Physicochemical Properties
| Molecular Formula | C18H16O4 |
| Molecular Weight | 296.322 |
| Exact Mass | 296.105 |
| Elemental Analysis | C, 72.96; H, 5.44; O, 21.60 |
| CAS # | 75464-11-8 |
| PubChem CID | 53271 |
| Appearance | Solid powder |
| Density | 1.324g/cm3 |
| Boiling Point | 490.1ºC at 760 mmHg |
| Flash Point | 264.3ºC |
| Index of Refraction | 1.641 |
| LogP | 3.143 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 22 |
| Complexity | 416 |
| Defined Atom Stereocenter Count | 0 |
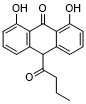
| SMILES | CCCC(=O)C1C2=C(C(=CC=C2)O)C(=O)C3=C1C=CC=C3O |
| InChi Key | AHZXFRRDQXXPJD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H16O4/c1-2-5-12(19)15-10-6-3-8-13(20)16(10)18(22)17-11(15)7-4-9-14(17)21/h3-4,6-9,15,20-21H,2,5H2,1H3 |
| Chemical Name | 9(10H)-Anthracenone, 1,8-dihydroxy-10-(1-oxobutyl)- |
| Synonyms | Butantrone; CCRIS2041; CCRIS-2041; CCRIS 2041 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References | 1: Männistö PT, Hanhijärvi H, Kosma VM, Collan Y. A 6-month dermal toxicity test with dithranol and butantrone in miniature swine. Contact Dermatitis. 1986 Jul;15(1):1-9. doi: 10.1111/j.1600-0536.1986.tb01253.x. PMID: 2944694. 2: Göransson A. Comparison of dithranol and butantrone in short contact therapy of psoriasis. Acta Derm Venereol. 1987;67(2):149-53. PMID: 2438880. 3: Hanhijärvi H, Nevalainen T, Männistö P. A six month dermal irritation test with anthralins in the Göttingen miniature swine. Arch Toxicol Suppl. 1985;8:463-8. doi: 10.1007/978-3-642-69928-3_107. PMID: 3913406. 4: Remitz A, Kariniemi AL, Lehtonen E, Nordling S. Inhibition of proliferation of HeLa cells by dithranol (anthralin) and 1o-butyryl dithranol (butantrone). Br J Dermatol. 1989 Apr;120(4):525-31. doi: 10.1111/j.1365-2133.1989.tb01326.x. PMID: 2730843. 5: Brandt H. Antipsoriatic activity of 10-acyl analogues of dithranol (anthralin). II. Clinical comparison of dithranol and butantrone sticks with special reference to side effects. Acta Derm Venereol. 1987;67(1):55-61. PMID: 2436415. 6: Remitz A. Short-contact therapy for psoriasis with 3.9% butantrone (10-butyryl dithranol). Acta Derm Venereol. 1989;69(4):351-4. PMID: 2568062. 7: Müller K, Gürster D. Hydroxyl radical damage to DNA sugar and model membranes induced by anthralin (dithranol). Biochem Pharmacol. 1993 Nov 17;46(10):1695-704. doi: 10.1016/0006-2952(93)90573-f. PMID: 8250955. 8: Viluksela M, Debets FM, van der Hoeven JC, Männistö PT. Dithranol (anthralin) and 10-butyryl dithranol (butantrone) do not morphologically transform cultured C3H 10T1/2 C18 mouse embryo fibroblasts. Acta Derm Venereol. 1994 Jan;74(1):54-6. doi: 10.2340/00015555745456. PMID: 7908485. 9: Göransson A. Irritation and staining by dithranol (anthralin) and butantrone (10-butyryl dithranol): further short contact and tape stripping experiments. Acta Derm Venereol. 1987;67(1):72-6. PMID: 2436419. 10: Viluksela M, Haasio K, Männistö PT. Studies on the contact sensitizing activity of dithranol (anthralin) and 10-butyryl dithranol (butantrone). Contact Dermatitis. 1990 Aug;23(2):103-10. doi: 10.1111/j.1600-0536.1990.tb03231.x. PMID: 2209005. 11: Männistö PT, Kirkland D, Viluksela M, Tikkanen L. Toxicological studies with dithranol and its 10-acyl analogues. Arch Toxicol. 1986 Oct;59(3):180-5. doi: 10.1007/BF00316330. PMID: 3813885. 12: Greaves MW. Irritation and staining by 10-butyryl dithranol (butantrone) in psoriasis. Int J Clin Pharmacol Res. 1986;6(4):315-6. PMID: 2428761. 13: Laakso S, Römer L, Mustakallio KK. Inhibition of soybean lipoxygenase-1 by 10-butyryl substituted 1,8-dihydroxy-9-anthrone (butantrone). Med Biol. 1986;64(1):43-5. PMID: 3088340. 14: Viluksela M, Puotunen E, Newman AJ, Männistö PT. Tumor-producing and skin- irritating activity of dithranol (anthralin) and its 10-acyl analogues in SENCAR mice. Carcinogenesis. 1986 Oct;7(10):1755-60. doi: 10.1093/carcin/7.10.1755. PMID: 3757176. 15: Göransson AM, Kolari PJ, Mustakallio KK. Irritation and staining by dithranol (anthralin) and related compounds. V. Short-contact and tape-stripping experiments with dithranol and butantrone. Acta Derm Venereol. 1984;64(2):134-9. PMID: 6203301. 16: Mustakallio KK, Brandt H. Antipsoriatic activity of 10-acyl analogues of dithranol (anthralin). I. Phase I clinical trial of 10-propionyl dithranol and 10-butyryl dithranol (butantrone). Acta Derm Venereol. 1984;64(1):63-6. PMID: 6203285. 17: Männistö PT, Vaissi L, Mustakallio KK, Viluksela M, Kosma VM, Collan Y. Tumor-producing activity of dithranol (anthralin) and two of its 10-acyl analogs in the dorsal skin of female NMRI mice. J Pharmacol Exp Ther. 1984 Apr;229(1):255-60. PMID: 6423812. 18: Männistö P, Havas A, Haasio K, Hanhijärvi H, Mustakallio K. Skin irritation by dithranol (anthralin) and its 10-acyl analogues in 3 animal models. Contact Dermatitis. 1984 Mar;10(3):140-5. doi: 10.1111/j.1600-0536.1984.tb00019.x. PMID: 6232099. 19: Rao K, Ch B, Narasu LM, Giri A. Antibacterial activity of Alpinia galanga (L) Willd crude extracts. Appl Biochem Biotechnol. 2010 Oct;162(3):871-84. doi: 10.1007/s12010-009-8900-9. Epub 2010 Apr 13. PMID: 20387130. 20: Mustakallio KK, Kolari PJ. Irritation and staining by dithranol (anthralin) and related compounds. IV. Visual estimation of erythema compared with contact thermometry and laser Doppler flowmetry. Acta Derm Venereol. 1983;63(6):513-8. PMID: 6198841. |
| Additional Infomation | Butantrone is the 10-butyryl derivative of dithranol with antipsoriatic activity. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3747 mL | 16.8737 mL | 33.7473 mL | |
| 5 mM | 0.6749 mL | 3.3747 mL | 6.7495 mL | |
| 10 mM | 0.3375 mL | 1.6874 mL | 3.3747 mL |