On April 03, 2024, the U.S. Food and Drug Administration approved Zevtera (ceftobiprole medocaril sodium for injection) for the treatment of adults with Staphylococcus aureus bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis; adults with acute bacterial skin and skin structure infections (ABSSSI); and adult and pediatric patients three months to less than 18 years old with community-acquired bacterial pneumonia (CABP). “The FDA is committed to fostering new antibiotic availability when they prove to be safe and effective, and Zevtera will provide an additional treatment option for a number of serious bacterial infections,” said Peter Kim, M.D., M.S., director of the Division of Anti-Infectives in the FDA’s Center for Drug Evaluation and Research. “The FDA will continue our important work in this area as part of our efforts to protect the public health.”
Physicochemical Properties
| Molecular Formula | C26H25N8NAO11S2 |
| Molecular Weight | 712.643473386765 |
| Exact Mass | 712.098 |
| Elemental Analysis | C, 43.82; H, 3.54; N, 15.72; Na, 3.23; O, 24.70; S, 9.00 |
| CAS # | 252188-71-9 |
| Related CAS # | Ceftobiprole medocaril;376653-43-9; 252188-71-9 (sodium); 209467-52-7 (ceftobiprole) |
| PubChem CID | 135413543 |
| Appearance | Off-white to light yellow solid powder |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 17 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 48 |
| Complexity | 1570 |
| Defined Atom Stereocenter Count | 3 |
| SMILES | CC1=C(OC(=O)O1)COC(=O)N2CC[C@H](C2)N3CC/C(=C\C4=C(N5[C@@H]([C@@H](C5=O)NC(=O)/C(=N\O)/C6=NSC(=N6)N)SC4)C(=O)[O-])/C3=O.[Na+] |
| InChi Key | MFAWUGGPPMTWPU-LCJFHXTKSA-M |
| InChi Code | InChI=1S/C26H26N8O11S2.Na/c1-10-14(45-26(41)44-10)8-43-25(40)32-4-3-13(7-32)33-5-2-11(20(33)36)6-12-9-46-22-16(21(37)34(22)17(12)23(38)39)28-19(35)15(30-42)18-29-24(27)47-31-18;/h6,13,16,22,42H,2-5,7-9H2,1H3,(H,28,35)(H,38,39)(H2,27,29,31);/q;+1/p-1/b11-6+,30-15-;/t13-,16-,22-;/m1./s1 |
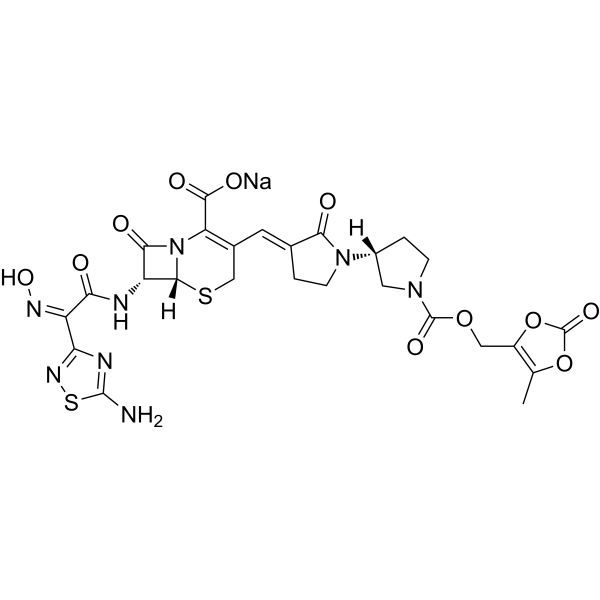
| Chemical Name | sodium;(6R,7R)-7-[[(2Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-hydroxyiminoacetyl]amino]-3-[(E)-[1-[(3R)-1-[(5-methyl-2-oxo-1,3-dioxol-4-yl)methoxycarbonyl]pyrrolidin-3-yl]-2-oxopyrrolidin-3-ylidene]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate |
| Synonyms | Ceftobiprole medocaril; Ceftobiprole medocaril sodium; BAL5788; Ro 65-5788; (6R,7R)-7-[[(2Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-hydroxyiminoacetyl]amino]-3-[(E)-[1-[(3R)-1-[(5-methyl-2-oxo-1,3-dioxol-4-yl)methoxycarbonyl]pyrrolidin-3-yl]-2-oxopyrrolidin-3-ylidene]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Penicillin-binding proteins (PBPs); bacterial cell wall synthesis. |
| ln Vitro |
Ceftobiprole Medocaril Sodium is the sodim salt form of ceftobiprole medocaril, a water-soluble prodrug of ceftobiprole, a pyrrolidinone cephalosporin antibiotic, with bactericidal activity. Ceftobiprole binds to and inactivates penicillin-binding proteins (PBPs), enzymes involved in the terminal stages of bacterial cell wall assembly and cell wall reshaping during bacterial growth and division. This agent exhibits a broad spectrum of activity against gram-negative and gram-positive pathogens including methicillin-resistant S. aureus (MRSA), vancomycin-intermediate S. aureus (VISA) and vancomycin-resistant S. aureus (VRSA). Ceftobiprole is refractory to hydrolysis by class A and class C lactamases.
