BI-749327 (BI749327) is a novel, potent and orally bioavailable antagonist of Transient receptor potential canonical type 6 (TRPC6) with the potential to be used for treating PAH (pulmonary arterial hypertension) and PH (pulmonary hypertension) due to respiratory diseases or hypoxemia. It inhibits TRPC6 with an IC50 of 13 nM in mice. BI-749327 exhibits 85- and 42-fold selectivity over the most closely related channels, TRPC3 and TRPC7. TRPC6 calcium conductance results in the stimulation of nuclear factor of activated T cells (NFAT) that triggers pathological cardiac and renal fibrosis and disease. BI 749327 suppresses NFAT activation in HEK293T cells expressing wild-type or gain-of-function TRPC6 mutants (P112Q, M132T, R175Q, R895C, and R895L) and blocks associated signaling and expression of prohypertrophic genes in isolated myocytes. In vivo, BI 749327 (30 mg/kg/day, yielding unbound trough plasma concentration ∼180 nM) improves left heart function, reduces volume/mass ratio, and blunts expression of profibrotic genes and interstitial fibrosis in mice subjected to sustained pressure overload. Additionally, BI 749327 dose dependently reduces renal fibrosis and associated gene expression in mice with unilateral ureteral obstruction. These results provide in vivo evidence of therapeutic efficacy for a selective pharmacological TRPC6 inhibitor with oral bioavailability and suitable pharmacokinetics to ameliorate cardiac and renal stress-induced disease with fibrosis.
Physicochemical Properties
| Molecular Formula | C23H21F3N4O2 |
| Molecular Weight | 442.433655500412 |
| Exact Mass | 442.161 |
| Elemental Analysis | C, 62.44; H, 4.78; F, 12.88; N, 12.66; O, 7.23 |
| CAS # | 2361241-23-6 |
| PubChem CID | 138377580 |
| Appearance | White to off-white solid powder |
| LogP | 3.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 32 |
| Complexity | 611 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | FC(C1C=CC(=CC=1)OC1C=CC(=CC=1)C(N1CCC(C2=CC=C(N)N=N2)CC1)=O)(F)F |
| InChi Key | RGYMFGHHIDRCBN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H21F3N4O2/c24-23(25,26)17-3-7-19(8-4-17)32-18-5-1-16(2-6-18)22(31)30-13-11-15(12-14-30)20-9-10-21(27)29-28-20/h1-10,15H,11-14H2,(H2,27,29) |
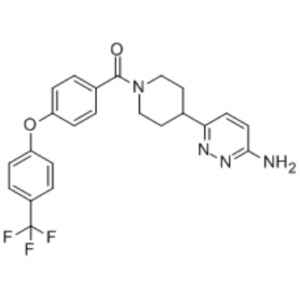
| Chemical Name | [4-(6-aminopyridazin-3-yl)piperidin-1-yl]-[4-[4-(trifluoromethyl)phenoxy]phenyl]methanone |
| Synonyms | BI749327; BI 749327; (4-(6-Aminopyridazin-3-yl)piperidin-1-yl)(4-(4-(trifluoromethyl)phenoxy)phenyl)methanone; BI 749327; [4-(6-aminopyridazin-3-yl)piperidin-1-yl]-[4-[4-(trifluoromethyl)phenoxy]phenyl]methanone; BI749327; SCHEMBL21274829; GTPL12530; BI-749327 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
mTRPC6 (IC50 = 13 nM); hTRPC6 (IC50 = 19 nM); guinea pig TRPC6 (IC50 = 15 nM)[1] BI-749327 is a potent and selective antagonist of the Transient Receptor Potential Canonical type 6 (TRPC6) channel. The half-maximal inhibitory concentration (IC₅₀) for mouse TRPC6 is 13 nM. It shows 85-fold selectivity over TRPC3 (IC₅₀ = 1,100 nM) and 42-fold selectivity over TRPC7 (IC₅₀ = 550 nM). It also exhibits >700-fold selectivity over TRPC5, and >500-fold selectivity over human TRPM8, TRPV1, TRPA1, and Nav1.5, as well as >150-fold selectivity over human Kv11.1 (hERG). [1] |
| ln Vitro |
In HEK293T cells expressing wild-type or gain-of-function TRPC6 mutants, BI-749327 reduces NFAT activation. It also prevents associated signaling and the production of pro-hypertrophic genes in isolated myocytes [1]. In this study, researchers report an orally bioavailable TRPC6 antagonist (BI 749327; IC50 13 nM against mouse TRPC6, t1/2 8.5-13.5 hours) with 85- and 42-fold selectivity over the most closely related channels, TRPC3 and TRPC7. TRPC6 calcium conductance results in the stimulation of nuclear factor of activated T cells (NFAT) that triggers pathological cardiac and renal fibrosis and disease. BI 749327 suppresses NFAT activation in HEK293T cells expressing wild-type or gain-of-function TRPC6 mutants (P112Q, M132T, R175Q, R895C, and R895L) and blocks associated signaling and expression of prohypertrophic genes in isolated myocytes. In HEK293T cells transfected with a Nuclear Factor of Activated T-cells (NFAT)-luciferase reporter and TRPC6, BI-749327 dose-dependently inhibited TRPC6-mediated NFAT activation, with 500 nM completely abrogating the signal. This