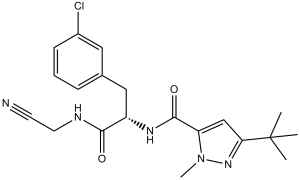
Cathepsin Inhibitor 1 (CTS-IN-1) is a novel and potent inhibitor of Cathepsin (L, L2, S, K, B) with the potential to be used in the treatment of osteoarthritis. With respective pIC50 values of 7.9, 6.7, 6.0, 5.5, and 5.2, it inhibits Cathepsin (L, L2, S, K, and B). With underlying bone damage and articular cartilage loss leading to joint instability and pain, osteoarthritis is now understood to be a chronic degenerative disease. One possible target for intervention in the treatment of osteoarthritis is the lysosomal cysteine protease2 Cathepsin L (CatL). Major components of articular cartilage, such as aggrecan4 and type II collagen, are proteoglycans that can be degraded by CatL once it is secreted into the extracellular matrix.
Physicochemical Properties
| Molecular Formula | C20H24CLN5O2 | |
| Molecular Weight | 401.89 | |
| Exact Mass | 401.162 | |
| Elemental Analysis | C, 59.77; H, 6.02; Cl, 8.82; N, 17.43; O, 7.96 | |
| CAS # | 225120-65-0 | |
| Related CAS # |
|
|
| PubChem CID | 44224135 | |
| Appearance | White to off-white solid powder | |
| Density | 1.23±0.1 g/cm3 (20 ºC 760 Torr) | |
| LogP | 3.766 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 28 | |
| Complexity | 612 | |
| Defined Atom Stereocenter Count | 1 | |
| SMILES | CC(C)(C1=NN(C(C(N[C@H](C(NCC#N)=O)CC2=CC(Cl)=CC=C2)=O)=C1)C)C |
|
| InChi Key | MZRVIHRERYCHBL-HNNXBMFYSA-N | |
| InChi Code | InChI=1S/C20H24ClN5O2/c1-20(2,3)17-12-16(26(4)25-17)19(28)24-15(18(27)23-9-8-22)11-13-6-5-7-14(21)10-13/h5-7,10,12,15H,9,11H2,1-4H3,(H,23,27)(H,24,28)/t15-/m0/s1 | |
| Chemical Name | 5-tert-butyl-N-[(2S)-3-(3-chlorophenyl)-1-(cyanomethylamino)-1-oxopropan-2-yl]-2-methylpyrazole-3-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Cathepsin L (pIC50 = 7.9); Cathepsin L2 (pIC50 = 6.7); Cathepsin S (pIC50 = 6); Cathepsin K (pIC50 = 5.5); Cathepsin B (pIC50 = 5.2) Cathepsin L: - IC₅₀ = 0.12 μM (fluorogenic substrate Z-Phe-Arg-AMC assay) [1] |
||
| ln Vitro |
Cathepsin Inhibitor 1 (compound 25) is a cathepsin (L, L2, S, K, B) inhibitor with pIC50 values of 7.9, 6.7, 6.0, 5.5, and 5.2, in that order. Cathepsin L inhibition: 1. Enzyme activity: Cathepsin Inhibitor 1 (0.01–1 μM) concentration-dependently inhibited recombinant human Cathepsin L. At 0.1 μM, inhibition rate reached ~85% (Z-Phe-Arg-AMC cleavage assay) [1] 2. Selectivity: No significant inhibition of Cathepsin B (1 μM, >90% activity remaining) or Cathepsin S (1 μM, >80% activity remaining) [1] |
||
| ln Vivo |
|
||
| Enzyme Assay |
Cathepsin L inhibition assay: 1. Protein preparation: Recombinant human Cathepsin L expressed in Escherichia coli, purified via nickel-chelate chromatography, activated with 10 mM DTT in 50 mM sodium acetate buffer (pH 5.5) [1] 2. Reaction setup: 100 μL mixture contained activated Cathepsin L (0.2 μg), fluorescent substrate Z-Phe-Arg-AMC (20 μM), Cathepsin Inhibitor 1 (0.01–1 μM), and 50 mM sodium acetate buffer (pH 5.5). Vehicle (DMSO) used as control [1] 3. Incubation and detection: Incubated at 37°C for 60 minutes; fluorescence intensity measured at 10-minute intervals (excitation 360 nm, emission 460 nm). Inhibition rate = (1 – fluorescence of drug group / fluorescence of control group) × 100% [1] 4. Data analysis: IC₅₀ calculated via four-parameter logistic fitting [1] |
||
| Animal Protocol |
|
||
| References |
[1]. Dipeptidyl nitrile inhibitors of Cathepsin L. Bioorg Med Chem Lett. 2009 Aug 1;19(15):4280-3. |
||
| Additional Infomation |
Background: Cathepsin Inhibitor 1 is a dipeptidyl nitrile compound designed as a selective inhibitor of Cathepsin L. It was developed to explore the role of Cathepsin L in pathological processes (e.g., cancer invasion, inflammation) [1] - Mechanism of action: Covalently binds to the active-site cysteine residue of Cathepsin L, irreversibly inhibiting its proteolytic activity [1] - Synthetic route: Prepared via solid-phase peptide synthesis, followed by nitrile group introduction. Total yield ≈ 25% over 5 steps [1] - Structural optimization: Introduction of a nitrile moiety improved binding affinity compared to earlier aldehyde-based inhibitors [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4882 mL | 12.4412 mL | 24.8824 mL | |
| 5 mM | 0.4976 mL | 2.4882 mL | 4.9765 mL | |
| 10 mM | 0.2488 mL | 1.2441 mL | 2.4882 mL |