Physicochemical Properties
| Molecular Formula | C24H28CL2N6O8 |
| Molecular Weight | 599.42 |
| Exact Mass | 598.134 |
| CAS # | 1196994-12-3 |
| Related CAS # | ST-1006;1196994-11-2 |
| PubChem CID | 165437225 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 40 |
| Complexity | 502 |
| Defined Atom Stereocenter Count | 0 |
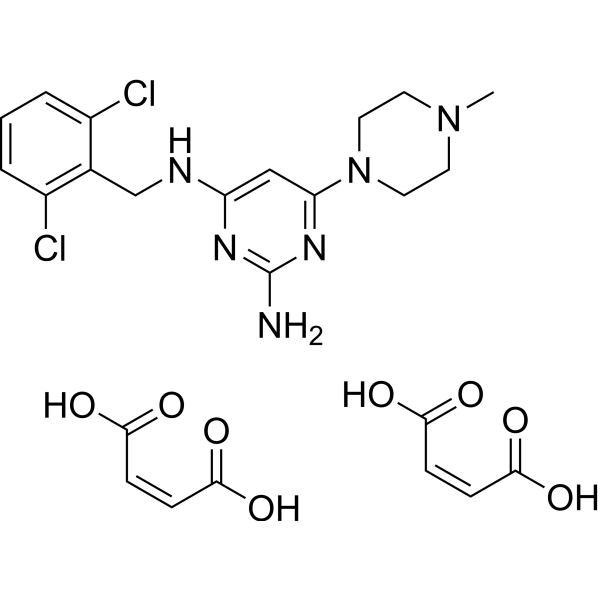
| SMILES | CN1CCN(CC1)C2=NC(=NC(=C2)NCC3=C(C=CC=C3Cl)Cl)N.C(=C\C(=O)O)\C(=O)O.C(=C\C(=O)O)\C(=O)O |
| InChi Key | NBLGNUAWAKUPSY-SPIKMXEPSA-N |
| InChi Code | InChI=1S/C16H20Cl2N6.2C4H4O4/c1-23-5-7-24(8-6-23)15-9-14(21-16(19)22-15)20-10-11-12(17)3-2-4-13(11)18;2*5-3(6)1-2-4(7)8/h2-4,9H,5-8,10H2,1H3,(H3,19,20,21,22);2*1-2H,(H,5,6)(H,7,8)/b;2*2-1- |
| Chemical Name | (Z)-but-2-enedioic acid;4-N-[(2,6-dichlorophenyl)methyl]-6-(4-methylpiperazin-1-yl)pyrimidine-2,4-diamine |
| Synonyms | ST-1006 Maleate; ST-1006 (Maleate); 1196994-12-3; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Histamine H4 receptor/H4 receptor (pKi = 7.94) |
| ln Vitro |
ST-1006 Maleate (10 μM) is a powerful inducer of basophil migration and causes basophil migration[2]. ST-1006 (0-100 μM) maleate inhibits basophil activation mediated by FceRI and lowers the expression levels of CD63 and CD203c on basophils triggered by FceRI[2]. We could show that highly purified basophils express the H1R, H2R, and H4R but not the H3R mRNA. Human basophils expressed higher H4R mRNA levels as compared to the expression levels of the H1R (P < 0.01). Histamine and the H4R agonist ST-1006 initiated active migration of basophils (P < 0.001). A significant reduction in FcεRI cross-linking-mediated surface expression of CD63 and CD203c was observed on basophils after pre-incubation with histamine or the specific H4R agonist ST-1006 (P < 0.01). The synthesis and release of sulfidoleukotrienes from basophils after activation with different stimuli, by FcεRI cross-linking or by stimulation with hymenoptera venom allergens, were significantly reduced by histamine or the H4R agonist ST-1006 (P < 0.05-0.001). Conclusion: These data imply that the H4R regulates IgE-dependent processes in human basophils and provides a novel function of the H4R preventing an overwhelming immune reaction by engagement of a negative feedback loop. [2] |
| ln Vivo |
ST-1006 (1-100 mg/kg; sc; male CD-1 mice) maleate exhibits antipruritic and anti-inflammatory properties[1]. The anti-inflammatory effects of histamine H4 receptor (H4R) antagonists opened new therapeutic options for the treatment of inflammatory/allergic diseases, but the role of H4R in inflammation is far from being solved. Aim of the present study was to investigate the role of structurally related H4R ligands of the aminopyrimidine class with different efficacies and functionalities (neutral antagonist ST-994, partial agonist ST-1006, inverse agonist ST-1012, and partial inverse agonist ST-1124) on croton oil-induced ear edema and pruritus in mice. The H4R ligands were administered subcutaneously before topical application of croton oil. While ST-1006 and ST-1124 were ineffective at any dose tested (10-100 mg/kg), both ST-994 and ST-1012 (30 and 100 mg/kg) significantly reduced croton oil-induced ear edema. Moreover, ST-994, ST-1006, and ST-1124, but not ST-1012, significantly inhibited croton oil-induced ear pruritus at 30 mg/kg. In accordance with results obtained with the reference H4R antagonist JNJ7777120 (100 mg/kg), histological examination of inflamed ear tissue indicated that treatment with ST-994 (30 mg/kg) led to a significant reduction in the inflammatory severity score and in the number of eosinophils infiltrating the tissue, while the number of degranulated mast cells in inflamed tissues was increased in comparison with the number of intact mast cells. These data indicate that croton oil-induced ear inflammation and pruritus seem to be clearly, but variably, affected by the H4R ligands tested. The potential advantage of dual effect of the H4R neutral antagonist ST-994 has to be carefully considered as a new therapeutic approach to the treatment of inflammatory diseases. |
