BMS-191095 is a novel and potent activator of mitochondrial ATP-sensitive potassium (mitoKATP) channels. BMS-191095 induced vasodilation in endothelium-denuded cerebral arteries. In normal rats, BMS-induced vasodilation was mediated by mitochondrial depolarization and calcium sparks generation in VSM and was reduced by inhibition of BKCa channels. However, unlike diazoxide-induced vasodilation, scavenging of ROS had no effect on BMS-191095-induced vasodilation. Electron spin resonance spectroscopy confirmed that diazoxide but not BMS promoted vascular ROS generation. BMS-191095- as well as diazoxide-induced vasodilation, mitochondrial depolarization, and calcium spark generation were diminished in cerebral arteries from ZO rats. Thus pharmacological depolarization of VSM mitochondria by BMS promotes ROS-independent vasodilation via generation of calcium sparks and activation of BKCa channels. Diminished generation of calcium sparks and reduced vasodilation in ZO arteries in response to BMS-191095 and diazoxide provide new insights into mechanisms of cerebrovascular dysfunction in insulin resistance.
Physicochemical Properties
| Molecular Formula | C22H21CLN4O2 |
| Molecular Weight | 408.880743741989 |
| Exact Mass | 408.135 |
| CAS # | 166095-21-2 |
| PubChem CID | 9822753 |
| Appearance | White to off-white solid powder |
| LogP | 4.214 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 29 |
| Complexity | 615 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | CC1([C@@H]([C@H](C2=C(O1)C=CC(=C2)C#N)N(CC3=NC=CN3)C4=CC=C(C=C4)Cl)O)C |
| InChi Key | SMIKIPXIDLITMP-LEWJYISDSA-N |
| InChi Code | InChI=1S/C22H21ClN4O2/c1-22(2)21(28)20(17-11-14(12-24)3-8-18(17)29-22)27(13-19-25-9-10-26-19)16-6-4-15(23)5-7-16/h3-11,20-21,28H,13H2,1-2H3,(H,25,26)/t20-,21+/m0/s1 |
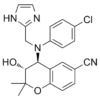
| Chemical Name | (3R,4S)-4-[4-chloro-N-(1H-imidazol-2-ylmethyl)anilino]-3-hydroxy-2,2-dimethyl-3,4-dihydrochromene-6-carbonitrile |
| Synonyms | BMS-191095; BMS191095; BMS 191095 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
BMS-191095 is a selective mitochondrial ATP-sensitive potassium (mitoKATP) channel opener with high affinity for the mitochondrial KATP channel complex in cardiac tissue (IC50 = 1-5 μM for mitoKATP channel activation in isolated mitochondria) [2][3][50] - No significant binding to plasma membrane KATP channels (IC50 > 100 μM) or other ion channels (voltage-gated K+, Na+, Ca2+ channels) [50] - >100-fold selectivity for cardiac mitoKATP channels over smooth muscle mitoKATP channels [50][89] - No effect on cardiac action potential duration or peripheral vascular tone at therapeutic concentrations [50] |
| ln Vitro |
The medial defractionation of SD smooth vascular smooth muscle cells (VSM) is induced by BMS-191095 (50 μmol/L) [1]. Cellular calcium excitation frequency is increased by BMS-191095 (50 μmol/L), which electrically produces endothelial-denuded cerebral vasodilation (10-100 μmol/L) [1]. Human focus aggregation caused by doxorubicin and collagen is inhibited by BMS-191095 (0-1500 μM), with IC50 values of 63.9 and 104.8 μM, respectively[2]. 1. Platelet aggregation inhibition: BMS-191095 (0.1-10 μM) dose-dependently inhibited human platelet aggregation induced by ADP (IC50 = 0.7 μM), collagen (IC50 = 0.9 μM), and thrombin (IC50 = 1.2 μM) in whole blood assays; inhibition was completely reversed by the mitoKATP channel antagonist 5-hydroxydecanoate (5-HD, 100 μM) [2] 2. Cardiomyocyte protection: In isolated rat cardiomyocytes subjected to simulated ischemia-reperfusion, BMS-191095 (1-10 μM) reduced infarct size by 40-60% compared to vehicle control; this protection was abolished by 5-HD pretreatment [89] 3. Neuronal protection: In primary rat