BD1047 dihydrobromide (BD-1047) is a selective functional antagonist of sigma receptors (σ receptors) with antipsychotic activity and may be used for schizophrenia. BD 1047 is demonstrated to attenuate the dystonia produced by the high affinity σ receptor ligands di-o-tolylguanidine and Haloperidol. BD 1047 is described to have similar activity at the σ-1 receptor to BD 1063 (sc-203838) and to show higher affinity at σ-2 than BD 1063. BD 1047 is demonstrated to antagonize the effects of Trazodone on neurotransmitter release. BD 1047 did not decrease amphetamine-induced hyperactivity in mice in a statistically significant manner. Likewise, it did not modify the hyperactivity induced by NMDA receptor antagonists, phencyclidine, memantine or dizocilpine. On the other hand, BD 1047 attenuated apomorphine-induced climbing in mice and phencyclidine-induced head twitches in rats, like rimcazole and panamesine did. Summing up, BD 1047 shows a moderate activity in models used in this study suggesting that its usefulness as an antipsychtic drug is doubtful. However, more detailed studies are required for definitive confirmation of this conclusion.
Physicochemical Properties
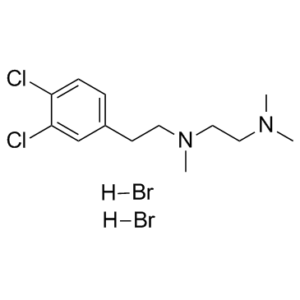
| Molecular Formula | C13H20CL2N2.2HBR | |
| Molecular Weight | 437.04 | |
| Exact Mass | 433.953 | |
| Elemental Analysis | C, 35.73; H, 5.07; Br, 36.57; Cl, 16.22; N, 6.41 | |
| CAS # | 138356-21-5 | |
| Related CAS # | 138356-20-4; 138356-21-5 (HBr); | |
| PubChem CID | 45073418 | |
| Appearance | White to off-white solid powder | |
| LogP | 4.945 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 2 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 19 | |
| Complexity | 212 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | CN(C)CCN(C)CCC1=CC(=C(C=C1)Cl)Cl.Br.Br |
|
| InChi Key | WOALTFHGLDVJHK-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C13H20Cl2N2.2BrH/c1-16(2)8-9-17(3)7-6-11-4-5-12(14)13(15)10-11;;/h4-5,10H,6-9H2,1-3H3;2*1H | |
| Chemical Name | N'-[2-(3,4-dichlorophenyl)ethyl]-N,N,N'-trimethylethane-1,2-diamine;dihydrobromide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Sigma 1 receptor |
| ln Vitro |
Cutamesine lowers the rate of cell death brought on by light exposure in mouse photoreceptor-derived 661w cells when BD-1047 (dihydrobromide) is administered[2]. Cutamesine lowers the elevated level of caspase 3/7 activity and mitochondrial damage, but BD-1047 (dihydrobromide) attenuates this effect[2].
Cutamesine reduced cell damage induced by light exposure in 661W cells [2] We examined whether the cutamesine protected the 661W cells against light-induced cell death. Representative photographs of the Hoechst 33342 and PI staining of the 661W cells pretreated with cutamesine and/or BD-1047 are shown in Fig. 1A. Hoechst 33342 stains all cells (both live and dead), whereas PI stains only dead cells. Pretreatment with 10 μM cutamesine protected against light-induced cell death (Fig. 1B), and the protective effect significantly disappeared after treatment with BD-1047, a sigma-1 receptor antagonist, at 1 μM (Fig. 1C). Cutamesine prevented the disruption of mitochondrial membrane potential [2] We investigated whether cutamesine might prevent the disruption of the mitochondrial membrane potential. Healthy cells were detected with JC-1 J-aggregates (red fluorescent cells) and apoptotic or unhealthy cells were detected with JC-1 monomers (green fluorescent cells) (Fig. 3A). Light irradiation increased the numbers of green fluorescent 661W cells, indicating a disruption of the mitochondrial membrane potential. Cutamesine significantly reduced the number of green fluorescent cells, and v significantly inhibited the effect of cutamesine (Fig. 3B). Cutamesine prevented caspase-3/7 activation induced by light exposure [2] To investigate the effect of cutamesine on caspase-3/7 activation induced by light irradiation, we measured caspase-3/7 activity using the Caspase-Glo 3/7 Assay System. Light irradiation significantly increased caspase-3/7 activity (Fig. 4). Cutamesine at 10 μM significantly reduced the activation of caspase 3/7, and this effect was attenuated by BD-1047 at 1 μM (Fig. 4). |
| ln Vivo |
At a dose of 10 mg/kg, BD-1047 (dihydrobromide) (1–10 mg/kg; ip) reduces the climbing behavior in mice induced by apomorphine (APO)[1]. ?The antidepressant-like effect caused by co-administration of pramipexole and sertraline (but not pramipexole and fluoxetine) is countered by BD-1047 (dihydrobromide)[3]. ?Dihydrobromide, or BD-1047, reverses the potentiation of Sig-1 R agonists in NMDA-induced pain behavior and pNR1 immunoreactivity, as well as the increasing expression of pNR1[4].