Ceftobiprole has demonstrated _in vitro_ activity against Gram-positive and Gram-negative bacteria. [Ceftobiprole], the active moiety of ceftobiprole medocaril, exhibits its bactericidal activity by inhibition of bacterial cell wall synthesis. This activity is mediated through binding to essential penicillin-binding proteins (PBPs) and inhibiting their transpeptidase activity, which is essential for the synthesis of the peptidoglycan layer of the bacterial cell wall. Ceftobiprole has demonstrated _in vitro_ activity against both Gram-positive and Gram-negative bacteria. In Gram-positive bacteria, including methicillin-resistant _Staphylococcus aureus_ (MRSA), Ceftobiprole binds to PBP2a. Ceftobiprole also binds to PBP2b in _Streptococcus pneumoniae_ (penicillin-intermediate), PBP2x in _S. pneumoniae_ (penicillin resistant), and to PBP5 in _Enterococcus faecalis_. |
| ln Vivo |
Ceftobiprole medocaril (sc; 3 × q12h; total daily dose of BAL9141 equivalent, 2.1, 4.2, or 8.4 mg/kg) induces infection in female Swiss albino mice (body weight 20 to 22 g) with Penr Cros Ctxs strain P-15986 [1]. The T1/2 of cefbiprole medocaril (10, 40, 160 mg/kg; single dose; subcutaneous injection) ranged from 20 minutes to 31 minutes as the dose was increased from 40 mg/kg to 160 mg/kg. In neutropenic thigh-infected mice, single ascending dosage AUC/dose values ranged from 0.585 to 1.33, while Cmax/dose values fell from 1.08 to 0.90 [2].
Ceftobiprole medocaril is an antibacterial indicated for the treatment of adult patients with _Staphylococcus aureus_ bloodstream infections (bacteremia) (SAB), including those with right-sided infective endocarditis. It is additionally indicated in adult patients with acute bacterial skin and skin structure infections (ABSSSI). It is indicated in adult and pediatric patients ≥3 months of age for the treatment of community-acquired bacterial pneumonia (CABP). In Canada, it is indicated for the treatment of both community- and hospital-acquired pneumonia (excluding ventilator-associated pneumonia). In a neutropenic murine thigh infection model, therapeutic efficacy is correlated with the time that the unbound plasma concentration of ceftobiprole exceeds the minimum inhibitory concentration (MIC) of _S. aureus_, _S. pneumoniae_, and _Enterobacterales spp._ It is not active against Gram-negative bacteria producing extended-spectrum beta-lactamases (ESBLs) from the TEM, SHV, or CTX-M families, serine carbapenemases (such as KPC), class B metallo-beta-lactamases, class C (AmpC cephalosporinases) if expressed at high levels, and Ambler class D beta-lactamases including carbapenemases. Ceftobiprole is not indicated for use in patients with ventilator-associated bacterial pneumonia (VABP) - in clinical trials, a statistically significant increase in mortality was seen in patients with VABP treated with ceftobiprole medocaril as compared to comparator-treated patients. |
| ADME/Pharmacokinetics |
Absorption Because ceftobiprole medocaril is administered intravenously, its bioavailability is 100%. The mean Cmax and AUC0-8h after multiple-dose administration are 33.0 µg/mL and 102 µg*h/mL, respectively. Route of Elimination Active [ceftobiprole] is eliminated primarily unchanged by renal excretion. Approximately 89% of the administered dose is recovered in the urine as active ceftobiprole (83%), the open-ring metabolite (5%) and ceftobiprole medocaril (<1%). Due to the significant degree of renal elimination, patients with renal impairment who are undergoing treatment with ceftobiprole may require lower doses. Volume of Distribution The steady-state volume of distribution of active [ceftobiprole] is 15.5-18.0 L, which approximates extracellular fluid volume in humans. Clearance The mean clearance of active [ceftobiprole] following multiple-dose administration is 4.98 L/h. Protein Binding Active [ceftobiprole] is minimally (16%) bound to plasma proteins. Metabolism / Metabolites Conversion of prodrug ceftobiprole medocaril to the active moiety ceftobiprole occurs rapidly and is mediated by non-specific plasma esterases. Ceftobiprole itself is minimally metabolized to a microbiologically inactive open-ring metabolite, which accounts for approximately 4% of the parent exposure in subject with a normal renal function. Biological Half-Life The half-life of active [ceftobiprole] following multiple-dose administration is approximately 3.3 hours. |
| Toxicity/Toxicokinetics |
For adults with SAB, the most common side effects of Zevtera included anemia, nausea, low levels of potassium in the blood (hypokalemia), vomiting, diarrhea, increased levels of certain liver tests (hepatic enzymes and bilirubin), increased blood creatinine, high blood pressure, low white blood cell count (leukopenia), fever, abdominal pain, fungal infection, headache and shortness of breath (dyspnea).