| Cell Assay |
Basophil activation test [2] Surface expression of the basophil activation markers CD63 and CD203c was measured using multicolor flow cytometry. Briefly, 100 μl of whole blood from healthy subjects was diluted in 100 μl stimulation buffer consisting of PBS supplemented with IL-3 (2 ng/ml). Cells were pre-incubated for 15 min at 37°C with stimulation buffer only, histamine (10 μM) or the H4R agonist ST-1006 (10 μM). For dose kinetic experiments, the cells were stimulated with different concentrations of histamine or the H4R agonist (0.1–100 μM). For the time kinetic experiments, the cells were pre-incubated for different periods of time including 15, 10, 5, and 0 min with histamine or the H4R agonist before the anti-IgE antibody was added for 20 min. To prove that the effects were specific for the H4R, a pre-incubation of the cells with the H4R antagonist JNJ7777120 for 30 min preceded histamine or H4R stimulation. Thereafter, the cells were activated for 20 min by adding the monoclonal anti-human IgE antibody which binds to the ε-chain of human IgE. Degranulation was stopped by chilling the cells on ice for 5 min. The cells were stained with FITC-conjugated anti-CD63, phycoerythrin-conjugated anti-CD203c, and PerCP-conjugated anti-FcεRI. Erythrocytes were removed by a lysing reaction, and the cells were analyzed by flow cytometry. Sulfidoleukotriene ELISA [2] A commercially available cellular antigen stimulation test ELISA was used to quantify the release of sulfidoleukotrienes including leukotriene C4 (LTC4) and its derivates LTD4 and LTE4, produced by isolated leukocytes upon contact with allergens or stimuli which fosters the degranulation. Isolation of leukocytes, cell stimulation, and leukotriene determination were performed according to the protocol of the supplier. Whole blood was collected from patients with a history of allergic diseases or from hymenoptera venom-sensitized patients. In brief, leukocytes were separated from whole blood by density gradient centrifugation on dextran. The isolated leukocytes were resuspended in stimulation buffer containing IL-3. Each sample was tested for the basal release and for the release after stimulation with different stimuli. To activate the basophils, the cells were stimulated for 40 min at 37°C (i) with an antibody directed against the high-affinity IgE receptor or (ii) with a mixture of C5a (0.1 μM), formyl-methionyl-leucyl-phenylalanine (FMLP) (1 μM), and platelet-activating factor (PAF) (10 μM). To test the allergen-dependent degranulation of basophils, the cells were incubated for 40 min at 37°C with bee and wasp venom (200 μg/ml). Histamine (10 μM), the H4R agonist ST-1006 (10 μM), the H1R agonist 2-pyridylethylamine (10 μM), and the H2R agonist amthamine (10 μM) were added simultaneously with the activating stimuli to all approaches as indicated. To show the concentration dependency, the cells were stimulated with different concentrations (0.1–100 μM) of the H2R agonist amthamine or the H4R agonist ST-1006. For the time kinetic experiments, the cells were pre-incubated for different periods of time including 10, 5, and 0 min with the H2R agonist or the H4R agonist each 10 μM before the wasp venom allergen was added for 40 min. Finally, the cells were centrifuged and the supernatants were collected. Sulfidoleukotriene release was estimated using the CAST ELISA according to the manufacturer's instructions. |
