Physicochemical Properties
| Molecular Formula | C41H56BCLF2N4O5 |
| Molecular Weight | 769.17 |
| Exact Mass | 768.4000351 |
| CAS # | 216571-88-9 |
| Appearance | Typically exists as solid at room temperature |
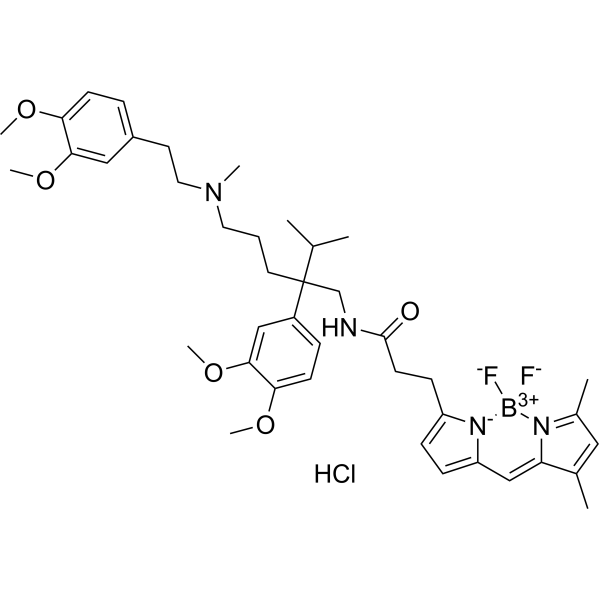
| SMILES | [B-]1(N2C(=CC(=C2C=C3[N+]1=C(C=C3)CCC(=O)NCC(CCCN(C)CCC4=CC(=C(C=C4)OC)OC)(C5=CC(=C(C=C5)OC)OC)C(C)C)C)C)(F)F.Cl |
| InChi Key | CVGLCXJYDRQISG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C41H55BF2N4O5.ClH/c1-28(2)41(32-12-17-37(51-7)39(25-32)53-9,20-10-21-46(5)22-19-31-11-16-36(50-6)38(24-31)52-8)27-45-40(49)18-15-33-13-14-34-26-35-29(3)23-30(4)47(35)42(43,44)48(33)34;/h11-14,16-17,23-26,28H,10,15,18-22,27H2,1-9H3,(H,45,49);1H |
| Chemical Name | 3-(2,2-difluoro-10,12-dimethyl-1-aza-3-azonia-2-boranuidatricyclo[7.3.0.03,7]dodeca-3,5,7,9,11-pentaen-4-yl)-N-[2-(3,4-dimethoxyphenyl)-5-[2-(3,4-dimethoxyphenyl)ethyl-methylamino]-2-propan-2-ylpentyl]propanamide;hydrochloride |
| Synonyms | 216571-88-9; orb2566798 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Fluorescent dye a (λ ex max: 503 nm, λ em max: 512 nm) [3] |
| ln Vitro |
To display binding sites, tissue that has been excised can be treated with BODIPY FL Verapamil HCl (10 μM) in phosphate buffer solution [1]. With IC50 values of 1.6-15.6 nM, BODIPY FL VerapamilHCl (0-100 nM) suppresses the efflux of ABCB1 and ABCC1 in combination with loxapine, mometasone, nicardipine, pimozide, and verapamil[2].