For adults with ABSSSI, the most common side effects of Zevtera included nausea, diarrhea, headache, injection site reaction, increased levels of hepatic enzymes, rash, vomiting and altered taste (dysgeusia). For adults with CABP, the most common side effects of Zevtera included nausea, increased levels of hepatic enzymes, vomiting, diarrhea, headache, rash, insomnia, abdominal pain, vein inflammation (phlebitis), high blood pressure and dizziness. For pediatric patients with CABP, the most common side effects of Zevtera included vomiting, headache, increased levels of hepatic enzymes, diarrhea, infusion site reaction, vein inflammation (phlebitis) and fever. Patients should not use Zevtera if they have a known history of severe hypersensitivity to ceftobiprole or any of the components of Zevtera, or other members of the cephalosporin antibacterial class. Zevtera comes with certain warnings and precautions such as increased mortality in ventilator-associated bacterial pneumonia patients (an unapproved use), hypersensitivity reactions, seizures and other central nervous system reactions and Clostridioides difficile-associated diarrhea. https://www.fda.gov/news-events/press-announcements/fda-approves-new-antibiotic-three-different-uses |
| References |
[1]. Efficacy of BAL5788, a prodrug of cephalosporin BAL9141, in a mouse model of acute pneumococcal pneumonia. Antimicrob Agents Chemother. 2004 Apr;48(4):1105-11. [2]. In vivo pharmacodynamics of ceftobiprole against multiple bacterial pathogens in murine thigh and lung infection models. Antimicrob Agents Chemother. 2008 Oct;52(10):3492-6. |
| Additional Infomation |
Ceftobiprole Medocaril Sodium is a water-soluble prodrug of ceftobiprole, a pyrrolidinone cephalosporin antibiotic, with bactericidal activity. Ceftobiprole binds to and inactivates penicillin-binding proteins (PBPs), enzymes involved in the terminal stages of bacterial cell wall assembly and cell wall reshaping during bacterial growth and division. This agent exhibits a broad spectrum of activity against gram-negative and gram-positive pathogens including methicillin-resistant S. aureus (MRSA), vancomycin-intermediate S. aureus (VISA) and vancomycin-resistant S. aureus (VRSA). Ceftobiprole is refractory to hydrolysis by class A and class C lactamases. Drug Indication Treatment of pneumonia Treatment of complicated skin and soft-tissue infections Ceftobiprole is a fifth-generation cephalosporin antibiotic having (E)-[(3'R)-2-oxo[1,3'-bipyrrolidin]-3-ylidene]methyl and [(2Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-(hydroxyimino)acetyl]amino side groups located at positions 3 and 7 respectively; developed for the treatment of hospital-acquired pneumonia (HAP, excluding ventilator-associated pneumonia, VAP) and community-acquired pneumonia (CAP). It has a role as an antimicrobial agent. It is a cephalosporin and a member of thiadiazoles. Ceftobiprole is a cephalosporin antibiotic with activity against methicillin-resistant Staphylococcus aureus. It was discovered by Basilea Pharmaceutica and is being developed by Johnson & Johnson Pharmaceutical Research and Development. Ceftobiprole is the first cephalosporin to demonstrate clinical efficacy in patients with infections due to methicillin-resistant staphylococci and, if approved by regulatory authorities, is expected to be a useful addition to the armamentarium of agents for the treatment of complicated skin infections and pneumonia. Ceftobiprole is a broad-spectrum, fifth-generation, pyrrolidinone cephalosporin with antibacterial activity. Ceftobiprole binds to and inactivates penicillin-binding proteins (PBPs) located on the inner membrane of the bacterial cell wall. PBPs are enzymes involved in the terminal stages of assembling the bacterial cell wall and in reshaping the cell wall during growth and division. Inactivation of PBPs interferes with the cross-linkage of peptidoglycan chains necessary for bacterial cell wall strength and rigidity. This results in the weakening of the bacterial cell wall and causes cell lysis. Drug Indication For the treatment of serious bacterial infections in hospitalised patients. Mechanism of Action Cephalosporins, such as ceftobiprole, are bactericidal and have the same mode of action as other beta-lactam antibiotics (such as penicillins). Cephalosporins disrupt the synthesis of the peptidoglycan layer of bacterial cell walls. The peptidoglycan layer is important for cell wall structural integrity, especially in Gram-positive organisms. The final transpeptidation step in the synthesis of the peptidoglycan is facilitated by transpeptidases known as penicillin binding proteins (PBPs). PBPs bind to the D-Ala-D-Ala at the end of muropeptides (peptidoglycan precursors) to crosslink the peptidoglycan. Beta-lactam antibiotics mimic this site and competitively inhibit