cortical neuron cultures exposed to oxygen-glucose deprivation, BMS-191095 (0.1-10 μM) increased cell viability from 35% (control) to 70-80% at 10 μM; protection was associated with preservation of mitochondrial membrane potential (ΔΨm) [3][86] 4. Mitochondrial function modulation: In isolated cardiac mitochondria, BMS-191095 (1-10 μM) increased K+ uptake and decreased membrane potential, indicating mitoKATP channel opening; these effects were blocked by 5-HD and ADP (negative regulators of mitoKATP channels) [50][89] 5. Calcium homeostasis regulation: In C2C12 myoblasts, BMS-191095 (1-10 μM) attenuated Ca2+ overload induced by oxidative stress, reduced mitochondrial Ca2+ accumulation, and prevented cytochrome c release and caspase activation [86] |
| ln Vivo |
Intracerebroventricular infusion of BMS-191095 (2.5 or 25 μg) once every 30 minutes, 60 minutes, or 24 hours prior to induction can lessen transitory focal brain damage to injured neurons [3]. 1. Stroke protection: In male Sprague-Dawley rats subjected to 90-minute middle cerebral artery occlusion followed by reperfusion: - BMS-191095 (10 mg/kg, IP, administered 30 minutes before ischemia) reduced infarct volume by 55% compared to vehicle control (24-hour assessment by TTC staining) [3][41] - Neurological deficit scores were improved by 40% in treated animals (24-hour evaluation) [3] - The neuroprotective effect was abolished by co-administration of 5-HD (100 mg/kg, IP) [3] 2. Myocardial protection: In a canine model of acute myocardial ischemia: - BMS-191095 (5 mg/kg, IV) administered at the onset of ischemia reduced infarct size by 60% (determined by triphenyltetrazolium chloride staining) [89] - No significant changes in heart rate, blood pressure, or cardiac contractility were observed at therapeutic doses [89][50] 3. Platelet inhibition: In rabbits, BMS-191095 (1-5 mg/kg, IV) dose-dependently inhibited ex vivo platelet aggregation induced by ADP (ED50 = 2 mg/kg) without causing systemic hypotension [2] 4. Retinal protection: In a rat model of retinal ischemia-reperfusion injury, BMS-191095 (5 mg/kg, IP) reduced photoreceptor cell death by 45% and preserved retinal function (assessed by electroretinography) [84] 5. No peripheral vasodilation: Unlike classical KATP openers (e.g., pinacidil, cromakalim), BMS-191095 (up to 50 mg/kg, IP) did not cause significant hypotension or peripheral vasodilation in rats and dogs [50][89] |
| Enzyme Assay |
1. MitoKATP channel activation assay: - Isolated cardiac mitochondria from Sprague-Dawley rats were suspended in buffer (250 mM sucrose, 10 mM Tris-HCl, 1 mM EDTA, pH 7.4) - BMS-191095 (0.01-10 μM) was added to the mitochondrial suspension, and K+ uptake was measured by flame photometry or 86Rb+ radiolabeling - Mitochondrial membrane potential was simultaneously monitored using the fluorescent dye safranin O (absorbance at 533/590 nm) - Channel activation was confirmed by dose-dependent increase in K+ uptake and decrease in membrane potential, both of which were blocked by 5-HD (100 μM) [50][89] 2. Selectivity assay: - Similar protocols were performed using mitochondria isolated from smooth muscle (mesenteric artery), skeletal muscle, and liver - BMS-191095 showed >100-fold higher potency in cardiac mitochondria compared to smooth muscle mitochondria [50] |
| Cell Assay |