| Animal Protocol |
Animal/Disease Models: Male CD-1 mice with pruritus (8-10 weeks and 25-30 g)[1] Doses: 1-100 mg/kg Route of Administration: subcutaneous (sc) injection Experimental Results: Had an antipruritic effect at the non-anti-inflammatory dose of 30 mg/kg. Edema [1] The technique originally described by Tubaro et al. (1986) and modified by Coruzzi et al. (2012) was followed. All experiments were carried out between 10 a.m. and 3 p.m., in order to avoid the influence of circadian variations in corticosteroid levels in the inflammatory responses. Cutaneous inflammation was induced in conscious mice by topical application of croton oil (2.5% in acetone). The irritant agent was applied with a micropipette (20 μl/ear) to the inner surface of the right ear. Acetone (20 μl/ear) was applied to the left ear, which served as a control (Tubaro et al. 1986). The H4R ligands ST-994, ST-1006, ST-1012, and ST-1124 (10–100 mg/kg); the reference H4R antagonist JNJ7777120 (30 and 100 mg/kg); or the vehicle were administered as single subcutaneous (s.c.) injection immediately before the topical application of croton oil. Control mice received s.c. injection with vehicle (20% DMSO and 80% 2-hydroxypropyl-β-cyclodextrin). Two or four hours after croton oil application, mice were sacrificed by cervical dislocation; both right (croton oil in acetone) and left (acetone) ears were removed by cutting horizontally across the indentation at the base of the ear. For each mouse, the extent of the edema was expressed as the difference in weight (Δ mg) between right (inflamed) and left (uninflamed) ear. Pruritus [1] As an indicator of pruritus, the scratching behavior of mice was determined (Fujii et al. 2010; Coruzzi et al. 2012). Single s.c. administration of vehicle, ST-994, ST-1006, ST-1012, or ST-1124 (30 mg/kg) was given immediately before croton oil application. Itch was measured by blinded counting of the number of scratching bouts in the 1-h period immediately following the application of croton oil. A bout of scratching was defined as three or more individual rapid scratch movements with the hind paw around the inflamed ear (Dunford et al. 2007). Hind paw movements directed away from the inflamed ear and grooming movements were not counted. Pruritus was evaluated as a difference in counting the scratching bouts between right (inflamed) and left (uninflamed) ear at 10-min intervals (time course, Δ scratching bouts/10 min) or as a difference in counting the total scratching bouts between right (inflamed) and left (uninflamed) ear over the 1-h observation period (Δ scratching bouts/1 h). Histology [1] For histological analysis, inflamed ears from vehicle-, ST-994-, ST-1006-, ST-1012-, ST-1124-, JNJ7777120-, or dexamethasone-treated CD-1 mice were removed 2 h after croton oil application, divided into two tissue samples, and fixed overnight in 10% formalin (Coruzzi et al. 2012). Subsequently, tissue samples were embedded longitudinally in paraffin, cut with a microtome into 4-μm sections, and mounted on glass slides. The cross-sections were stained with hematoxylin and eosin for the evaluation of inflammation intensity (total severity score) and the count of eosinophils. Each section was scored by two observers unaware of the treatment, according to the following score system: 0 = no inflammatory cells; 0.5 = rare leucocytes in the interstitium; 1 = rare leucocyte clusters infiltrating perivascular spaces (2–5 per section); 2 = leucocyte clusters infiltrating perivascular spaces (6–10 per section); and 3 = leucocyte clusters infiltrating the interstitium. The total severity score was then determined in the inflamed ear. The maximum severity score was six. Moreover, in the hematoxylin and eosin-stained cross-sections, the number of eosinophils was counted in ten randomly fields per inflamed ear and the total number was considered (number of eosinophils/ear). |
