Physicochemical Properties
| Molecular Formula | C24H38O3 |
| Molecular Weight | 374.56 |
| Exact Mass | 374.282 |
| CAS # | 1253909-57-7 |
| PubChem CID | 78210211 |
| Appearance | Colorless to light yellow liquid |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 496.5±45.0 °C at 760 mmHg |
| Flash Point | 157.1±22.2 °C |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C |
| Index of Refraction | 1.504 |
| LogP | 7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 27 |
| Complexity | 484 |
| Defined Atom Stereocenter Count | 0 |
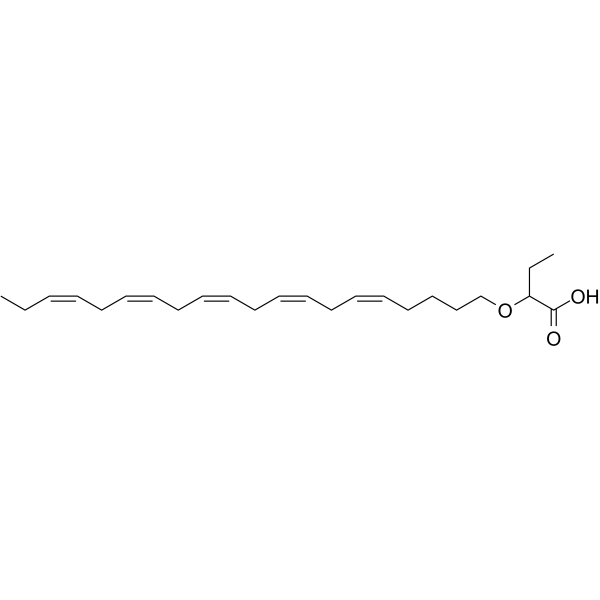
| SMILES | C(O)(=O)C(OCCCC/C=C\C/C=C\C/C=C\C/C=C\C/C=C\CC)CC |
| InChi Key | VOGXDRFFBBLZBT-AAQCHOMXSA-N |
| InChi Code | InChI=1S/C24H38O3/c1-3-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22-27-23(4-2)24(25)26/h5-6,8-9,11-12,14-15,17-18,23H,3-4,7,10,13,16,19-22H2,1-2H3,(H,25,26)/b6-5-,9-8-,12-11-,15-14-,18-17- |
| Chemical Name | 2-[(5Z,8Z,11Z,14Z,17Z)-icosa-5,8,11,14,17-pentaenoxy]butanoic acid |
| Synonyms | PRB01022; PRC-4016; PRB-01022 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Oral gavage of 100 mg/kg of icosalbutanil produced portal plasma flow rates (522 mL/h) that were significantly higher than mesenteric lymph fluid flow rates (0.5 mL/h). This suggests that icosalbutanol is almost entirely absorbed via the lymphatic route through the portal vein (>99%) in 8-week-old male Wistar rats [1]. Icosalbutanil ([14-C]-Icosalbutanil; oral gavage; 100 mg/kg; once) demonstrated peak radioactivity concentrations in most tissues 4–8 hours after administration (radiactivity levels in most other tissues are lower than those in the plasma of male albino Wistar rats [1]). Icosalbutinate (dietary administration; 135 mg/kg/day; 5 weeks) significantly improved glucose tolerance following oral glucose loading, significantly reducing AUC (0–120 min) by 60%, without affecting body weight, and lowering Plasma alanine aminotransferase (ALT) levels improve glucose metabolism by significantly lowering blood glucose, hemoglobin A1c, plasma insulin, and HOMA-IR in mice (-50%, -47%, -76%, and -87%, respectively) [ 1”. Oral icoxabutate (112 mg/kg/day; 20 weeks) inhibited hepatocellular hypertrophy (-82%) and microvesicular steatosis (-35%) but not macrovesicular steatosis. Despite a similar decrease in liver inflammatory cell aggregates, only icosabutate decreased the amount of liver collagen after 20 weeks of treatment [1]. |
| Animal Protocol |
Animal/Disease Models: 6-8 weeks old male ob/ob mice [1] Doses: 135 mg/kg Route of Administration: 135 mg/kg/day, administered through diet; 5-week Experimental Results: Improved glucose metabolism and insulin resistance. Animal/Disease Models: 8-15 weeks old male APOE*3 Leiden.CETP mice, fed a high-fat and high-cholesterol diet [1] Doses: 112 mg/kg/day Route of Administration: po (oral gavage); 20-week Experimental Results: Improved microbiome Alveolar steatosis, liver inflammation, and fibrosis. |
| References |
[1]. Icosabutate Exerts Beneficial Effects Upon Insulin Sensitivity, Hepatic Inflammation, Lipotoxicity, and Fibrosis in Mice.Hepatol Commun. 2019 Dec 24;4(2):193-207. [2]. Icosabutate, a Structurally Engineered Fatty Acid, Improves the Cardiovascular Risk Profile in Statin-Treated Patients with Residual Hypertriglyceridemia.Cardiology. 2016;135(1):3-12. |
| Additional Infomation |
Icosabutate is under investigation in clinical trial NCT02373176 ([14C] Icosabutate -A Phase I Absorption, Metabolism and Excretion Study). Drug Indication Treatment of non-alcoholic fatty liver disease including non-alcoholic steatohepatitis |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~266.98 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.67 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (6.67 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.67 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6698 mL | 13.3490 mL | 26.6980 mL | |
| 5 mM | 0.5340 mL | 2.6698 mL | 5.3396 mL | |
| 10 mM | 0.2670 mL | 1.3349 mL | 2.6698 mL |