Physicochemical Properties
| Molecular Formula | C10H13CLN2O |
| Molecular Weight | 212.677 |
| Exact Mass | 212.071 |
| CAS # | 15545-48-9 |
| Related CAS # | Chlorotoluron-d6;1219803-48-1 |
| PubChem CID | 27375 |
| Appearance |
COLORLESS CRYSTALS white powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 345.9±52.0 °C at 760 mmHg |
| Melting Point | 147-148ºC |
| Flash Point | 163.0±30.7 °C |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.541 |
| LogP | 2.62 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 14 |
| Complexity | 208 |
| Defined Atom Stereocenter Count | 0 |
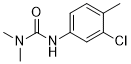
| SMILES | CC1=C(Cl)C=C(NC(N(C)C)=O)C=C1 |
| InChi Key | JXCGFZXSOMJFOA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C10H13ClN2O/c1-7-4-5-8(6-9(7)11)12-10(14)13(2)3/h4-6H,1-3H3,(H,12,14) |
| Chemical Name | 3-(3-chloro-4-methylphenyl)-1,1-dimethylurea |
| Synonyms | Chlorotoluron Dikurin Dicuran |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion ...(TOLYL-(14)C)CHLORTOLURON WAS ADMIN DAILY TO RATS FOR...2 WK...CONCN OF RADIOACTIVITY IN BLOOD & VARIOUS ORGANS REACHED THEIR HIGHEST LEVEL ON 1ST OR 2ND DAY...& DID NOT INCR FOR REMAINDER OF EXPT. ...WHEN FEEDING OF LABEL WAS ABANDONED, RADIOACTIVITY IN ALL TISSUES DECR BELOW DETECTABLE LEVELS WITHIN...9 DAYS. ...PHENYLUREAS LABELED WITH (14)C IN DIFFERENT POSITIONS WERE ADMIN ORALLY TO RATS, IT WAS OBSERVED THAT TOTAL RADIOACTIVITY WAS RAPIDLY & EFFICIENTLY ELIMINATED IN URINE & FECES. ...90% OR MORE OF RADIOACTIVITY APPLIED WAS ACCOUNTED FOR IN EXCRETA WITHIN 72 HR AFTER ADMIN...(TOLYL-(14)C)CHLORTOLURON. WITH MOST MENTIONED CMPD /(TOLYL-(14)C)CHLORTOLURON/ MAJOR PORTION OF LABEL WAS ELIMINATED IN URINE, WHEReAS AMT OF FECAL RADIOACTIVITY WAS USUALLY NO MORE THAN 20% IN TERMS OF DOSE APPLIED. Metabolism / Metabolites RECOGNIZED N-DEMETHYLATION &/OR N-DEMETHOXYLATION REACTIONS OF PHENYLUREA HERBICIDES IN BIOLOGICAL ENVIRONMENTS--CHLORTOLURON-- SOIL/MICROBIAL MEDIA: IN SILTY CLAY SOIL; PLANTS: IN WHEAT; MAMMALS: IN RAT /FROM TABLE/ ...FATE OF (14)C-TOLYL-LABELED HERBICIDE /CHLORTOLURON/ IN RAT. ...METHYL RING SUBSTITUENT WAS SUBJECTED TO STEPWISE OXIDATION, YIELDING FIRST CORRESPONDING HYDROXYMETHYL AND THEN CARBOXY DERIVATIVE. ... COMBINED N-DEMETHYLATION/SIDE-CHAIN OXIDATION PATHWAY IN RATS IS /SRP: 3-(3-CHLORO-4-HYDROXYMETHYLPHENYL)-1,1-DIMETHYLUREA; 3-(3-CHLORO-4-CARBOXYPHENYL)-1,1-DIMETHYLUREA; 3-(3-CHLORO-4-HYDROXYMETHYLPHENYL)-1-METHYLUREA; 3-(3-CHLORO-4-CARBOXYPHENYL)-1-METHYLUREA; 3-(3-CHLORO-4-HYDROXYMETHYLPHENYL)UREA AND 3-(3-CHLORO-4-CARBOXYPHENYL)UREA/. THE METABOLISM OF CHLORTOLURON IN WHEAT EXHIBITED ESSENTIALLY THE SAME QUALITATIVE PATTERN OF DEGRADATION. HOWEVER, THE MAJOR PORTION OF THE HYDROXYMETHYL METABOLITE (3-(3-CHLORO-4-HYDROXYMETHYLPHENYL)-1,1-DIMETHYLUREA) WAS FIRST TRAPPED AS WATER-SOL CONJUGATE(S). FURTHER OXIDATION TO CORRESPONDING CARBOXY DERIVATIVE OCCURRED ONLY IN LATER STAGES OF PLANT GROWTH. IN ADDN, N-DEMETHYLATION DID NOT PROCEED BEYOND N-MONOMETHYL STAGE. The metabolism of chlortoluron was investigated in several plant