| References |
[1]. Differential effects of functionally different histamine H4 receptor ligands on acute irritant dermatitis in mice. Naunyn Schmiedebergs Arch Pharmacol. 2018 Dec;391(12):1387-1397. [2]. Human basophil chemotaxis and activation are regulated via the histamine H4 receptor. Allergy. 2016 Sep;71(9):1264-73. |
| Additional Infomation |
Results of the present study indicate that croton oil-induced ear inflammation and pruritus in CD-1 mice seem to be clearly, but variably, affected by the structurally related H4R ligands tested (Sander et al. 2009; Kottke et al. 2011; Gschwandtner et al. 2013). Subcutaneous injections of both the hH4R neutral antagonist ST-994 and the hH4R full inverse agonist ST-1012 inhibited croton oil-induced ear edema at any time point considered (2 and 4 h), while both the hH4R partial agonist ST-1006 and the hH4R partial inverse agonist ST-1124 were inactive. Croton oil-induced itch was not modified by ST-1012 at the anti-inflammatory dose of 30 mg/kg s.c., whereas the anti-inflammatory effect of ST-994 was associated to its antipruritic properties. Surprisingly, both ST-1006 and ST-1124 reduced croton oil-induced scratching bouts at a dose of 30 mg/kg s.c. that did not affect croton oil-induced ear edema, giving further evidence for the complex regulation behavior: (1) H4R blockade induced by ST-994 did not completely overcome results seen with the reference H4R antagonist JNJ7777120 as ST-994 reduced both croton oil-induced ear edema and pruritus while JNJ7777120 reduced inflammation but not scratching bouts (Coruzzi et al. 2012); (2) the anti-inflammatory effect of ST-994 is not explained by the antipruritic effect, since ST-1012 reduced croton oil-induced ear edema but not itch; (3) both ST-1006 and ST-1124 did not possess proinflammatory or anti-inflammatory effects in this model of acute inflammation when s.c. injected, but reductions of croton oil-induced pruritus indicate that their ineffectiveness depends on the parameter considered; and (4) differently from the hH4R neutral antagonist ST-994, the hH4R full inverse agonist ST-1012 (100 mg/kg s.c.) reduced only the ear edema developing at 2 h after challenge, whereas the late inflammatory response (at 4 h) was not affected; a possible explanation could be related to an unfavorable kinetics, but the effectiveness of ST-1012 (30 mg/kg s.c.) at 2 and 4 h after croton oil application minimizes this hypothesis. Taken together, the present and literature data suggest that the effects of H4R ligands are highly dependent on the strain (Coruzzi et al. 2012) and the specific model and/or parameter considered (present study). The anti-inflammatory effect of ST-994 or ST-1012 is in accordance with the H4R blockade in different settings in mice (Cowden et al. 2010; Coruzzi et al. 2012; Lucarini et al. 2016), but different from the ineffectiveness of JNJ7777120 on ear swelling induced by the two haptens, 2,4-dinitrochlorobenzene (Rossbach et al. 2008) or 2,4,6-trinitrochlorobenzene (Seike et al. 2010). Several factors may be responsible for these contrasting effects, including the type of chemical irritant, the presence/absence of a prior sensitization or the different cytokine profile involved, the use of different strains of mice and, in the same strain, the assay employed (Coruzzi et al. 2012) and the possibility of different pharmacokinetics of the different compounds.