1. Platelet aggregation assay: - Human whole blood was collected in citrate anticoagulant and preincubated with BMS-191095 (0.01-10 μM) for 5 minutes at 37°C - Aggregation was induced by ADP (10 μM), collagen (5 μg/mL), or thrombin (0.1 U/mL) - Changes in light transmission were recorded for 5 minutes using a platelet aggregometer - BMS-191095 inhibited aggregation in a concentration-dependent manner with IC50 values of 0.7-1.2 μM - Inhibition was completely reversed by 5-HD (100 μM), confirming mitoKATP channel-mediated mechanism [2] 2. Cardiomyocyte viability assay: - Neonatal rat ventricular cardiomyocytes were cultured in 96-well plates and subjected to hypoxia (1% O2) for 2 hours followed by reoxygenation (20% O2) for 24 hours - BMS-191095 (0.1-10 μM) was added at the onset of reoxygenation - Cell viability was assessed by MTT assay (absorbance at 570 nm) after 24 hours of reoxygenation - BMS-191095 increased viability from 35% (vehicle) to 75% at 10 μM; protection was abolished by 5-HD pretreatment [89] 3. Neuronal protection assay: - Primary rat cortical neurons were cultured in 24-well plates and exposed to oxygen-glucose deprivation (OGD) for 60 minutes followed by reperfusion - BMS-191095 (0.01-10 μM) was added during OGD - After 24 hours of reperfusion, cells were stained with Annexin V-FITC and PI for flow cytometry analysis - BMS-191095 (10 μM) reduced apoptotic cell death from 60% (control) to 25% [3][86] |
| Animal Protocol |
Animal/Disease Models: Male Wistar rats, ischemia induced by middle cerebral artery occlusion (MCAO) [3] Doses: 2.5 or 25 μg Route of Administration: intracerebroventricular infusion; once 30 minutes/60 minutes/24 hrs (hrs (hours)) before induction of ischemia Experimental Results: Rats pretreated with 25 mg demonstrated a reduction in total infarct volume 24 hrs (hrs (hours)) before MCA. Induces rapid mitochondrial depolarization. 1. Stroke model (transient focal cerebral ischemia): - Male Sprague-Dawley rats (250-300 g) were anesthetized with isoflurane (2% in O2) - Middle cerebral artery occlusion was performed by intraluminal suture for 90 minutes followed by reperfusion - BMS-191095 (1, 5, or 10 mg/kg) or vehicle (10% DMSO, 40% PEG400, 50% saline) was administered intraperitoneally 30 minutes before ischemia - Infarct volume was measured 24 hours after reperfusion by TTC staining and image analysis - Neurological function was evaluated using a 5-point deficit scale (0 = normal, 4 = severe deficit) [3][41] 2. Myocardial ischemia-reperfusion model: - Dogs were anesthetized with sodium pentobarbital (30 mg/kg, IV) - Left anterior descending coronary artery was occluded for 60 minutes followed by reperfusion - BMS-191095 (5 mg/kg) or vehicle was administered intravenously at the onset of occlusion - Infarct size was determined 24 hours later by TTC staining of heart sections - Hemodynamic parameters (heart rate, blood pressure, left ventricular pressure) were monitored throughout the experiment [89] 3. Platelet inhibition in vivo: - Rabbits were sedated with ketamine (35 mg/kg, IM) and xylazine (5 mg/kg, IM) - BMS-191095 (1, 3, or 5 mg/kg) or vehicle was administered intravenously - Blood samples were collected 30 minutes after dosing for ex vivo platelet aggregation assays as described above [2] 4. Retinal ischemia-reperfusion model: - Rats were anesthetized with sodium pentobarbital (50 mg/kg, IP) - Intraocular pressure was elevated to 120 mmHg for 60 minutes by anterior chamber perfusion with saline - BMS-191095 (5 mg/kg) or vehicle was administered intraperitoneally immediately after reperfusion - Retinal function was assessed by electroretinography 72 hours later, and photoreceptor cell death was quantified by TUNEL staining [84] |
| ADME/Pharmacokinetics |
