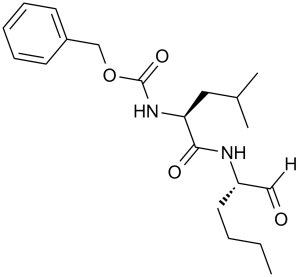
Calpeptin (sequence: Z-Leu-nLeu-H) is a novel, potent, cell-permeable calpain inhibitor that has anti-fibrotic properties. With IC50s of 52 nM, 34 nM, 138 nM, and 40 nM, respectively, it inhibits Calpain I (porcine erythrocytes), Calpain II (porcine kidney), and Calpain I (human platelets). In an rat model of multiple sclerosis, it reduces apoptosis, inflammation, cell death, and axonal damage. Calpeptin also inhibited the migration of cells that was dependent on angiopoietin-1 and the proliferation of cells that was dependent on IL-6. This may be the fundamental mechanism underlying Calpeptin's protective effect against pulmonary fibrosis.
Physicochemical Properties
| Molecular Formula | C20H30N2O4 | |
| Molecular Weight | 362.46 | |
| Exact Mass | 362.22 | |
| Elemental Analysis | C, 66.27; H, 8.34; N, 7.73; O, 17.66 | |
| CAS # | 117591-20-5 | |
| Related CAS # |
|
|
| PubChem CID | 73364 | |
| Appearance | White to off-white solid powder | |
| Density | 1.1±0.1 g/cm3 | |
| Boiling Point | 550.7±45.0 °C at 760 mmHg | |
| Melting Point | 60-75 °C | |
| Flash Point | 286.8±28.7 °C | |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C | |
| Index of Refraction | 1.508 | |
| LogP | 4.37 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 12 | |
| Heavy Atom Count | 26 | |
| Complexity | 434 | |
| Defined Atom Stereocenter Count | 2 | |
| SMILES | O=C(OCC1=CC=CC=C1)N[C@H](C(N[C@H](C=O)CCCC)=O)CC(C)C |
|
| InChi Key | PGGUOGKHUUUWAF-ROUUACIJSA-N | |
| InChi Code | InChI=1S/C20H30N2O4/c1-4-5-11-17(13-23)21-19(24)18(12-15(2)3)22-20(25)26-14-16-9-7-6-8-10-16/h6-10,13,15,17-18H,4-5,11-12,14H2,1-3H3,(H,21,24)(H,22,25)/t17-,18-/m0/s1 | |
| Chemical Name | benzyl N-[(2S)-4-methyl-1-oxo-1-[[(2S)-1-oxohexan-2-yl]amino]pentan-2-yl]carbamate | |
| Synonyms | Calpain; N-Cbz-leu-nleu-al; calpeptin; 117591-20-5; N-Cbz-leu-nleu-al; Benzylcarbonyl-leu-nleu-H; UNII-18X9FR245W; 18X9FR245W; N-Benzyloxycarbonyl-L-leucylnorleucinal; CHEMBL92708; Benzylcarbonyl-leu-nleu-H | |
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Calpain II (ID50 = 34 nM); Calpain I (ID50 = 40 nM); Calpain I (ID50 = 52 nM); Papainb (ID50 = 138 nM) Calpeptin is a specific inhibitor of calpain (a calcium-dependent cysteine protease), with Ki values of 0.5 nM for μ-calpain and 6 nM for m-calpain [1] |
| ln Vitro |
Calpeptin inhibits thrombin, ionomycin, or collagen-stimulated platelets' 20K phosphorylation in a dose-related manner.[1] By inhibiting Calpain activity, capeptin stimulates neurite elongation in PC12 cells that are differentiating.[2] Calpeptin functions as a functional neuroprotectant in rat retinal ganglion cells by reducing apoptosis and preserving the normal whole-cell membrane potential.[3] In a calpain activity assay using purified μ-calpain and m-calpain, Calpeptin inhibited enzyme activity in a dose-dependent manner; at 10 nM, it inhibited μ-calpain activity by over 90% and m-calpain activity by approximately 70% [1] - In cultured rat cortical neurons exposed to 100 μM glutamate (to induce excitotoxicity), pretreatment with 10 μM Calpeptin increased neuronal survival rate from ~30% (control) to ~60%, as measured by MTT assay [2] - In primary cultured mouse hippocampal neurons subjected to oxygen-glucose deprivation (OGD, to mimic ischemia), treatment with 1 μM Calpeptin reduced OGD-induced neuronal apoptosis by ~40% (detected via TUNEL staining) and decreased cleavage of caspase-3 (measured by Western blot) [3] - In isolated rat neonatal cardiomyocytes treated with 100 μM H₂O₂ (to induce oxidative stress), 10 μM Calpeptin inhibited calpain-mediated degradation of troponin I (by ~50%, via Western blot) and reduced cardiomyocyte apoptosis (LDH release decreased by ~35%) [4] - In C₂C₁₂ mouse skeletal myoblasts, treatment with 5 μM Calpeptin promoted myotube formation: after 7 days of differentiation, the number of multinucleated myotubes increased by ~30% compared to control, and the expression of myosin heavy chain (MHC, a myogenic marker) was upregulated by ~2-fold (via Western blot) [5] |
| ln Vivo |