effect was specific to TRPC6, as 500 nM BI-749327 had no impact on NFAT activity in cells transfected with empty vector or TRPC3, and only mild inhibition was observed with TRPC7. [1] BI-749327 (500 nM) also fully blocked NFAT activation induced by several gain-of-function TRPC6 mutants associated with human familial glomerulosclerosis (P112Q, M132T, R175Q, R895C, R895L). [1] In neonatal rat ventricular myocytes, stimulation with angiotensin II (Ang II, 100 nM for 48 h) upregulated mRNA expression of pro-hypertrophic genes (Nppa, Nppb, Myh7) and calcineurin/NFAT pathway genes (Rcan1, Trpc6). Co-treatment with BI-749327 (250 nM or 500 nM) attenuated this upregulation. BI-749327 alone did not alter the expression of these genes. [1] |
| ln Vivo |
BI-749327 (30 mg/kg/day; ig) improves left ventricular function, decreases volume/mass ratio, and attenuates the expression of profibrotic genes and interstitial fibrosis in mice with chronic pressure overload [1]. BI-749327 dose-dependently decreases renal fibrosis and associated gene expression in mice with unilateral ureteral obstruction [1]. BI-749327 has a lengthy terminal half-life (t1/2 8.5-13.5 hours) in mice (3-30 mg/kg; oral) [1]. In vivo, BI 749327 (30 mg/kg/day, yielding unbound trough plasma concentration ∼180 nM) improves left heart function, reduces volume/mass ratio, and blunts expression of profibrotic genes and interstitial fibrosis in mice subjected to sustained pressure overload. Additionally, BI 749327 dose dependently reduces renal fibrosis and associated gene expression in mice with unilateral ureteral obstruction. These results provide in vivo evidence of therapeutic efficacy for a selective pharmacological TRPC6 inhibitor with oral bioavailability and suitable pharmacokinetics to ameliorate cardiac and renal stress-induced disease with fibrosis[1]. In a mouse model of cardiac pressure overload induced by transverse aortic constriction (TAC), daily oral administration of BI-749327 (30 mg/kg/day) starting one week post-surgery gradually improved left ventricular fractional shortening compared to vehicle over 7 weeks. [1] Invasive pressure-volume analysis revealed that BI-749327 treatment significantly reduced both end-diastolic and end-systolic left ventricular volumes in both sham and TAC-operated mice, without altering heart rate, peak systolic pressure, or load-independent contractility indices (preload recruitable stroke work and end-systolic elastance). This suggests a reduction in cardiac preload. [1] BI-749327 treatment suppressed TAC-induced myocardial expression of fetal genes (Nppa, Nppb), fibrotic genes (Col1a2, Col3a2, Fn1, Mmp2, Tgfb1, Timp2), and the feedback regulator Rcan1. It also reduced Trpc6 mRNA levels in the heart. [1] Myocardial interstitial fibrosis, assessed by histology, was reduced by approximately 40% in TAC mice treated with BI-749327 compared to vehicle. [1] In a mouse model of renal fibrosis induced by unilateral ureteral obstruction (UUO), daily oral BI-749327 treatment (3, 10, 30 mg/kg/day) initiated at surgery dose-dependently reduced interstitial collagen deposition (measured by picrosirius red staining) and the expression of the myofibroblast marker smooth muscle α-actin (αSMA). [1] BI-749327 treatment also reduced UUO-induced renal expression of Tgfb1, Col4a1, and Fn1 mRNA, decreased T-cell infiltration (CD3+ staining), and reduced the number of interstitial lesions. [1] |
| Enzyme Assay |
A whole-cell patch clamp electrophysiology assay was used to determine the inhibitory potency of BI-749327 on TRPC6 channels. HEK293 cells were transfected to express mouse, human, or guinea pig TRPC6. Currents were activated by the diacylglycerol analog OAG (1-oleoyl-2-acetyl-sn-glycerol). Cells were voltage-clamped, and currents were recorded in response to a voltage ramp protocol (from -80 mV to +80 mV). BI-749327 was applied, and the concentration-dependent inhibition of the OAG-activated current was measured to calculate IC₅₀ values. [1] |
| Cell Assay |
