Physicochemical Properties
| Molecular Formula | C17H17N3O3 |
| Molecular Weight | 311.33518 |
| Exact Mass | 311.127 |
| CAS # | 178616-26-7 |
| PubChem CID | 4377504 |
| Appearance | Light yellow to khaki solid powder |
| Density | 1.32g/cm3 |
| Index of Refraction | 1.637 |
| LogP | 2.056 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 23 |
| Complexity | 460 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | MJKADKZSYQWGLL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H17N3O3/c1-22-14-7-11-8-16(21)19-20-17(13(11)9-15(14)23-2)10-3-5-12(18)6-4-10/h3-7,9H,8,18H2,1-2H3,(H,19,21) |
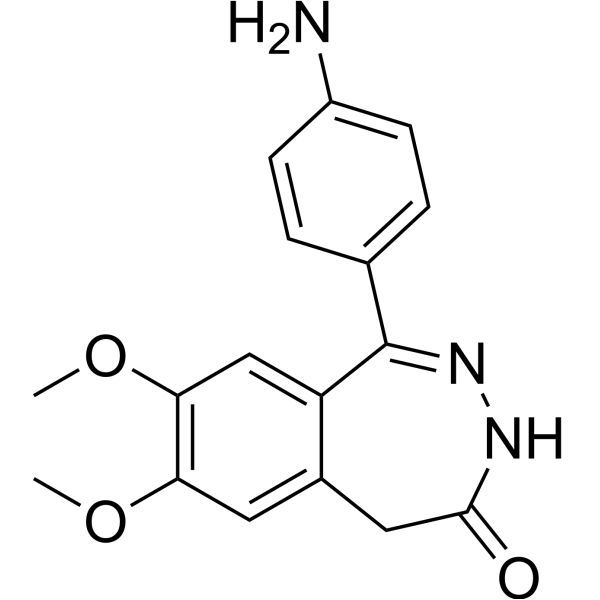
| Chemical Name | 1-(4-aminophenyl)-7,8-dimethoxy-3,5-dihydro-2,3-benzodiazepin-4-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | The extracellular signal-regulated kinase (ERK1/2) pathway is inhibited by CFM-2. CFM-2 upregulates tumor suppressor protein p21 and cell cycle regulatory factors while decreasing the phosphorylation of cAMP response element binding protein (CREB) and inhibiting the expression of cyclin D1. and p53 resulted in a drop in the proportion of lung adenocarcinoma cells in the G2 and S cell cycle stages. |
| ln Vitro | The extracellular signal-regulated kinase (ERK1/2) pathway is inhibited by CFM-2. CFM-2 upregulates tumor suppressor protein p21 and cell cycle regulatory factors while decreasing the phosphorylation of cAMP response element binding protein (CREB) and inhibiting the expression of cyclin D1. and p53 resulted in a drop in the proportion of lung adenocarcinoma cells in the G2 and S cell cycle stages. |
| ln Vivo |
CFM-2 pretreatment slows the advancement of seizure grade when pentylenetetrazole is administered repeatedly. The mean epilepsy score at the end of the 6-week repeated pentylenetetrazole treatment period was 0 in the vehicle-treated controls, 4.3 in the animals treated with vehicle + pentylenetetrazole (treated with CFM-2 (20 μmol/kg; peritoneal 2.2) + pentylenetetrazole in rats chronically treated (intravenous injection), and 1.0 in rats repeatedly treated with CFM-2 (50 μmol/kg; i.p.) + pentylenetetrazole). In pentylenetetrazole-kindled animals, CFM-2 can likewise counteract the long-term increase in sensitivity to the convulsive effects of GABA function inhibitors [1]. The ipsilateral hind paw's mechanical and thermal hypersensitivity was considerably reduced at two and twenty-four hours following CFA injection by intrathecal administration of two selective noncompetitive AMPAR antagonists, CFM-2 (25 and 50 μg) and GYKI 52466 (50 μg). hypersensitivity response. When subjected to mechanical and thermal stimulation, the contralateral basal response is unaffected by either GYKI 52466 or CFM-2 [4]. CFM-2 demonstrated anticonvulsant activity in the pentylenetetrazole (PTZ) kindling model in Sprague-Dawley rats. Pretreatment with CFM-2 (20 or 50 µmol kg⁻¹, i.p., 30 min before PTZ) delayed the progression of seizure severity during repeated PTZ administration (25 mg kg⁻¹, i.p., three times per week for 6 weeks). At the end of the 6-week period, the mean seizure score was 4.3 in vehicle+PTZ controls, 2.2 in rats treated with CFM-2 (20 µmol kg⁻¹) + PTZ, and 1.0 in rats treated with CFM-2 (50 µmol kg⁻¹) + PTZ. [1] CFM-2 antagonized the long-term increase in sensitivity to convulsant effects of GABA function inhibitors in PTZ-kindled animals. A challenge dose of PTZ (15 mg kg⁻¹, i.p.) 15 days after the end of chronic treatment induced seizures in 9 out of 10 vehicle+PTZ kindled rats, but only in 2 out of 8 rats from the CFM-2 (20 µmol kg⁻¹) + PTZ group and 1 out of 8 rats from the CFM-2 (50 µmol kg⁻¹) + PTZ group. Similarly, a challenge with picrotoxin (1.5 mg kg⁻¹, i.p.) 30 days after treatment induced seizures in all 10 vehicle+PTZ kindled rats but not in rats chronically treated with CFM-2 (20 or 50 µmol kg⁻¹). [1] No development of tolerance to the anticonvulsant effects of CFM-2 was observed following repeated administration in the PTZ kindling model. [1] |
| Animal Protocol |