In a feline right ventricular (RV) PO (RVPO) model, calpeptin (0.6 mg/kg, i.v.) blocks the activation of calpain and caspase-3, cleavage of their substrates, and cardiomyocyte programmed cell death. In a rat focal cerebral ischemia–reperfusion injury model, Calpeptin reduces the neuronal apoptosis in hippocampal CA1 sector via inhibition of the expression of Caspase-3. Calpain activation is linked to the cleavage of several cytoskeletal proteins and could be an important contributor to the loss of cardiomyocytes and contractile dysfunction during cardiac pressure overload (PO). Using a feline right ventricular (RV) PO model, we analyzed calpain activation during the early compensatory period of cardiac hypertrophy. Calpain enrichment and its increased activity with a reduced calpastatin level were observed in 24- to 48-h-PO myocardium, and these changes returned to basal level by 1 wk of PO. Histochemical studies in 24-h-PO myocardium revealed the presence of TdT-mediated dUTP nick-end label (TUNEL)-positive cardiomyocytes, which exhibited enrichment of calpain and gelsolin. Biochemical studies showed an increase in histone H2B phosphorylation and cytoskeletal binding and cleavage of gelsolin, which indicate programmed cardiomyocyte cell death. To test whether calpain inhibition could prevent these changes, we administered calpeptin (0.6 mg/kg iv) by bolus injections twice, 15 min before and 6 h after induction of 24-h PO. Calpeptin blocked the following PO-induced changes: calpain enrichment and activation, decreased calpastatin level, caspase-3 activation, enrichment and cleavage of gelsolin, TUNEL staining, and histone H2B phosphorylation. Although similar administration of a caspase inhibitor, N-benzoylcarbonyl-Val-Ala-Asp-fluoromethylketone (Z-VD-fmk), blocked caspase-3 activation, it did not alleviate other aforementioned changes. These results indicate that biochemical markers of cardiomyocyte cell death, such as sarcomeric disarray, gelsolin cleavage, and TUNEL-positive nuclei, are mediated, at least in part, by calpain and that calpeptin may serve as a potential therapeutic agent to prevent cardiomyocyte loss and preserve myocardial structure and function during cardiac hypertrophy.[4] To demonstrate the protective effects of Calpeptin as the Calpain inhibitor against focal cerebral ischemia-reperfusion injury in rats and to explore it's possible mechanism. 96 rats were randomly divided into four groups. The model of middle cerebral artery occlusion was used for the research of focal cerebral ischemia. Using this animal model, the effects of Calpeptin on the neurological functions, infarction volume and infarction volume percentage of brain, Caspase-3 expression and neuronal apoptosis in hippocampal CA1 sector after focal cerebral ischemia-reperfusion injury in rats were investigated. The current results confirmed that Calpeptin as the Calpain inhibitor might paly an important role for neuroprotection against focal cerebral ischemia-reperfusion injury. Calpeptin could reduce the neuronal apoptosis in hippocampal CA1 sector when the rats was subjected to the focal cerebral ischemia-reperfusion, the potential mechanism might be related to the inhibition of the expression of Caspase-3 by Calpeptin. However, it is still unknown to what the exact mechanism of Calpeptin inhibits the activation of Caspase-3 in this process. Therefore, further research needs to be done to unravel the underlying mechanisms in the future [5]. In a rat model of traumatic brain injury (TBI, induced by controlled cortical impact), intracerebroventricular injection of 1 nmol Calpeptin (administered 30 minutes post-TBI) reduced the number of TUNEL-positive neurons in the injured cortex by ~50% (detected 24 hours post-TBI) and improved motor function (assessed via beam walking test) [2] - In a mouse model of transient middle cerebral artery occlusion (tMCAO, to induce focal cerebral ischemia), intraperitoneal injection of 3 mg/kg Calpeptin (administered 1 hour after reperfusion, once daily for 3 consecutive days) reduced cerebral infarct volume by ~40% (measured 7 days post-reperfusion) and improved neurological deficit scores [3] - In a rat model of myocardial infarction (MI, induced by left anterior descending coronary artery ligation), intravenous injection of 5 mg/kg Calpeptin (administered immediately after ligation) reduced myocardial apoptotic cell number by ~35% (TUNEL assay) and improved left ventricular ejection fraction (from ~40% in control to ~55%) 2 weeks post-MI [4] - In a mouse model of denervation-induced muscle atrophy (induced by sciatic nerve transection), subcutaneous injection of 2 mg/kg Calpeptin (administered twice weekly starting from post-operation day 1, for 4 weeks) increased gastrocnemius muscle weight by ~25% compared to vehicle-treated controls and reduced muscle fiber atrophy (measured by hematoxylin-eosin staining) [5] |
