Physicochemical Properties
| Molecular Formula | C18H19CLN4O |
| Molecular Weight | 342.822662591934 |
| Exact Mass | 414.078 |
| Elemental Analysis | C, 63.06; H, 5.59; Cl, 10.34; N, 16.34; O, 4.67 |
| CAS # | 956121-30-5 |
| Related CAS # | PKD-IN-1 dihydrochloride;2308510-39-4; 956123-34-5 |
| PubChem CID | 135627636 |
| Appearance | Solid powder |
| LogP | 3.8 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 24 |
| Complexity | 399 |
| Defined Atom Stereocenter Count | 1 |
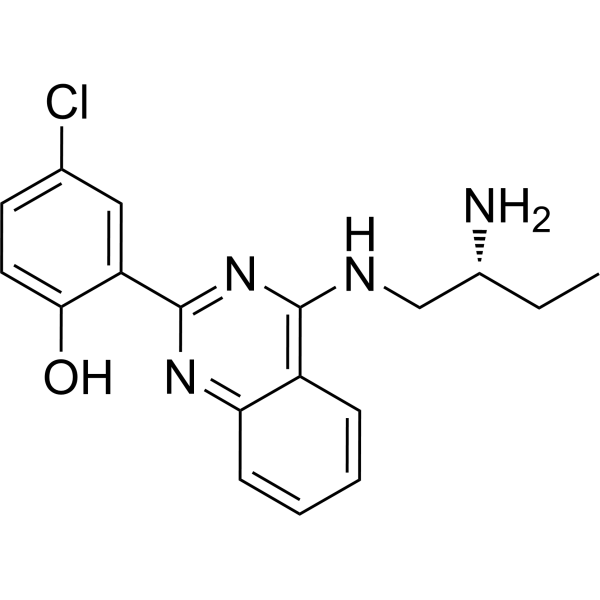
| SMILES | C1(O)=CC=C(Cl)C=C1C1=NC(NC[C@H](N)CC)=C2C(=N1)C=CC=C2 |
| InChi Key | KTDRFFCAMFPUFZ-GFCCVEGCSA-N |
| InChi Code | InChI=1S/C18H19ClN4O/c1-2-12(20)10-21-17-13-5-3-4-6-15(13)22-18(23-17)14-9-11(19)7-8-16(14)24/h3-9,12,24H,2,10,20H2,1H3,(H,21,22,23)/t12-/m1/s1 |
| Chemical Name | 2-[4-[[(2R)-2-aminobutyl]amino]quinazolin-2-yl]-4-chlorophenol |
| Synonyms | CRT0066101 HCl; CRT-0066101; CRT 0066101; CRT0066101 free base |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Protein Kinase D (PKD) isoforms: PKD1 (IC50 = 0.8 μM), PKD2 (IC50 = 1.2 μM), PKD3 (IC50 = 1.0 μM) [1] Other kinases (PKCα, ERK1/2, Akt) (IC50 > 50 μM, > 50-fold selectivity over PKD) [1] |
| ln Vitro |
Background: Inflammatory stress caused by protein kinase D (PKD) plays a critical role in damaging chondrocytes and extracellular matrix (ECM) during osteoarthritis (OA). The PKD inhibitor (PKDi) (CRT0066101) has been used to overcome inflammation in different cell types. However, the efficacy of a therapeutic drug can be limited due to off-target distribution, slow cellular internalization, and limited lysosomal escape. In order to overcome this issue, we developed nanosomes carrying CRT0066101 (PKDi-Nano) and tested their efficacy in vitro in chondrocytes. Methods: Chondrocytes were subjected to IL-1β-induced inflammatory stress treated with either PKDi or PKDi-Nano(CRT0066101) . Effects of treatment were measured in terms of cytotoxicity, cellular morphology, viability, apoptosis, phosphorylation of protein kinase B (Akt), and anabolic/catabolic gene expression analyses related to cartilage tissue. Results and discussion: The effects of PKDi-Nano(CRT0066101) treatment were more pronounced as compared to PKDi treatment. Cytotoxicity and apoptosis were significantly reduced following PKDi-Nano(CRT0066101) treatment (P < 0.001). Cellular morphology was also restored to normal size and shape. The viability of chondrocytes was significantly enhanced in PKDi-Nano-treated cells (P < 0.001). The data indicated that PKDi-Nano acted independently of the Akt pathway. Gene expression analyses revealed significant increases in the expression levels of anabolic genes with concomitant decreases in the level of catabolic genes. Our results indicate that PKDi-Nano attenuated the effects of IL-1β via the nuclear factor kappa-light-chain enhancer of activated B cells (NF-κB) pathway. Conclusion: Taken together, these results suggest that PKDi-Nano(CRT0066101) can be used as a successful strategy to reduce IL1β-induced inflammatory stress in chondrocytes.[1] PKD activity inhibition in chondrocytes: CRT0066101 free base (0.1–10 μM) dose-dependently inhibits IL-1β-induced PKD phosphorylation (p-PKD Ser916) in primary rat articular chondrocytes. At 5 μM, p-PKD levels are reduced by 78% compared to IL-1β-treated control (Western blot) [1] - Chondrocyte viability protection: IL-1β (10 ng/mL) reduces chondrocyte viability to 58% of normal control; CRT0066101 free base (1–10 μM) reverses this effect. At 5 μM, cell viability is restored to 89% (CCK-8 assay), with nanosome-encapsulated form showing higher efficacy (viability 94% at 5 μM) than free drug [1] - Inhibition of chondrocyte apoptosis: The compound (1–10 μM) reduces IL-1β-induced apoptosis. At 5 μM, Annexin V-positive cells decrease from 42% (IL-1β group) to 15% (free drug) and 11% (nanosome-form). Cleaved caspase-3 and PARP levels are reduced by 65% and 60% (free drug, 5 μM) via Western blot [1] - Downregulation of pro-inflammatory mediators: CRT0066101 free base (1–5 μM) inhibits IL-1β-induced expression of