For the pentylenetetrazole (PTZ) kindling study, Sprague-Dawley rats (250-280 g) were implanted with fronto-parietal screw electrodes under chloral hydrate anesthesia. After a recovery period of at least 7 days, the kindling phase began. PTZ (25 mg kg⁻¹, i.p.) was administered three times per week (Mondays, Wednesdays, Fridays) for up to 6 weeks. [1] CFM-2 was administered intraperitoneally at doses of 20 or 50 µmol kg⁻¹, 30 minutes before each PTZ injection during the chronic treatment period. Control rats received an equivalent volume of sterile saline. Seizure severity was scored for up to 60 minutes after PTZ administration based on a defined scale (0 = no seizure to 5 = generalized tonic seizures with hindlimb extension). Electrocortical activity was recorded. [1] For challenge experiments, 15 or 30 days after the end of the chronic PTZ treatment, animals were injected with a subconvulsant dose of PTZ (15 mg kg⁻¹, i.p.) or picrotoxin (1.5 mg kg⁻¹, i.p.) and observed for 1 hour for seizure incidence. [1] Motor function was assessed using a rotarod test. Rats were trained to remain on a rotating rod (4 cm diameter, 4.5 rpm) for 5 minutes. Impairment was defined as the inability to remain on the rod for the test period. Performance was assessed at 30, 60, and 90 minutes after acute CFM-2 administration, and also on days 1, 14, and 28 during chronic treatment, 30 minutes after drug injection. [1] The hydrochloride salt of CFM-2 was dissolved in sterile saline for administration. [1] |
| Toxicity/Toxicokinetics |
The TD₅₀ values (dose causing motor impairment in 50% of animals) for CFM-2 in the rotarod test were determined in Sprague-Dawley rats. Following acute administration, TD₅₀ values were 57.4 (48-68.7) µmol kg⁻¹ at 30 min, 46.9 (39.1-56.4) µmol kg⁻¹ at 60 min, and 94.3 (76.2-116.7) µmol kg⁻¹ at 90 min. After 28 days of repeated treatment, the TD₅₀ values were 58.8 (47.2-73.2) µmol kg⁻¹ at 30 min and 48.6 (38.4-61.5) µmol kg⁻¹ at 60 min post-injection. [1] The doses of CFM-2 (20 and 50 µmol kg⁻¹) used in chronic anticonvulsant studies did not induce observable adverse effects such as ataxia, decreased muscle tone, reduction in locomotor activity, or loss of righting reflex. [1] The therapeutic index (TI = TD₅₀ / ED₅₀ for clonic seizures) for CFM-2 is not explicitly calculated in the text, but the data suggest a separation between anticonvulsant and motor-impairing doses. [1] |
| References |
[1]. Effects of some AMPA receptor antagonists on the development of tolerance in epilepsy-prone rats and in pentylenetetrazole kindled rats. Eur J Pharmacol. 1999 Mar 5;368(2-3):149-59. [2]. Determination of new 2,3-benzodiazepines in rat plasma using high-performance liquid chromatography with ultraviolet detection. J Chromatogr B Biomed Sci Appl. 1999 Aug 20;731(2):207-15. [3]. AMPA antagonists inhibit the extracellular signal regulated kinase pathway and suppress lung cancer growth. Cancer Biol Ther. 2007 Dec;6(12):1908-15. [4]. Role of spinal cord alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors in complete Freund's adjuvant-induced inflammatory pain. Mol Pain. 2008 Dec 30;4:67. |
| Additional Infomation |
1-(4-aminophenyl)-7,8-dimethoxy-3,5-dihydro-2,3-benzodiazepin-4-one is a benzodiazepine. CFM-2 (1-(4'-aminophenyl)-3,5-dihydro-7,8-dimethoxy-4H-2,3-benzodiazepin-4-one hydrochloride) is a novel 2,3-benzodiazepine derivative acting as a non-selective AMPA receptor antagonist. [1] The study suggests that AMPA receptor antagonists like CFM-2 may have clinical potential for certain types of epilepsy, particularly because, unlike classical benzodiazepines, tolerance to their anticonvulsant effects did not develop upon repeated administration in the tested models. [1] The data indicate that activation of AMPA receptors is important for the behavioral and electrical expression of kindled seizures, and antagonism of these receptors can modulate the kindling process and the post-kindling state. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~160.60 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.75 mg/mL (8.83 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2119 mL | 16.0596 mL | 32.1192 mL | |
| 5 mM | 0.6424 mL | 3.2119 mL | 6.4238 mL | |
| 10 mM | 0.3212 mL | 1.6060 mL | 3.2119 mL |