The uptake of BV was evaluated in drug sensitive and resistant cell lines as well as in clinical specimens from patients. Figure 1a shows results obtained with P388 and P388/ADR cells. A significantly higher ability to extrude the fluorescent dye was evident in the resistant counterpart, which is known to express high levels of P-glycoprotein. Similar results were obtained with the proximal tubular cell line LLC-PK1 and its resistant counterpart LLC-PK1/ADR (Fig. 1b). In P388 cells the uptake of BV was extremely rapid, and a plateau was reached within 15 min; in the resistant cell line, BV concentrations were, as expected, lower, and the equilibrium was reached even earlier, at 5 min (Fig. 2). We next studied the kinetics of BV efflux from P388/ADR cells. As shown in Fig. 3, the efflux was extremely rapid, and the process was almost completed in 15 min. The effect of reversing agents was evaluated on the intracellular concentrations of BV. The P-glycoprotein inhibitors verapamil and vinblastine were extremely effective in increasing the intracellular concentrations of the fluorescent compound in P388/ADR cells (Fig. 4a), which express high levels of P-glycoprotein, but not in the parental line P388 (data not shown). In addition, vinblastine was also active in reducing BV efflux from preloaded P388/ADR cells (Fig. 3).[3] |
| Cell Assay | Uptake of BODIPY FL Verapamil (BV) LLC-PK1, LLC-PK1/ADR, P388, P388/ADR and PANC-1 cells (5 × 105 cells/0.5 ml) were incubated in triplicate with increasing concentrations of BV (Molecular Probes, USA) at 37◦C for different experimental times in culture medium. After incubation, ice cold PBS was added, and the cells were centrifuged at 150×g for 5 min at 4◦C. The pellet was resuspended in 0.5 ml PBS and immediately subjected to FACS analysis. Leukemic cells (2.5 × 106) from patients were incubated in RPMI-1640 medium (Biochrom KG, Germany) at 37◦C with 5% CO2 for 15 min with 0.125 µM BV. After two washes in cold PBS, cells were kept on ice and immediately analyzed by FACS. [3] |
| References |
[1]. Histological demonstration of voltage dependent calcium channels on calcitonin gene-related peptide-immunoreactive nerve fibres in the mouse knee joint. Neurosci Lett. 2001 Oct 26;312(3):133-6. [2]. Fluorescent substrates for flow cytometric evaluation of efflux inhibition in ABCB1, ABCC1, and ABCG2 transporters. Anal Biochem. 2013 Jun 1;437(1):77-87. [3]. Bodipy-FL-Verapamil: A Fluorescent Probe for the Study of Multidrug Resistance Proteins. Cell Oncol. 2004;26(1-2):3-11. |
| Additional Infomation | Most of the substances used as fluorescent probes to study drug transport and the effect of efflux blockers in multidrug resistant cells have many drawbacks, such as toxicity, unspecific background, accumulation in mitochondria. New fluorescent compounds, among which Bodipy‐FL‐verapamil (BV), have been therefore proposed as more useful tools. The uptake of BV has been evaluated by cytofluorimetry and fluorescence microscopy using cell lines that overexpress P‐glycoprotein (P388/ADR and LLC‐PK1/ADR) or MRP (multidrug resistance‐related protein) (PANC‐1) and clinical specimens from patients. The effect of specific inhibitors for P‐glycoprotein (verapamil and vinblastine) or MRP (MK571 and probenecid) has been also studied. BV intracellular concentrations were significantly lower in the two P‐glycoprotein overexpressing cell lines in comparison with the parental lines. In addition, verapamil and vinblastine increased the intracellular concentrations of the dye; MK571 and probenecid, two MRP inhibitors, increased BV levels in PANC‐1 cells, that express this protein. These findings were confirmed in clinical specimens from patients. Fluorescence microscopy revealed a faint fluorescence emission in P‐glycoprotein or MRP expressing cell lines; however, treatment with specific inhibitors significantly increased the fluorescence. BV is a useful tool for studying multidrug resistance proteins with different techniques such as cytofluorimetry and fluorescence microscopy, but does not discriminate between P‐glycoprotein and MRP. In comparison with other classic fluorescent probes, the assay with this dye is extremely rapid, simple, not toxic for cells, devoid of fluorescent background, and can be useful in the clinical settings. [3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3001 mL | 6.5005 mL | 13.0010 mL | |
| 5 mM | 0.2600 mL | 1.3001 mL | 2.6002 mL | |
| 10 mM | 0.1300 mL | 0.6501 mL | 1.3001 mL |