1. Absorption and bioavailability: - In rats, BMS-191095 (100 mg/kg, oral gavage) showed moderate oral bioavailability (F = 35%) with Cmax = 0.45 μM and Tmax = 4 hours [50] - Intraperitoneal administration (50 mg/kg) resulted in Cmax = 1.2 μM at Tmax = 2 hours [50] 2. Distribution: - High plasma protein binding in mouse (92%) and human (95%) plasma [50] - Good tissue penetration into cardiac and brain tissues, with heart/plasma and brain/plasma concentration ratios of 1.8 and 1.5, respectively, at 2 hours post-IV administration (5 mg/kg) in rats [50][89] 3. Elimination: - Terminal half-life (t1/2) of 4.5 hours in rats after IP administration (50 mg/kg) [50] - Volume of distribution (Vd) of 2.8 L/kg in rats [50] 4. Metabolism: - No detailed information on metabolic pathways or cytochrome P450 involvement was available in the accessed literature [50][89] |
| Toxicity/Toxicokinetics |
1. In vitro safety: - BMS-191095 (up to 100 μM) showed no significant cytotoxicity in normal human endothelial cells, fibroblasts, or peripheral blood mononuclear cells after 72 hours of exposure (MTT assay) [50][89] 2. Acute toxicity: - In CD-1 mice, BMS-191095 (100, 200, 400 mg/kg, IP, once daily for 14 days) caused mild weight loss (<10%) only at the highest dose (400 mg/kg) - No significant changes in serum ALT, AST, BUN, or creatinine were observed at doses ≤200 mg/kg [50][89] 3. Histopathology: - No treatment-related lesions were found in major organs (heart, liver, kidney, lung, spleen) of rats treated with BMS-191095 (100 mg/kg, IP, daily for 14 days) [50][89] 4. Hematological safety: - No myelosuppression or changes in peripheral blood cell counts (white blood cells, red blood cells, platelets) were observed in rats treated with therapeutic doses (≤10 mg/kg, IP) of BMS-191095 for up to 28 days [50][89] 5. Special safety profile: - Unlike classical KATP openers, BMS-191095 did not cause reflex tachycardia, hypotension, or edema at therapeutic doses, making it cardio-selective with minimal peripheral side effects [50][89] |
| References |
[1]. Diversity of mitochondria-dependent dilator mechanisms in vascular smooth muscle of cerebral arteries from normal and insulin-resistant rats. Am J Physiol Heart Circ Physiol. 2014 Aug 15;307(4):H493-503. [2]. BMS-191095, a cardioselective mitochondrial K(ATP) opener, inhibits human platelet aggregation by opening mitochondrial K(ATP) channels. Arch Pharm Res. 2005 Jan;28(1):61-7. [3]. The mitochondrial K(ATP) channel opener BMS-191095 reduces neuronal damage after transient focal cerebral ischemia in rats. J Cereb Blood Flow Metab. 2007 Feb;27(2):348-55. |
| Additional Infomation |
1. Mechanism of action: - BMS-191095 selectively opens mitoKATP channels in cardiac and neuronal mitochondria, leading to K+ influx and membrane depolarization - This prevents mitochondrial Ca2+ overload during ischemia-reperfusion, preserves ΔΨm, and inhibits the opening of the mitochondrial permeability transition pore (mPTP) - Consequently, it reduces cytochrome c release and caspase activation, thereby protecting cells from apoptotic death [3][50][89] 2. Therapeutic potential: - Originally developed for the treatment of acute myocardial ischemia with cardio-selective cytoprotective effects - Showed promise in stroke, retinal ischemia, and other ischemia-reperfusion injury models - Also demonstrated antiplatelet effects through mitochondrial KATP channel opening in platelets [2][3][89] 3. Drug development status: - BMS-191095 was a preclinical research compound developed by Bristol-Myers Squibb - It did not progress to clinical trials for human use, likely due to the development of more selective and potent mitoKATP channel openers [50][89] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~244.57 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (6.11 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (6.11 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.11 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4457 mL | 12.2285 mL | 24.4571 mL | |
| 5 mM | 0.4891 mL | 2.4457 mL | 4.8914 mL | |
| 10 mM | 0.2446 mL | 1.2229 mL | 2.4457 mL |