PBP crosslinking of peptidoglycan. Pharmacodynamics Ceftobiprole, a cephalosporin antibiotic, is active against methicillin-resistant Staphylococcus aureus. Ceftobiprole medocaril is a cephalosporin. It has a role as a prodrug. Ceftobiprole medocaril is a prodrug of [ceftobiprole], a fifth-generation semisynthetic cephalosporin antibacterial. Ceftobiprole is a broad-spectrum agent with demonstrated activity against both Gram-positive and Gram-negative bacteria, including antibiotic-resistant strains of Staphylcoccus aureus (methicillin-resistant Staphylococcus aureus; MRSA). The EMA's Committee for Medicinal Products for Human Use (CHMP) adopted a negative opinion of ceftobiprole medocaril in February 2010, recommending the refusal of its marketing authorization in the European Union primarily due to data quality issues in pivotal clinical studies. It received its first approval in Canada in October 2017 for use in certain patients with bacterial pneumonia, and was subsequently approved in the United States with additional indications for skin and skin structure infections and bacteremia in April 2024. Ceftobiprole Medocaril Sodium is the sodim salt form of ceftobiprole medocaril, a water-soluble prodrug of ceftobiprole, a pyrrolidinone cephalosporin antibiotic, with bactericidal activity. Ceftobiprole binds to and inactivates penicillin-binding proteins (PBPs), enzymes involved in the terminal stages of bacterial cell wall assembly and cell wall reshaping during bacterial growth and division. This agent exhibits a broad spectrum of activity against gram-negative and gram-positive pathogens including methicillin-resistant S. aureus (MRSA), vancomycin-intermediate S. aureus (VISA) and vancomycin-resistant S. aureus (VRSA). Ceftobiprole is refractory to hydrolysis by class A and class C lactamases. Zevtera’s efficacy in treating SAB was evaluated in a randomized, controlled, double-blind, multinational, multicenter trial. In the trial, researchers randomly assigned 390 subjects to receive Zevtera (192 subjects) or daptomycin plus optional aztreonam [the comparator] (198 subjects). The primary measure of efficacy for this trial was the overall success (defined as survival, symptom improvement, S. aureus bacteremia bloodstream clearance, no new S. aureus bacteremia complications and no use of other potentially effective antibiotics) at the post-treatment evaluation visit, which occurred 70 days after being randomly assigned an antibiotic. A total of 69.8% of subjects who received Zevtera achieved overall success compared to 68.7% of subjects who received the comparator. Zevtera’s efficacy in treating ABSSSI was evaluated in a randomized, controlled, double-blind, multinational trial. In the trial, researchers randomly assigned 679 subjects to receive either Zevtera (335 subjects) or vancomycin plus aztreonam [the comparator] (344 subjects). The primary measure of efficacy was early clinical response 48-72 hours after start of treatment. Early clinical response required a reduction of the primary skin lesion by at least 20%, survival for at least 72 hours and the absence of additional antibacterial treatment or unplanned surgery. Of the subjects who received Zevtera, 91.3% achieved an early clinical response within the necessary timeframe compared to 88.1% of subjects who received the comparator. Zevtera’s efficacy in treating adult patients with CABP was evaluated in a randomized, controlled, double-blind, multinational, multicenter trial. In the trial, researchers randomly assigned 638 adults hospitalized with CABP and requiring IV antibacterial treatment for at least 3 days to receive either Zevtera (314 subjects) or ceftriaxone with optional linezolid [the comparator] (324 subjects). The primary measurement of efficacy were clinical cure rates at test-of-cure visit, which occurred 7-14 days after end-of-treatment. Of the subjects who received Zevtera, 76.4% achieved clinical cure compared to 79.3% of subjects who received the comparator. An additional analysis considered an earlier timepoint of clinical success at Day 3, which was 71% in patients receiving Zevtera and 71.1% in patients receiving the comparator. Given the similar course of CABP in adults and pediatric patients, today’s approval of Zevtera in pediatric patients three months to less than 18 years with CABP was supported by evidence from the CABP trial of Zevtera in adults and a trial in 138 pediatric subjects three months to less than 18 years of age with pneumonia. https://www.fda.gov/news-events/press-announcements/fda-approves-new-antibiotic-three-different-uses |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~350.81 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4032 mL | 7.0162 mL | 14.0323 mL | |
| 5 mM | 0.2806 mL | 1.4032 mL | 2.8065 mL | |
| 10 mM | 0.1403 mL | 0.7016 mL | 1.4032 mL |