Physicochemical Properties
| Molecular Formula | C23H20FN5O3S |
| Molecular Weight | 465.5034 |
| Exact Mass | 465.127 |
| Elemental Analysis | C, 59.35; H, 4.33; F, 4.08; N, 15.05; O, 10.31; S, 6.89 |
| CAS # | 1259028-99-3 |
| PubChem CID | 49841251 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Index of Refraction | 1.652 |
| LogP | 2.97 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 33 |
| Complexity | 742 |
| Defined Atom Stereocenter Count | 0 |
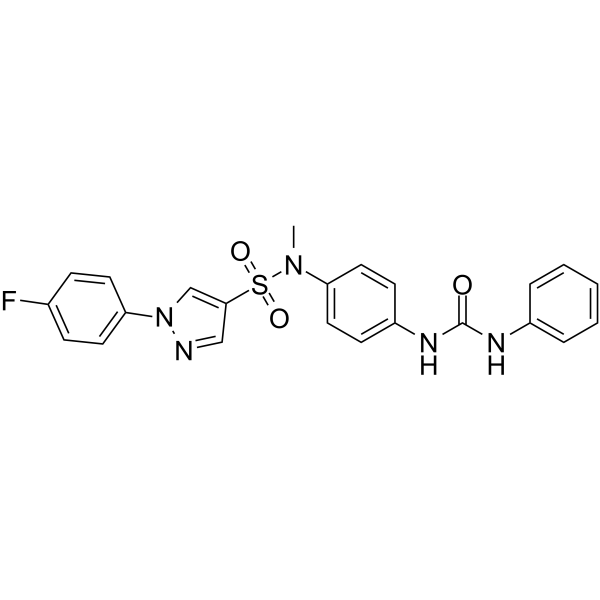
| SMILES | S(C1C=NN(C2C=CC(=CC=2)F)C=1)(N(C)C1C=CC(=CC=1)NC(NC1C=CC=CC=1)=O)(=O)=O |
| InChi Key | NSLIQOPYDUKWTA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H20FN5O3S/c1-28(33(31,32)22-15-25-29(16-22)21-11-7-17(24)8-12-21)20-13-9-19(10-14-20)27-23(30)26-18-5-3-2-4-6-18/h2-16H,1H3,(H2,26,27,30) |
| Chemical Name | 1-[4-[[1-(4-fluorophenyl)pyrazol-4-yl]sulfonyl-methylamino]phenyl]-3-phenylurea |
| Synonyms | BTT3033; BTT 3033; BTT-3033 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | BTT-3033 (1 nM-100 μM, 2 hours) is more selective for α2β1 than α3β1, α4β1, α5β1, and αv, and it reduces the adherence of CHO-α2wt cells to rat tail collagen I (EC50: 130 nM)[1]. With an EC50 value of 6 μM in mouse whole blood, BTT-3033 (10 μM, 5 minutes) inhibits human platelet binding to type I collagen-coated capillaries under flow [1]. Under shear stress conditions, CHO cells expressing α2 are unable to bind to type I collagen when exposed to BTT-3033 (10 μM) for five minutes [1]. Human prostate smooth muscle contractions generated by thromboxane A2 and neurogenic stimuli are inhibited by BTT-3033 (1 μM, 60 min) [3]. BTT-3033 (25 and 50 μM, 48 hours) causes G1 cell cycle arrest in LNcap-FGC and DU-145 cells, which decreases cell survival and proliferation [4]. Using a combination of caspase-3 activation, ΔΨm depletion, Bax protein overexpression, and ROS activation, BTT-3033 (50 μM) causes apoptosis in 48 hours [4]. In human articular cartilage-derived chondrocytes, BTT-3033 (10 μM, 15/28 days) promotes the expression of MMP1 and MT-MMP1 and suppresses the production of MMP13 [5]. |
| ln Vivo | In a mouse air sac model, BTT-3033 (orally, 10 mg/kg, 24 and 2 hours before PAF induction) demonstrated anti-inflammatory effects [2]. In an arachidonic acid-induced ear edema model, BTT-3033 (orally, 10 mg/kg, 48, 24 and 2 hours before ear swelling) demonstrated anti-inflammatory effects [2]. |
| Cell Assay |
Cell Viability Assay [4] Cell Types: LNcap-FGC and DU-145 Cell Tested Concentrations: 0.05, 0.5, 5, 25 and 50 μM Incubation Duration: 48 hrs (hours) Experimental Results: diminished cell viability at 25 μM and 50 μM. Cell viability assay [4] Cell Types: LNcap-FGC and DU-145 Cell Tested Concentrations: 5, 25 and 50 μM Incubation Duration: 48 hrs (hours) Experimental Results: Induced apoptosis by approximately 20%, 32% and 47% (LNcap-FGC) and 26%, 41%, and 59% at 5, 25, and 50 μM (DU-145). Western Blot Analysis [4] Cell Types: LNcap-FGC and DU-145 Cell Tested Concentrations: 25 μM Incubation Duration: 48 hrs (hours) Experimental Results: Result in downregulation of N-cadherin and upregulation of E-cadherin (EMT related protein). |
| Animal Protocol |
Animal/Disease Models: PAF (platelet activating factor)-induced mouse air sac model [2] Doses: 24 hrs (hrs (hours)) and 2 hrs (hrs (hours)) before PAF induction 1, 10 mg/kg Route of Administration: Oral Experimental Results:Leukocyte infiltration diminished by approximately 50% 10 mg/kg kilogram. Animal/Disease Models: Male DBA/1 mice (pharmacokinetic/PK/PK determination) [2] Doses: Single dose 10 mg/kg Route of Administration: Oral Experimental Results: Plasma level: Approximately 1 ng/mL 24 hrs (hrs (hours)) after administration. |
| References |
[1]. Novel α2β1 integrin inhibitors reveal that integrin binding to collagen under shear stress conditions does not require receptor preactivation. J Biol Chem. 2012 Dec 28;287(53):44694-702. [2]. Sulfonamide inhibitors of α2β1 integrin reveal the essential role of collagen receptors in in vivo models of inflammation. Pharmacol Res Perspect. 2015 Jun;3(3):e00146. [3]. Inhibition of neurogenic and thromboxane A 2 -induced human prostate smooth muscle contraction by the integrin α2β1 inhibitor BTT-3033 and the integrin-linked kinase inhibitor Cpd22. Prostate. 2020 Aug;80(11):831-849. [4]. Integrin α2β1 inhibition attenuates prostate cancer cell proliferation by cell cycle arrest, promoting apoptosis and reducing epithelial-mesenchymal transition. J Cell Physiol. 2021 Jul;236(7):4954-4965. [5]. Integrin α2β1 plays an important role in the interaction between human articular cartilage-derived chondrocytes and atelocollagen gel. Sci Rep. 2021 Jan 19;11(1):1757. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~537.06 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1482 mL | 10.7411 mL | 21.4823 mL | |
| 5 mM | 0.4296 mL | 2.1482 mL | 4.2965 mL | |
| 10 mM | 0.2148 mL | 1.0741 mL | 2.1482 mL |