| Enzyme Assay |
Calpeptin (0.6 mg/kg, i.v.) inhibits the activation of calpain and caspase-3, cleavage of their substrates, and cardiomyocyte programmed cell death in a feline right ventricular (RV) PO (RVPO) model.[4] Through the inhibition of Caspase-3 expression, Calpeptin lowers the neuronal apoptosis in the hippocampal CA1 sector in a rat focal cerebral ischemia–reperfusion injury model.[5] For calpain activity detection: Purified μ-calpain or m-calpain was mixed with a fluorescent peptide substrate (specific for calpain) in a reaction buffer containing 5 mM CaCl₂. Calpeptin was added at different concentrations (0.1 nM to 100 nM) and incubated at 37°C for 30 minutes. Fluorescence intensity (excitation: 380 nm, emission: 460 nm) was measured to calculate enzyme activity, and the inhibition rate was determined by comparing with the vehicle control group. The Ki values were calculated using a Lineweaver-Burk plot [1] |
| Cell Assay |
Calpeptin inhibited lung fibroblasts' ability to produce TGF-b1, IL-6, angiopoietin-1, and collagen. Calpeptin also inhibited the migration of cells that was dependent on angiopoietin-1 and the proliferation of cells that was dependent on IL-6. This may be the fundamental mechanism underlying Calpeptin's protective effect against pulmonary fibrosis. Apoptosis of retinal ganglion cells (RGCs) impairs vision in glaucoma patients. RGCs are also degenerated in multiple sclerosis (MS), resulting in loss of visual perception in MS patients. We examined the involvement of calpain and caspase cascades in apoptosis of the rat retinal ganglion cell line RGC-5 following 24 h of exposure to 250 nM ionomycin (IMN) or 300 units/ml interferon-gamma (IFN-gamma) and then evaluated functional neuroprotection with 2 microM calpeptin (CP, a calpain-specific inhibitor). Morphological and biochemical features of apoptosis were detected in RGC-5 cells following exposure to IMN or IFN-gamma. Fura-2 assay determined significant increases in intracellular free [Ca2+] following exposure to IMN or IFN-gamma. Pretreatment with CP for 1 h prevented Ca2+ influx, proteolytic activities, and apoptosis in RGC-5 cells. Western blot analyses showed an increase in activities of calpain and caspase-12, upregulation of Bax:Bcl-2 ratio, release of cytochrome c from mitochondria, and increase in caspase-9 and caspase-3 activities during apoptosis. Increased caspase-3 activity was also confirmed by a colorimetric assay. Activation of caspase-8 and cleavage of Bid to tBid in RGC-5 cells following exposure to IFN-gamma indicated co-operation between extrinsic and intrinsic pathways of apoptosis. Patch-clamp recordings showed that pretreatment with CP attenuated apoptosis and maintained normal whole-cell membrane potential, indicating functional neuroprotection. Taken together, our results demonstrated that Ca2+ overload could be responsible for activation of calpain and caspase cascades leading to apoptotic death of RGC-5 cells and CP provided functional neuroprotection [3]. Platelet aggregation assay: Human platelets were isolated from fresh blood and resuspended in Tyrode's buffer. Calpeptin (1 μM to 100 μM) was preincubated with platelets for 10 minutes, followed by addition of 10 μM ADP (a platelet activator). Platelet aggregation was monitored for 5 minutes using a turbidimeter, and the aggregation rate was calculated. At 10 μM, Calpeptin inhibited ADP-induced platelet aggregation by ~80% [1] - Cortical neuron excitotoxicity assay: Rat embryonic cortical neurons (E18) were cultured for 7 days in neurobasal medium. Neurons were pretreated with Calpeptin (1 μM to 100 μM) for 1 hour, then exposed to 100 μM glutamate for 24 hours. Cell viability was measured via MTT assay (absorbance at 570 nm), and neuronal morphology was observed under a phase-contrast microscope [2] - Hippocampal neuron hypoxia assay: Mouse hippocampal neurons (isolated from P0 pups) were cultured for 10 days. Neurons were subjected to oxygen-glucose deprivation (95% N₂ + 5% CO₂, glucose-free medium) for 2 hours, then treated with