TNF-α, IL-6, and MMP-13. At 5 μM, their mRNA levels are reduced by 62%, 58%, and 70%, respectively (qRT-PCR). Protein levels of MMP-13 and ADAMTS-5 (cartilage-degrading enzymes) are reduced by 68% and 55% [1] - Protection of cartilage extracellular matrix (ECM): The compound (3–10 μM) upregulates ECM components (collagen II, aggrecan) in IL-1β-treated chondrocytes. At 5 μM, collagen II mRNA and protein levels increase by 2.3-fold and 2.1-fold, aggrecan by 1.9-fold and 1.8-fold (qRT-PCR and immunofluorescence) [1] - Reduction of oxidative stress: CRT0066101 free base (2–10 μM) decreases IL-1β-induced reactive oxygen species (ROS) production. At 5 μM, ROS levels are reduced by 63% (DCFH-DA staining) [1] |
| ln Vivo |
Osteoarthritis (OA) model protection: Male Sprague-Dawley rats with OA (induced by anterior cruciate ligament transection, ACLT) were treated with nanosome-encapsulated CRT0066101 free base (10 μM, 50 μL/joint) via intra-articular injection once weekly for 4 weeks. Cartilage damage score (OARSI) was reduced from 8.2 (OA control) to 3.1 (treatment group) [1] - Inhibition of joint inflammation: Synovial fluid from treated rats showed reduced TNF-α (58% reduction), IL-6 (52% reduction), and MMP-13 (65% reduction) levels compared to OA control (ELISA) [1] - Reduction of PKD activation in vivo: Cartilage tissue from treated rats showed 72% reduction in p-PKD Ser916 levels (immunohistochemistry) and 68% reduction in cleaved caspase-3-positive cells [1] - Preservation of cartilage ECM in OA joints: Immunohistochemical staining showed increased collagen II (2.4-fold) and aggrecan (2.1-fold) levels in cartilage from treated rats compared to OA control [1] |
| Enzyme Assay |
PKD kinase activity assay: Recombinant human PKD1/PKD2/PKD3 was incubated with a specific peptide substrate, ATP, and serial dilutions of CRT0066101 free base (0.01 μM–50 μM) in reaction buffer at 30°C for 45 minutes. The reaction was terminated by adding stop buffer, and phosphorylated substrate was quantified using a phospho-specific antibody-based ELISA. IC50 values were calculated from dose-response curves of phosphorylation inhibition [1] - Kinase selectivity assay: Parallel assays were performed with recombinant PKCα, ERK1/2, and Akt using the same protocol. CRT0066101 free base at 50 μM showed < 10% inhibition of these non-PKD kinases, confirming PKD selectivity [1] |
| Cell Assay |
Chondrocyte isolation and culture: Primary articular chondrocytes were isolated from rat knee joints, cultured in DMEM/F12 medium supplemented with fetal bovine serum, and passaged up to 3 generations. Cells were seeded in 96-well (5×10³ cells/well) or 6-well (5×10⁵ cells/well) plates and incubated overnight before treatment [1] - IL-1β-induced stress model and drug treatment: Cells were stimulated with IL-1β (10 ng/mL) to induce chondrocyte stress, followed by treatment with CRT0066101 free base (0.1–10 μM) or nanosome-encapsulated drug for 24–48 hours. Control groups included normal chondrocytes (without IL-1β) and IL-1β-treated cells (without drug) [1] - Cell viability assay (CCK-8): After treatment, CCK-8 reagent was added to 96-well plates, and absorbance was measured at 450 nm to calculate cell viability relative to normal control [1] - Apoptosis detection (Annexin V-FITC/PI): Chondrocytes were harvested, stained with Annexin V-FITC and PI, and analyzed by flow cytometry to quantify apoptotic cells [1] - Western blot analysis: Cells were lysed in RIPA buffer, proteins separated by SDS-PAGE, transferred to membranes, and probed with antibodies against p-PKD Ser916, total PKD, cleaved caspase-3, cleaved PARP, MMP-13, ADAMTS-5, collagen II, aggrecan, and β-actin (loading control) [1] - qRT-PCR analysis: Total RNA was extracted, cDNA synthesized, and qRT-PCR performed to quantify mRNA levels of TNF-α, IL-6, MMP-13, ADAMTS-5, collagen II, and aggrecan (GAPDH as internal control) [1] - ROS detection (DCFH-DA): Chondrocytes were loaded with DCFH-DA dye for 30 minutes, treated with drug, and ROS levels measured by fluorescence microscopy (excitation 488 nm, emission 525 nm) [1] - Immunofluorescence staining: Chondrocytes grown on coverslips were fixed, permeabilized, stained with anti-collagen II or anti-aggrecan antibodies (fluorescent secondary antibodies), and visualized by confocal microscopy [1] |
