Physicochemical Properties
| Molecular Formula | C31H43CLF3NO2 |
| Molecular Weight | 554.12700 |
| Exact Mass | 553.293 |
| CAS # | 586976-24-1 |
| Related CAS # | 586976-24-1; |
| PubChem CID | 10311918 |
| Appearance | White to light yellow solid powder |
| LogP | 10.716 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 38 |
| Complexity | 633 |
| Defined Atom Stereocenter Count | 0 |
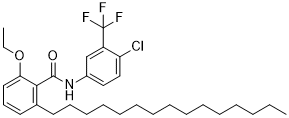
| SMILES | CCCCCCCCCCCCCCCC1=C(C(=CC=C1)OCC)C(=O)NC2=CC(=C(C=C2)Cl)C(F)(F)F |
| InChi Key | OYBMVMAXKOGYDC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C31H43ClF3NO2/c1-3-5-6-7-8-9-10-11-12-13-14-15-16-18-24-19-17-20-28(38-4-2)29(24)30(37)36-25-21-22-27(32)26(23-25)31(33,34)35/h17,19-23H,3-16,18H2,1-2H3,(H,36,37) |
| Chemical Name | N-[4-chloro-3-(trifluoromethyl)phenyl]-2-ethoxy-6-pentadecylbenzamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | CTPB is a synthetic HAT activator that increases the cleavage of H3 and H4 in nucleosomes to enhance activation [1]. |
| References |
[1]. Investigation of activation mechanism and conformational stability of N-(4-chloro-3-trifluoromethyl-phenyl)-2-ethoxybenzamide and N-(4-chloro-3-trifluoromethyl-phenyl)-2-ethoxy-6-pentadecyl-benzamidein the active site of p300 histone acetyl transferase enzyme by molecular dynamics and binding free-energy studies. J Biomol Struct Dyn. 2018 Oct 9:1-38. |
| Additional Infomation | CTPB is a benzamide obtained by formal condensation of the carboxy group of 2-ethoxy-6-pentadecylbenzoic acid with one of the amino groups of 4-chloro-3-(trifluoromethyl)aniline. It has a role as a histone acetyltransferase activator. It is a member of benzamides, an aromatic ether, a member of monochlorobenzenes and an organofluorine compound. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~62.5 mg/mL (~112.79 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.08 mg/mL (3.75 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.75 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (3.75 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8046 mL | 9.0232 mL | 18.0463 mL | |
| 5 mM | 0.3609 mL | 1.8046 mL | 3.6093 mL | |
| 10 mM | 0.1805 mL | 0.9023 mL | 1.8046 mL |