At 2 h, both ST-994 and ST-1012 induced ear edema reductions as effectively as inhibitions induced by JNJ7777120 or the first-generation H1R blocker pyrilamine suggesting that both H1Rs and H4Rs are involved almost in the early phase of the inflammation. Even more interesting is the evidence that pyrilamine abolished croton oil-induced pruritus, while JNJ7777120 induced only a nonsignificant reduction (Coruzzi et al. 2012; Adami et al. 2014). In the same model of acute skin inflammation, the second-generation H1R antagonist cetirizine, that differs from first generation because of its high specificity and affinity for peripheral H1Rs, scarcely reduced ear edema and pruritus (Adami et al. 2014) thus opening the question if central penetration of the compound is necessary for both anti-inflammatory effect and/or antipruritic activity. Differences in the ability of antihistamines to interact with P-glycoprotein (P-gp) efflux pump at the blood-brain barrier (BBB) may determine their central nervous system (CNS) penetration (Chen et al. 2003; Obradovic et al. 2007) and as a consequence the presence or absence of central side effects. Sedating H1R antagonists are not P-gp substrates, while some non-sedating H1R antagonists are P-gp substrates: affinity for P-gp at BBB may explain the lack of CNS side effects of the second-generation H1R antagonists (Chen et al. 2003). The lack of effect on croton oil-induced ear edema by ST-1006 or ST-1124, together with their effectiveness in diminishing croton oil-induced itch, is surprising and even paradoxical for ST-1006 considering its pruritogen-evoked scratching after intradermal injection into the back skin of female BALBc mice (Rossbach et al. 2014). This paradoxical result is however in agreement with reductions of local reactions and pruritus in atopic dermatitis patients induced by intracutaneous injections of histamine (Heyer et al. 1998). Despite differences in strain can markedly influence scratching responses to histamine (Inagaki et al. 2001), it has to underline that the previously reported high susceptibility of NMRI mice to H4R-induced itching was not correlated to higher H4R densities in this mouse strain (Bäumer et al. 2008). Thus, any conclusion should be taken with caution because it may depend on the prototypical H4R ligand used, not neglecting its different efficacies in different cell/disease states (Gbahou et al. 2003; Sivertsen et al. 2013).[1] As life-threatening anaphylactic reactions to bee or wasp stings occur in 0.8–5% of the general population, we focussed in the main part of the study on the role of histamine and the H4R in this scenario, using tools which are used in the daily routine diagnostic of insect venom allergy. We could show the capability of histamine and the H4R agonist ST-1006 to suppress the FcεRI-induced expression of the activation markers CD63 and CD203c on the cell surface of basophils in a dose- and time-dependent manner by means of flow cytometry. Moreover, when using leukotriene secretion as an indicator of basophil degranulation, we could demonstrate that the synthesis and release of sulfidoleukotrienes from effector cells after activation with different stimuli or by FcεRI cross-linking were markedly reduced by selective stimulation with histamine and the H4R agonist ST-1006 at the same time period. In addition, we showed that the stimulation with histamine or the H4R agonist ST-1006 alone did not alter the basal degranulation in human basophils. These observations are consistent with Hofstra et al. who showed that also murine bone marrow mast cells were not affected by stimulation with histamine alone in terms of leukotriene and prostaglandin release. Several immunologic effects of histamine during basophil or mast cell activation in both in vitro assays and in the course of allergen immunotherapy were described in the literature. Former studies provided evidence that histamine is able to inhibit its own release in human leukocytes or mast cells. It was shown in one of these early studies that histamine and different H2R agonists inhibit the histamine release from human leukocytes with high potency. This effect was specifically blocked by an H2R antagonist, indicating that the H2R is involved 17. We could show the suppression of allergen-induced release of sulfidoleukotrienes from human basophils in the presence of histamine, the selective H2R agonist amthamine, or the selective H4R agonist ST-1006, confirming these early observations with regard to the H2R and expanding them to the H4R.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6683 mL | 8.3414 mL | 16.6828 mL | |
| 5 mM | 0.3337 mL | 1.6683 mL | 3.3366 mL | |
| 10 mM | 0.1668 mL | 0.8341 mL | 1.6683 mL |