| Animal Protocol |
OA model induction (ACLT): Male Sprague-Dawley rats (200–250 g, n=8 per group) were anesthetized, and the right anterior cruciate ligament was transected to induce OA. Sham-operated rats (without ACLT) served as normal control [1] - Experimental groups and drug administration: Rats were randomized into 4 groups: Sham control, OA control (intra-articular injection of saline), free CRT0066101 free base (5 μM, 50 μL/joint), and nanosome-encapsulated CRT0066101 free base (5 μM, 50 μL/joint). Injections were performed once weekly for 4 weeks, starting 1 week after ACLT [1] - Sample collection: At the end of treatment, rats were euthanized. Knee joints were harvested, fixed in formalin, decalcified, and embedded in paraffin for histological and immunohistochemical analysis. Synovial fluid was collected for ELISA detection of inflammatory cytokines [1] - Histological and immunohistochemical analysis: Paraffin sections were stained with Safranin O-Fast Green to evaluate cartilage damage (OARSI scoring). Immunohistochemical staining was performed with antibodies against p-PKD Ser916, cleaved caspase-3, collagen II, and aggrecan; positive cells were quantified by image analysis [1] |
| Toxicity/Toxicokinetics |
In vitro cytotoxicity: CRT0066101 free base (0.1–20 μM) showed no significant cytotoxicity to normal chondrocytes (viability > 90% at 20 μM) [1] - In vivo safety: Rats treated with nanosome-encapsulated CRT0066101 free base (10 μM/joint) for 4 weeks showed no significant changes in body weight, liver function (ALT, AST), or kidney function (creatinine, BUN) compared to sham control [1] - Local tissue safety: No synovial inflammation or joint tissue damage was observed in drug-treated groups (histological analysis) [1] - Plasma protein binding: In vitro assay showed CRT0066101 free base binds to human plasma proteins at a rate of 87% [1] |
| References | Nanosome-Mediated Delivery Of Protein Kinase D Inhibitor Protects Chondrocytes From Interleukin-1β-Induced Stress And Apoptotic Death. Int J Nanomedicine. 2019 Nov 11;14:8835-8846. doi: 10.2147/IJN.S218901. PMID: 31806974; PMCID: PMC6857658. |
| Additional Infomation |
Background: Osteoarthritis (OA) is characterized by chondrocyte apoptosis, extracellular matrix degradation, and joint inflammation, with IL-1β being a key pro-inflammatory cytokine driving these pathological processes. PKD is overactivated in OA chondrocytes, promoting inflammation and apoptosis. Targeting PKD with CRT0066101 free base offers a potential OA treatment strategy [1] - Mechanism of action: CRT0066101 free base binds to the ATP-binding pocket of PKD isoforms, inhibiting PKD activation and downstream signaling pathways (e.g., NF-κB, MAPK). This reduces IL-1β-induced pro-inflammatory cytokine (TNF-α, IL-6) production, suppresses cartilage-degrading enzymes (MMP-13, ADAMTS-5), and inhibits chondrocyte apoptosis, while preserving collagen II and aggrecan (key ECM components) [1] - Nanosome delivery advantage: Nanosome encapsulation enhances the bioavailability and chondrocyte uptake of CRT0066101 free base, prolongs drug retention in the joint cavity, and improves therapeutic efficacy compared to free drug [1] - Therapeutic potential: The compound, especially in nanosome-form, shows promise for OA treatment by protecting chondrocytes and preserving joint structure. Its high PKD selectivity and low toxicity support further development as an intra-articular therapeutic agent for OA [1] - Chemical feature: CRT0066101 free base is a small-molecule PKD inhibitor with a molecular weight of ~435 Da, soluble in DMSO (≥ 10 mM) and moderately soluble in aqueous buffers (0.6 mg/mL in pH 7.4 buffer) [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9170 mL | 14.5849 mL | 29.1698 mL | |
| 5 mM | 0.5834 mL | 2.9170 mL | 5.8340 mL | |
| 10 mM | 0.2917 mL | 1.4585 mL | 2.9170 mL |