In Vitro Cellular Assays.[1] Neonatal rat ventricular myocytes were isolated from newborn Sprague-Dawley rat pups and stimulated with angiotensin II followed by vehicle or BI 749327 treatment. HEK293T cells were cultured to 70% confluence, transfected with plasmids expressing a NFAT luciferase reporter and another expressing WT-TRPC6, gain-of-function TRPC6 mutants (P112Q, M132T, R175Q, R895C, and R895L), or empty vector pcDNA3.1. Renilla-luciferase plasmid was transfected as an internal control. For NFAT-luciferase reporter assays, HEK293T cells were cultured and transfected at 70% confluence with plasmids expressing an NFAT-responsive luciferase reporter, a Renilla-luciferase internal control plasmid, and either wild-type TRPC6, gain-of-function TRPC6 mutants (P112Q, M132T, R175Q, R895C, R895L), TRPC3, TRPC7, or an empty vector control. After transfection, cells were treated with BI-749327 or vehicle. Luciferase activity was measured, normalized to Renilla luminescence, and expressed as fold-change relative to control. [1] For primary cardiomyocyte assays, neonatal rat ventricular myocytes were isolated from Sprague-Dawley rat pups. Cells were stimulated with angiotensin II (100 nM) and co-treated with BI-749327 or vehicle for 48 hours. RNA was then extracted, and mRNA levels of target genes (Trpc6, Rcan1, Nppa, Nppb, Myh7) were quantified by real-time PCR, normalized to Gapdh. [1] |
| Animal Protocol |
Animal/Disease Models: C57BL/6J mice[1] Doses: 30 mg/kg/day Route of Administration: po (oral gavage) Experimental Results: The left ventricular function of mice under sustained stress was improved, the volume/mass ratio was diminished, and profibrotic genes and interstitial fibrosis expression attenuated by overload. Animal/Disease Models: CD-1 mice[1] Doses: 3 mg/kg, 10 mg/kg, 30 mg/kg Route of Administration: Oral Experimental Results: t1/2 8.5-13.5 hrs (hrs (hours)) Pharmacokinetic Studies.[1] Pharmacokinetics of BI 749327 were investigated in male CD-1 mice at dose levels of 3, 10, and 30 mg/kg and at 30 mg/kg in B6129F1 mice. Serial blood sampling was performed via puncture of the saphenous vein and samples were collected using EDTA-coated microtainers. In Vivo Pressure-Overload Model.[1] Mice received daily gavage with methyl-cellulose/Tween-80 vehicle starting 1 wk before TAC surgery. After 1 wk post-TAC, mice were orally gavaged with daily vehicle or BI 749327 treatment. Cardiac function was assessed by serial echocardiography followed by terminal pressure-volume analysis after 8 wk post-TAC. In Vivo Unilateral Ureteral Obstruction Model.[1] A midline laparotomy was performed and the left ureter was isolated and ligated at the boundary of the lower renal pole to induce irreversible UUO. For the cardiac pressure overload model, C57BL/6J mice underwent transverse aortic constriction (TAC) or sham surgery. All mice received vehicle (methylcellulose/Tween-80) by daily oral gavage for one week prior to surgery for acclimation. One week after TAC, mice were randomized to receive either vehicle or BI-749327 (30 mg/kg/day) via daily oral gavage for an additional 7 weeks. Cardiac function was assessed weekly by echocardiography. Terminal hemodynamic assessment was performed via left ventricular pressure-volume catheterization at the study endpoint. [1] For the renal fibrosis model, mice underwent unilateral ureteral obstruction (UUO) surgery. BI-749327 treatment (3, 10, or 30 mg/kg/day) or vehicle was initiated immediately post-surgery and administered daily by oral gavage. Kidneys were harvested 10-14 days post-surgery for histological and molecular analysis. [1] For pharmacokinetic studies, BI-749327 was formulated and administered orally to CD-1, B6129F1, or C57BL/6J mice at specified doses. Blood samples were serially collected via the saphenous vein into EDTA-coated tubes for plasma drug concentration analysis. [1] |
| ADME/Pharmacokinetics |
To test the efficacy of BI 749327 in vivo, we first performed single dose pharmacokinetic (PK) experiments to guide the studies. Oral administration of 3, 10, or 30 mg/kg BI 749327 to CD-1 mice led to a dose-proportional increase of maximum plasma concentrations (Cmax) and total systemic exposure (SI Appendix, Fig. S2A and Table S3). The long terminal half-life (t1/2) of 8.5–13.5 h qualified the compound for once daily oral dosing. Oral administration of 30 mg/kg BI 749327 to B6129F1, CD-1, and C57BL/6J mice yielded comparable exposures (SI Appendix, Fig. S2). These datasets were then used to develop a two compartment PK model to simulate multiple dosing. Mouse plasma protein binding of BI 749327 measured by equilibrium dialysis was high at 98.4 ± 0.1% (at a concentration of 1 µM) with a corresponding unbound fraction of 1.6%. Given this, the trough unbound concentration was ∼180 nM using daily 30 mg/kg oral dosing, 10× the IC50 for TRPC6, and 1/5–1/6 the IC50 for TRPC7 and TRPC3.