The sigma receptors were first classified as a subtype of opioid receptors but later they were found to be a distinct pharmacological entity. Many preclinical and clinical data have indicated that sigma receptor ligands have to be involved in neuropsychiatric disorders, including schizophrenia. Numerous data have suggested that potential antipsychotic activity of sigma ligands results from their "antagonistic" activity. However, the subcellular mechanisms by which sigma ligands exert their effects have not been elucidated in detail, therefore, the terms "agonist" or "antagonist" and their functional implications are not entirely unequivocal. The aim of the present study was to find out if BD-1047, described recently as a selective functional antagonist of sigma receptors, shows antipsychotic activity in animal models predictive of efficacy in schizophrenia. In contrast to rimcazole and panamesine, two selective sigma ligands whose antipsychotic activity was confirmed clinically, BD-1047 did not decrease amphetamine-induced hyperactivity in mice in a statistically significant manner. Likewise, it did not modify the hyperactivity induced by NMDA receptor antagonists, phencyclidine, memantine or dizocilpine. On the other hand, BD 1047 attenuated apomorphine-induced climbing in mice and phencyclidine-induced head twitches in rats, like rimcazole and panamesine did. Summing up, BD-1047 shows a moderate activity in models used in this study suggesting that its usefulness as an antipsychtic drug is doubtful. However, more detailed studies are required for definitive confirmation of this conclusion. [1] Progesterone and BD-1047 (a sigma(1) receptor antagonist) counteracted the antidepressant-like effect induced by co-administration of pramipexole and sertraline (but not pramipexole and fluoxetine). In that test, active behavior did not reflect the increases in general activity, since combined administration of pramipexole and fluoxetine or sertraline failed to enhance the locomotor activity of rats. None of the tested drugs (SCH 23390, sulpiride, S 33084, WAY 100635, BD-1047 and progesterone) - alone or in combination with pramipexole and fluoxetine or sertraline - changed locomotor activity. The results described in the present paper indicate that co-administration of pramipexole and fluoxetine or sertraline may induce a more pronounced antidepressive activity than does treatment with pramipexole alone, and that in addition to other mechanisms, dopamine D(2/3) and 5-HT(1A) receptors may contribute to the antidepressant-like activity of pramipexole and fluoxetine or sertraline in the forced swimming test in rats. Moreover, sigma(1) receptors may constitute one of the possible mechanisms by which co-administration of pramipexole and sertraline induces antidepressant-like activity in that test. [3] |
| Cell Assay |
Light-induced cell death model in 661W cell cultures [2] The 661W cells were seeded at a density of 3 × 103 cells per well in 96-well plates, and then incubated for 24 h under a humidified atmosphere of 5% CO2 at 37 °C. Then, they were treated with 1 or 10 μM cutamesine and/or 1 μM N-[2-(3,4-dichlorophenyl)ethyl]-N-methyl-2-(dimethylamino)ethylamine (BD-1047) dihydrobromide and incubated for 1 h under a humidified atmosphere of 5% CO2 at 37 °C. The cells were exposed to 2500 lux of white fluorescent light for 24 h with each agent under a humidified atmosphere of 5% CO2 at 37 °C. The dark control cells and light-irradiated 661W cells were all from the same stock, eliminating any preexisting bias (such as light and temperature), as previously described by Kanan et al. (2007). |
| Animal Protocol |
Animal/Disease Models: Male Albino Swiss mice (50 days old, 25–28 g)[1] Doses: 1 mg/kg, 3 mg/kg, 10 mg/kg Route of Administration: intraperitoneal (ip)injection Experimental Results: diminished the APO-induced climbing at the dose of 10 mg/kg in mice. Exposure to visible light [2] The mice were kept in dark conditions for 24 h prior to light exposure for dark adaptation. The pupils of the mice were dilated using 1% cyclopentolate hydrochloride eye drops 30 min prior to exposure to light. The mice, which were not anesthetized, were exposed to visible light (8000 lux) emitted by white fluorescent lamps for 3 h in cages with reflective interiors. The temperature during exposure to light was maintained at 25 ± 1.5 °C. After the exposure to light, all of the mice were returned to darkness for 24 h and then kept under normal light/dark cycling conditions. Cutamesine (50 or 500 μM, injected volume; 2 μL), BD-1047 (500 μM), cutamesisne (500 μM) plus BD-1047 (500 μM), or PBS was injected into the intravitreal space 1 h prior to the light exposure. The concentrations of cutamesine in the vitreous body just after the intravitreal administration at 50 and 500 μM were estimated to be approximately 10 and 100 μM, respectively. |
| References |