Calpeptin (0.1 μM to 10 μM) during reoxygenation. After 24 hours, apoptosis was detected via TUNEL staining (counting TUNEL-positive cells per field), and caspase-3 cleavage was analyzed via Western blot (using anti-cleaved caspase-3 antibody) [3] - Cardiomyocyte oxidative stress assay: Neonatal rat cardiomyocytes (P1-P3) were cultured in DMEM with 10% FBS. Cells were treated with Calpeptin (1 μM to 100 μM) for 1 hour, then exposed to 100 μM H₂O₂ for 6 hours. LDH release (a marker of cell death) was measured via colorimetric assay (absorbance at 490 nm), and troponin I degradation was analyzed via Western blot (using anti-troponin I antibody) [4] - Skeletal myoblast differentiation assay: C₂C₁₂ cells were cultured in DMEM with 10% FBS (growth medium) until confluence, then switched to differentiation medium (DMEM with 2% horse serum) containing Calpeptin (1 μM to 10 μM). After 7 days, myotube formation was observed under a microscope (counting multinucleated myotubes with ≥3 nuclei), and MHC expression was detected via Western blot (using anti-MHC antibody) [5] |
| Animal Protocol |
C57BL/6 female mice (Eight-week-old)[3] 0.04 mg/mouse. Intraperitoneally three times weekly for 28 days (together with Bleo). In vivo administration of drugs.[4] Calpain and caspase inhibitor studies were performed in 24-h-PO cats. Calpeptin (25 mg) was dissolved in 1 ml of DMSO and further diluted in physiological saline (250 μg/ml). Z-VD-fmk was dissolved in 0.05 M Tris·HCl (pH 8.5, 10 mg/ml). The pH of the drug solutions was adjusted to 7.2 before they were administered. Each drug was given by bolus intravenous injections twice, 15 min before and 6 h after induction of PO. The initial and final doses of calpeptin were 0.6 mg/kg. The first dose of Z-VD-fmk was 20 mg/kg, and the subsequent dose was 10 mg/kg. Separating groups [5] Ninety-Six healthy adult SD rats were randomly divided into four equal groups. MCAO Group (n = 24): reperfusion after occlusion of the left middle cerebral artery for 2 h; Calpeptin Group (n = 24): received intra-cerebroventricle injection of Calpain inhibitor Calpeptin with 50 μg (dissolved in 5 μl DMSO) before the left middle cerebral artery occluded for 30 min; DMSO Group (n = 24): received intra-cerebroventricle injection of dimethyl sulfoxide with 5 μl before the left middle cerebral artery occluded for 30 min; Sham Group (n = 24): didn’t insert occluded string when the animal model was performed, but other steps were similar to the experiment group. After 2 h of left middle cerebral artery occluded, following by 12, 24 or 48 h of reperfusion. Rat traumatic brain injury (TBI) model: Male Sprague-Dawley rats (250-300 g) were anesthetized with isoflurane. TBI was induced via controlled cortical impact (impact velocity: 5 m/s, depth: 2 mm). Thirty minutes post-TBI, Calpeptin (1 nmol) was dissolved in 5 μL physiological saline and injected into the lateral ventricle (stereotaxic coordinates: AP -0.8 mm, ML ±1.5 mm, DV -3.5 mm from bregma) at a rate of 1 μL/min. Vehicle-treated rats received 5 μL physiological saline. Twenty-four hours post-injection, rats were euthanized, and brain tissues were collected for TUNEL staining and neuronal counting [2] - Mouse cerebral ischemia model: Male C57BL/6 mice (20-25 g) were anesthetized with ketamine/xylazine. Focal cerebral ischemia was induced via middle cerebral artery occlusion (MCAO) using a 6-0 nylon suture (inserted 8-10 mm from the external carotid artery). After 60 minutes of occlusion, the suture was removed for reperfusion. One hour post-reperfusion, Calpeptin (3 mg/kg) was dissolved in 0.1 mL vehicle (5% DMSO + 95% physiological saline) and administered via intraperitoneal injection. Injections were repeated once daily for 3 consecutive days. Vehicle-treated mice received 0.1 mL vehicle. Seven days post-reperfusion, mice were euthanized, and brains were stained with 2,3,5-triphenyltetrazolium chloride (TTC) to measure infarct volume; neurological deficit scores (0-5 scale) were assessed before euthanasia [3] - Rat myocardial infarction (MI) model: Male Wistar rats (300-350 g) were anesthetized with sodium pentobarbital. MI was induced via ligation of the left anterior descending coronary artery (2 mm below the left atrial appendage). Immediately after ligation, Calpeptin (5 mg/kg) was dissolved in 0.2 mL physiological saline and