species. Chlortoluron was degraded via both N-demethylation and ring-methyl oxidation reactions. The latter pathway was the major degradative route in wheat and barley. Since the products of ring-methyl oxidation are nonphytotoxic, the existence of such a detoxification mechanism in these cereals may account for their enhanced resistance to chlortoluron. The major metabolites in the susceptible cereal weeds, Avena fatua, Alopecurus myosuroides and Lolium perenne, and in resistant cotton were products of N-demethylation. This reaction in the cereal weeds proceeded only very slightly beyond the state the mono-N-demethylated metabolites some of which retain considerable phytotoxicity. In cotton, however, N-demethylation was more efficient, giving rise to substantial amounts of N-didemethylated metabolites which are non-phytotoxic. For more Metabolism/Metabolites (Complete) data for CHLORTOLURON (7 total), please visit the HSDB record page. |
| Toxicity/Toxicokinetics |
Interactions Levels of cytochrome p450s and rates of monooxygenase activities were studied in microsomes prepared from wheat cell suspension cultures. Cytochrome contents and enzymatic activities, namely, the enzymatic systems responsible for chlorotoluron ring-methy1 hydroxy1ation and N-demethylation, were enhanced after pretreatment of cells with 2,4-D, procloraz, mecoprop, chlorotoluron, and oxime ether safeners. Effects of various agrochemicals were also determined on chlorotoluron ring-methyl hydroxylase and N-demethylase, following their direct addition to microsomal preparations. Plant growth regulators and fungicide, as well as piperonyl butoxide decreased both activities, tetcyclacis, and procloraz being marked1y inhibitory. Naphthalic anhydride, oxime ether safeners, dich1ormid, and tridiphane had only weak effects. The substrate specificities of chlorotoluron ring-methyl hydroxylase and N-demethylase were also investigated using structural analogues of the herbicide. Diuron was the strongest inhibitor among the tested phenylureas. Other herbicides that can be metabolized by wheat affected both activities to different extents. However, diclofop enhanced only chlorotoluron N-demethylase. Non-Human Toxicity Values LD50 Rat oral greater than 10000 mg/kg LD50 Rat subcutaneous greater than 2000 mg/kg |
| Additional Infomation |
Chlorotoluron is a member of the class of phenylureas that is urea in which one of the nitrogens is substituted by two methyl groups while the other is substituted by a 3-chloro-4-methylphenyl group. A herbicide that is non-toxic to honeybees but moderately toxic to mammals, birds, earthworms and most aquatic organisms. It has a role as a xenobiotic, an environmental contaminant, an agrochemical and a herbicide. It is a member of monochlorobenzenes and a member of phenylureas. Mechanism of Action Inhibits photosynthesis. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~235.09 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7019 mL | 23.5095 mL | 47.0190 mL | |
| 5 mM | 0.9404 mL | 4.7019 mL | 9.4038 mL | |
| 10 mM | 0.4702 mL | 2.3509 mL | 4.7019 mL |