[1]. Effect of BD 1047, a sigma1 receptor antagonist, in the animal models predictive of antipsychotic activity. Pharmacol Rep. 2006 Sep-Oct;58(5):626-635. [2]. Effect of a sigma-1 receptor agonist, cutamesine dihydrochloride (SA4503), on photoreceptor cell death against light-induced damage. Exp Eye Res. 2015 Mar;132:64-72. [3]. Mechanism of synergistic action following co-treatment with pramipexole and fluoxetine or sertraline in the forced swimming test in rats. Pharmacol Rep. 2006 Jul-Aug;58(4):493-500. |
| Additional Infomation |
Next, we investigated the effect of cutamesine on the disruption of the mitochondrial membrane potential. Cutamesine recovered the disruption of the mitochondrial membrane potential, and BD-1047 inhibited this effect of cutamesine. These results suggest that cutamesine activated the sigma-1 receptor and enhanced its function to restore abnormal mitochondrial Ca2+ as well, as previously reported by another group (Tagashira et al., 2013). Therefore, the protective effect of cutamesine against light-induced damage might be dependent on recovering mitochondrial function. The activation of caspase-3, a cell death effector, was reported at the photoreceptors in retinal degeneration model animals (Jomary et al., 2001). Abnormal mitochondrial Ca2+ also mediates caspase-3 activation via cytochrome c release (He et al., 2000). In the present study, irradiation of white fluorescent light caused the caspase 3/7 activation in the 661W cells. Cutamesine reduced this elevated level, and this effect was prevented by BD-1047. These reports suggest that cutamesine inhibits caspase activity by recovering mitochondrial function. Finally, we investigated the protective effect of cutamesine against light-induced damage in an in vivo murine model. The intravitreal injection of 50 and 500 μM of cutamesine suppressed the light-induced damage to retinal function and histological changes in mice. The concentrations of cutamesine in the vitreous body just after the intravitreal administration were estimated to be approximately 10 and 100 μM, respectively. The protective effect was similar to the effective concentration at 10 μM in vitro. Furthermore, the protective effect of cutamesine on the light-induced damage was eliminated by co-administration with BD-1047, a sigma-1 receptor antagonist. Therefore, this effect might depend on the activation of sigma-1 receptor protein, which leads to normalized mitochondrial function and the prevention of caspase activities, as in the present in vitro study. [2] The aim of the present study was to examine the effect of combined treatment of male Wistar rats with pramipexole and fluoxetine or sertraline in the forced swimming test. The obtained results showed that co-treatment with pramipexole (0.1 mg/kg) and fluoxetine (10 mg/kg) or sertraline (5 mg/kg) (in doses inactive per se) exhibited antidepressant-like activity in the forced swimming test. Sulpiride (a dopamine D(2/3) receptor antagonist) and WAY 100635 (a 5-HT(1A) receptor antagonist), either being ineffective in the forced swimming test, inhibited the antidepressant-like effect induced by co-administration of pramipexole and fluoxetine or sertraline. However, SCH 23390 (a dopamine D(1) receptor antagonist) only partly did not alter the effect of pramipexole given jointly with antidepressant drugs; on the other hand, S 33084 (a dopamine D(3) receptor antagonist) only partly decreased (in a statistically insignificant manner) that effect. Moreover, progesterone and BD-1047 (a sigma(1) receptor antagonist) counteracted the antidepressant-like effect induced by co-administration of pramipexole and sertraline (but not pramipexole and fluoxetine). In that test, active behavior did not reflect the increases in general activity, since combined administration of pramipexole and fluoxetine or sertraline failed to enhance the locomotor activity of rats. None of the tested drugs (SCH 23390, sulpiride, S 33084, WAY 100635, BD-1047 and progesterone) - alone or in combination with pramipexole and fluoxetine or sertraline - changed locomotor activity. The results described in the present paper indicate that co-administration of pramipexole and fluoxetine or sertraline may induce a more pronounced antidepressive activity than does treatment with pramipexole alone, and that in addition to other mechanisms, dopamine D(2/3) and 5-HT(1A) receptors may contribute to the antidepressant-like activity of pramipexole and fluoxetine or sertraline in the forced swimming test in rats. Moreover, sigma(1) receptors may constitute one of the possible mechanisms by which co-administration of pramipexole and sertraline induces antidepressant-like activity in that test.[3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.72 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.72 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: 33.33 mg/mL (76.26 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2881 mL | 11.4406 mL | 22.8812 mL | |
| 5 mM | 0.4576 mL | 2.2881 mL | 4.5762 mL | |
| 10 mM | 0.2288 mL | 1.1441 mL | 2.2881 mL |