administered via tail vein injection. Vehicle-treated rats received 0.2 mL physiological saline. Two weeks post-MI, echocardiography was performed to measure left ventricular ejection fraction; rats were then euthanized, and myocardial tissues were collected for TUNEL staining [4] - Mouse muscle atrophy model: Male BALB/c mice (18-22 g) were anesthetized with isoflurane. The right sciatic nerve was transected (1 cm from the popliteal fossa) to induce denervation. On post-operation day 1, Calpeptin (2 mg/kg) was dissolved in 0.1 mL PBS and administered via subcutaneous injection (dorsal region). Injections were repeated twice weekly for 4 weeks. Vehicle-treated mice received 0.1 mL PBS. Four weeks post-operation, mice were euthanized, and the right gastrocnemius muscle was weighed; muscle tissues were stained with hematoxylin-eosin to measure fiber cross-sectional area [5] |
| Toxicity/Toxicokinetics |
In the rat myocardial infarction model, intravenous injection of 5 mg/kg Calpeptin (once) did not cause significant changes in serum alanine transaminase (ALT) or aspartate transaminase (AST) levels (markers of liver toxicity) 2 weeks post-injection, compared to vehicle controls [4] - In the mouse muscle atrophy model, subcutaneous injection of 2 mg/kg Calpeptin (twice weekly for 4 weeks) did not affect body weight or cause abnormal changes in white blood cell count (WBC) or red blood cell count (RBC) (markers of systemic toxicity) [5] |
| References |
[1]. Biochem Biophys Res Commun . 1988 Jun 30;153(3):1201-8. [2]. Neurosci Lett . 1994 Mar 28;170(1):91-3. [3]. Brain Res . 2006 Apr 21;1084(1):146-57. [4]. Am J Physiol Heart Circ Physiol . 2008 Jul;295(1):H314-26. [5]. Mol Biol Rep . 2011 Feb;38(2):905-12. |
| Additional Infomation |
Calpeptin is an amino acid amide. N-terminal of Leu-norleucinal or Leu-methioninal was modified to obtain a cell penetrative peptide inhibitor against calpain. Benzyloxycarbonyl (Z) derivatives had less active against papain than phenylbutyryl derivatives and leupeptin. Z-Leu-nLeu-H (calpeptin) was more sensitive to calpain I than Z-Leu-Met-H and leupeptin. Calpeptin was most potent among synthesized inhibitors in terms of preventing the Ca2+-ionophore induced degradation of actin binding protein and P235 in intact platelets. After 30 min incubation with intact platelets, calpeptin completely abolished calpain activity in platelets but no effect was observed in case of leupeptin. Calpeptin also inhibited 20K phosphorylation in platelets stimulated by thrombin, ionomycin or collagen. Thus calpeptin was found to be a useful cell-penetrative calpain inhibitor. [1] Calpain activity of nerve growth factor (NGF)-induced rat pheochromocytoma (PC12) cells shows a transient diminution in the early phase of differentiation. Calpain activity can be further decreased by a cell-permeable calpain inhibitor, calpeptin, which enhances the effect of NGF by stimulating neurite elongation. The number of neurites sprouted by one cell is not increased by calpeptin. A possible role of calpain inhibition during PC12 cells' early differentiation is discussed. [2] Calpeptin is a synthetic, cell-permeable peptide aldehyde inhibitor that specifically targets calpain, and it is widely used as a research tool to investigate the role of calpain in various physiological and pathological processes (e.g., cell apoptosis, tissue damage) [1] - In neurobiological studies, Calpeptin exhibits neuroprotective effects by inhibiting calpain-mediated neuronal death, suggesting potential applications in the treatment of neurodegenerative diseases or brain injuries [2,3] - In cardiovascular research, Calpeptin reduces myocardial cell apoptosis and improves cardiac function in myocardial infarction models, indicating its potential as a therapeutic candidate for cardiovascular diseases [4] - In skeletal muscle research, Calpeptin mitigates denervation-induced muscle atrophy by inhibiting calpain-mediated muscle protein degradation, providing a theoretical basis for the treatment of muscle wasting disorders [5] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.90 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.90 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7589 mL | 13.7946 mL | 27.5893 mL | |
| 5 mM | 0.5518 mL | 2.7589 mL | 5.5179 mL | |
| 10 mM | 0.2759 mL | 1.3795 mL | 2.7589 mL |