[1] Following single oral administration to CD-1 mice, BI-749327 (3, 10, 30 mg/kg) showed dose-proportional increases in maximum plasma concentration (Cₘₐₓ) and total systemic exposure (AUC). [1] The terminal half-life (t₁/₂) ranged from 8.5 to 13.5 hours in mice, supporting once-daily oral dosing. [1] Plasma protein binding in mice, measured by equilibrium dialysis at 1 μM, was high (98.4 ± 0.1%), resulting in an unbound fraction of 1.6%. [1] With a daily oral dose of 30 mg/kg in mice, the simulated trough unbound plasma concentration was approximately 180 nM. [1] |
| Toxicity/Toxicokinetics |
Daily oral administration of BI-749327 at 30 mg/kg/day for up to 8 weeks in mice with pressure overload did not significantly alter mean arterial pressure or heart rate in conscious animals. [1] The article does not report specific in vitro or in vivo toxicity data such as lethal dose, organ toxicity (beyond efficacy-related histology), drug-drug interactions, or detailed toxicokinetic parameters. [1] |
| References |
[1]. In vivo selective inhibition of TRPC6 by antagonist BI 749327 ameliorates fibrosis and dysfunction in cardiac and renal disease. Proc Natl Acad Sci U S A. 2019 May 14;116(20):10156-10161. |
| Additional Infomation |
Thisstudy has some limitations. First, evidence of target engagement by BI 749327 remains indirect, leveraging biomarkers engaged by the pathway. For the kidney, proof may require further cell-specific gene expression analysis. We did not test each disease model in TRPC6 KO mice to assess selectivity, as these models themselves alter the disease condition: TRPC6 gene deletion occurs before disease and up-regulation of other channels may compensate. The TAC cardiac model could not test if TRPC6 antagonism reverses lung congestion, a potential consequence of preload decline, as it modeled partially compensated hypertrophy and not heart failure. The UUO model generates interstitial fibrosis but not glomerular disease or proteinuria. Future studies in models that engage these abnormalities, such as diabetic nephropathy and other renal diseases, will be needed to test the therapeutic potential of BI 749327 for these conditions. Mice expressing gain-of-function TRPC6 mutations causing severe renal damage in humans rarely develop much disease, so such testing may ultimately be best done in humans.[1] In summary, we reveal in vivo evidence of therapeutic efficacy of an orally bioavailable TRPC6 antagonist to counter chronic profibrotic disease conditions. In addition to cardiac and renal disease, TRPC6 hyperstimulation is linked to important pathophysiology in the lung and in other tissues, and the favorable oral profile of BI 749327 supports exploration of its therapeutic role in these organs as well.[1] BI-749327 was identified through a high-throughput screen and subsequent medicinal chemistry optimization to achieve nanomolar potency, high selectivity (particularly over TRPC3 and TRPC7), and pharmacokinetic properties suitable for once-daily oral dosing with low peak-to-trough exposure ratios. [1] It is an orally bioavailable pharmacological tool that allows for the inhibition of TRPC6 in adult animals after disease onset, contrasting with genetic knockout models. [1] Its mechanism of therapeutic action is linked to the inhibition of TRPC6-mediated calcium influx, which suppresses the calcineurin-NFAT signaling pathway, a key driver of pathological gene expression, myofibroblast activation, and fibrosis in the heart and kidney. [1] The reduction in cardiac chamber volumes observed with BI-749327 treatment, independent of changes in contractility, suggests potential systemic effects on venous tone or circulatory volume, which could be beneficial in heart failure with congestion. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~31.25 mg/mL (~70.63 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.70 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.70 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.70 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2602 mL | 11.3012 mL | 22.6024 mL | |
| 5 mM | 0.4520 mL | 2.2602 mL | 4.5205 mL | |
| 10 mM | 0.2260 mL | 1.1301 mL | 2.2602 mL |