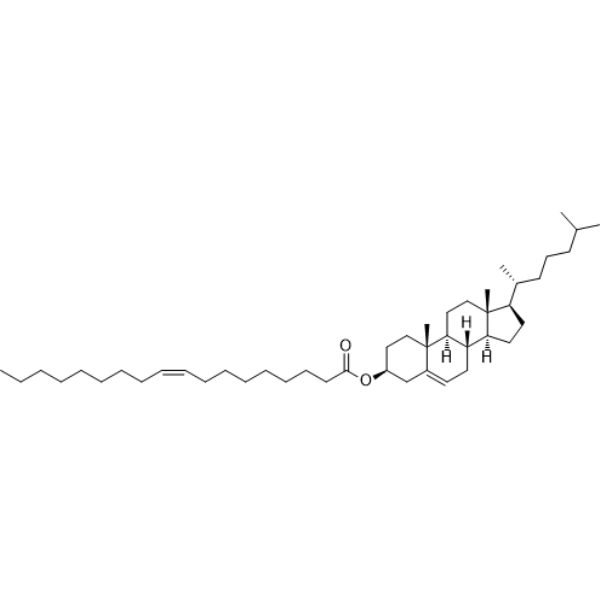
| SMILES | CCCCCCCC/C=C\CCCCCCCC(=O)O[C@H]1CC[C@@]2([C@H]3CC[C@]4([C@H]([C@@H]3CC=C2C1)CC[C@@H]4[C@H](C)CCCC(C)C)C)C |
| InChi Key | RJECHNNFRHZQKU-RMUVNZEASA-N |
| InChi Code | InChI=1S/C45H78O2/c1-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-25-43(46)47-38-30-32-44(5)37(34-38)26-27-39-41-29-28-40(36(4)24-22-23-35(2)3)45(41,6)33-31-42(39)44/h14-15,26,35-36,38-42H,7-13,16-25,27-34H2,1-6H3/b15-14-/t36-,38+,39+,40-,41+,42+,44+,45-/m1/s1 |
| Chemical Name | [(3S,8S,9S,10R,13R,14S,17R)-10,13-dimethyl-17-[(2R)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-yl] (Z)-octadec-9-enoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Cholesteryl oleate serves as a lipid component in cationic solid lipid nanoparticles (cSLNs) for siRNA delivery; cSLNs loaded with Cholesteryl oleate enhance cellular uptake of siRNA in HeLa cells, achieving a target gene (GFP) silencing efficiency of 78% at a siRNA concentration of 50 nM[2] Cholesteryl oleate-containing cSLNs show low cytotoxicity in HeLa cells; after 24-hour incubation with cSLNs (final Cholesteryl oleate concentration up to 20 μg/mL), cell viability remains above 85% compared to the control group[2] |
| Cell Assay |
For gene silencing and cellular uptake assay: Culture HeLa cells stably expressing GFP in RPMI 1640 medium supplemented with fetal bovine serum; seed cells in 24-well plates and incubate until 70-80% confluence; treat cells with Cholesteryl oleate-containing cSLNs (formulated with siRNA at 10-50 nM) for 4 hours; replace with fresh medium and continue culturing for 20 hours; analyze GFP expression via flow cytometry to quantify silencing efficiency, and observe cellular uptake of fluorescently labeled cSLNs via confocal laser scanning microscopy[2] For cytotoxicity assay: Seed HeLa cells in 96-well plates (5×10³ cells/well); treat cells with serial concentrations of Cholesteryl oleate-containing cSLNs (final Cholesteryl oleate concentration 5-20 μg/mL) for 24 hours; add MTT reagent and incubate for 4 hours; dissolve formazan crystals with DMSO; measure absorbance at 570 nm to evaluate cell viability[2] |
| Toxicity/Toxicokinetics | Cholesteryl oleate exhibits minimal in vitro cytotoxicity; when incorporated into cSLNs, it does not induce significant HeLa cell death at concentrations effective for gene silencing (≤20 μg/mL)[2] |
| References |
[1]. Study of the miscibility of cholesteryl oleate in a matrix of ceramide, cholesterol and fatty acid. Chem Phys Lipids. 2011 Oct;164(7):664-71. [2]. Cholesteryl oleate-loaded cationic solid lipid nanoparticles as carriers for efficient gene-silencing therapy. Int J Nanomedicine. 2018 May 30;13:3223-3233. |
| Additional Infomation |
Cholesteryl oleate is the (Z)-stereoisomer of cholesteryl octadec-9-enoate. It has a role as a mouse metabolite. It is a cholesteryl octadec-9-enoate and a CE(18:1). It is functionally related to an oleic acid. Cholesteryl oleate has been reported in Homo sapiens with data available. Cholesteryl oleate shows miscibility in ceramide-cholesterol-fatty acid matrices; it forms homogeneous mixtures with the matrix at Cholesteryl oleate mass fractions of 0.1-0.3, with no phase separation observed via differential scanning calorimetry and X-ray diffraction[1] The miscibility of Cholesteryl oleate in the lipid matrix is influenced by the fatty acid chain length; optimal miscibility is achieved with fatty acids containing 16-18 carbon atoms[1] Cholesteryl oleate is a key lipid component in cationic solid lipid nanoparticles, contributing to the stability of the nanoparticle structure and improving siRNA encapsulation efficiency and intracellular delivery[2] |
Solubility Data
| Solubility (In Vitro) |
Acetone : 50 mg/mL (~76.79 mM) Ethanol : ~12.5 mg/mL (~19.20 mM ) DMSO :< 1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (1.92 mM) (saturation unknown) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear EtOH stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 1.25 mg/mL (1.92 mM) (saturation unknown) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear EtOH stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 1.25 mg/mL (1.92 mM) (saturation unknown) in 10% EtOH + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear EtOH stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: ≥ 0.52 mg/mL (0.80 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.2 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 5: ≥ 0.52 mg/mL (0.80 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.2 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 6: ≥ 0.52 mg/mL (0.80 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.2 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5359 mL | 7.6793 mL | 15.3586 mL | |
| 5 mM | 0.3072 mL | 1.5359 mL | 3.0717 mL | |
| 10 mM | 0.1536 mL | 0.7679 mL | 1.5359 mL |